Abstract
Titanium and its alloys are conventionally used to produce medical devices, but their biocompatibility has not yet been optimized. Surface modification, especially control of the surface roughness of titanium, is one strategy for improving biocompatibility and providing effective binding to hard tissue. However, the soft tissue compatibility of metallic materials is currently poorly understood, and effective techniques for tight binding between metal surfaces and soft tissue are still under development. Therefore, we here investigated whether the surface roughness of titanium affects fibroblast adhesion and proliferation. Our results showed that a surface roughness of ∼100 nm reduces fibroblast function. On such surfaces, distinct focal adhesion was not observed. These findings improve the general understanding of the binding compatibility between soft tissues and metallic materials.
Introduction
Titanium and its alloys are used to produce medical devices because of their excellent mechanical properties and high corrosion resistance (1, 2). However, the biocompatibility of titanium has yet to be optimized. In particular, compatibility of soft tissue is one of the important biocompatibility properties of metallic materials. For penetrating devices such as dental implants, bacteria sometimes invade the body of the patient via the interface between the metal surface and soft tissues (3-4-5). Therefore, to prevent bacterial infection, improvement of titanium's soft tissue compatibility is desirable (6, 7). Fibrin is commonly used for gluing soft tissues (8); however, it is not effective for binding between solid surfaces and soft tissue.
Fibroblasts are the major cellular constituent of the connective tissue (9, 10). Hence, fibroblast attachment activity at a material's surface is closely related to its soft tissue compatibility. In general, cells are able to adhere to materials via specific interactions with constituents of the extracellular matrix (ECM), such as fibronectin, type I collagen, and laminin (11-12-13). Focal adhesions are intracellular molecular complexes formed via the binding between the ECM and integrin. These complexes generate intracellular signals associated with growth, proliferation, migration, and differentiation (14-15-16-17).
Surface roughness is also an important factor for improving the biological function of materials (18). For example, titanium that has a micrometer-scale roughened surface exhibits more cell attachment activity than titanium with smooth surface (19). In addition, several studies reported that micrometer-scale roughness also promotes the proliferation activity and differentiation of osteoblasts on given surfaces (20, 21). These rough surfaces are usually produced by methods such as alkaline and/or acid etching, and are often accompanied by variation in the chemical composition of the substrate surface (22). Variation in surface chemical properties often has negative or positive biological effects (23). Hence, discrimination between topographical effects and chemical effects on biocompatibility is difficult.
In this study, we investigated whether surface roughness at a scale of a few nanometers to 100 nm influences fibroblast function. We created surface roughness on titanium materials by using a simple method involving silicon carbide (SiC) polishing paper. This method does not affect the chemical composition of the oxide film on a substrate's surface. We investigated the effect of nanometer-scale surface roughness on fibroblast attachment activity, proliferation, and morphology. Our findings provide new insights into the soft tissue compatibility of metallic materials.
Materials and Methods
Preparation and characterization of materials
To prepare smooth surfaces, commercially sourced pure, grade 2 titanium disks (8.0 mm diameter × 2.0 mm thickness; Rare Metallic Co., Ltd., Tokyo, Japan) were mirror-polished by using SiC paper and SiO2 particles. To prepare rough surfaces, the mirror-polished titanium was repolished with SiC paper. These smooth and rough materials were termed s-Ti and r-Ti, respectively. After polishing, the disks were ultrasonically rinsed in acetone and deionized water, and then dried in a stream of nitrogen gas (99.9%). The surfaces of r-Ti and s-Ti were observed with an atomic force microscope (SPM-9600; Shimadzu, Kyoto, Japan.). A silicon probe mounted on a cantilever (NCHR-20; Nano World AG, Switzerland) was employed. Surface roughness (Ra) was calculated by examining Z-range images.
Cell culture
The r-Ti and the s-Ti were sterilized with 70% ethanol for 1 hour under UV light and then air-dried. After sterilization, these samples were set in a non-treated 48-well plate (Asahi Techno Glass Co., Ltd, Chiba, Japan). NIH/3T3 mouse embryo fibroblasts were seeded onto the materials with Dulbecco's modified Eagle medium (DMEM) (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum, 100 U/mL−1 penicillin, and 100 μg/mL−1 streptomycin. The cells were incubated at 37°C in a humidified 5% CO2 incubator. To evaluate cell attachment activity, fibroblasts were seeded at a density of 50,000 cells/cm2 and incubated. After 4 hours of incubation, non-adherent cells were removed by washing twice with phosphate buffered saline (PBS). The attached cells were quantified using the Cell Counting Kit-8 (CCK-8; Dojindo Chemical, Kumamoto, Japan). For the proliferation assay, cells were seeded onto materials at a density of 5000 cells/cm2 in DMEM. After 1, 3, 5, and 7 days of culture, the number of cells was quantified by CCK-8.
Observation of cell morphology and structure
Cell morphology and structure were observed by using a fluorescence microscope (IX-7; Olympus Co., Ltd., Tokyo, Japan). Images were captured with a CCD camera and processed with Image J (http://imagej.nih.gov/ij/). To facilitate observation, cells were seeded onto the materials at a density of 3000 cells/cm2, and after 4 hours of culture, they were stained with calcein-AM (Dojindo Chemical). The cell spreading area and circularity, in addition to the aspect ratio of cells, were calculated using Image J. To observe the actin cytoskeleton and vinculin, the cells were first fixed with 4% paraformaldehyde. After fixation, cells were treated with anti-vinculin primary antibody (Sigma-Aldrich) and then treated with FITC-labeled anti-IgG secondary antibody (Sigma-Aldrich). Finally, the cells were stained with 0.7% rhodamine/phalloidin (Cytoskeleton Inc., Denver, USA) for 1 hour and observed by fluorescence microscope.
Statistical Analysis
Five samples (n = 5) were used in each analysis. One-way ANOVA was used to examine the differences among different groups.
Results
To characterize the surface, we evaluated the surface roughness and surface area. The Ra value and surface area of each substrate are listed in Table I. The r-Ti had a Ra value of 105.6 ± 1.5 nm and a surface area of 415.3 ± 1.6 μm2. For s-Ti, Ra and surface area were 2.0 ± 1.5 nm and 400.3 ± 0.2 μm2, respectively. Figure 1 shows the atomic force microscopic images of r-Ti and s-Ti within an area of 20 × 20 μm. Non-directional scratches by the SiC paper can be observed on the surface of r-Ti.
The Ra value and surface area of r-Ti, and s-Ti

Data represent mean ± SD (n = 5).
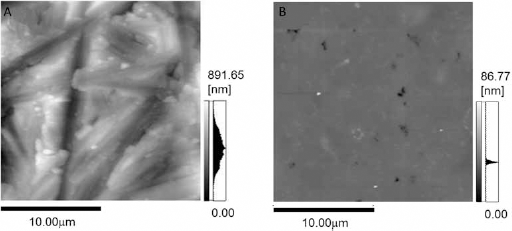
AFM topological images each substrate (
To determine the initial attachment activity, the number of cells were analyzed 4 hours after seeding on r-Ti, s-Ti, and tissue culture polystyrene (TCPS) (Fig. 2). There were no significant differences between s-Ti and TCPS; however, the initial attachment activity of r-Ti was drastically reduced compared to the other surfaces.
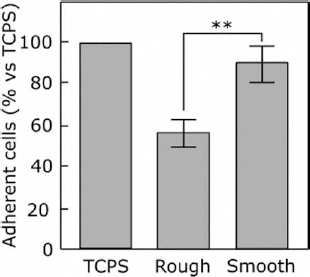
Cell attachment analysis. Attachment activity of cells cultured on the r-Ti, s-Ti, and tissue culture polystyrene (TCPS) after 4 hours of incubation. Data are mean ± SD (n = 5); ** p<0.01.
Cell morphology on each substrate was determined from microscopic images using Image J because cell morphology closely relates to cellular function. Cell adhesive area, circularity, and aspect ratio were calculated for cells cultured on r-Ti, s-Ti, and TCPS after 4 hours of incubation. The area of cells coverage was markedly increased on TCPS and s-Ti compared with r-Ti (Fig. 3). The circularity of cells cultured on r-Ti was higher than that on the other substrate (Fig. 4A), whereas the aspect ratio of cells cultured on r-Ti was the lowest among all substrates (Fig. 4B). Figure 5 shows the actin cytoskeleton and vinculin expression of cells cultured on the titanium substrates. Vinculin was detected at the tips of pseudopodia in cells cultured on s-Ti.
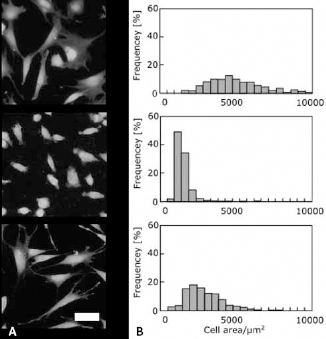
Cell spreading area analysis. Fluorescent microscopic images of cells cultured on the tissue culture polystyrene (TCPS) (top), r-Ti (middle), and s-Ti (bottom) for 4 hours (
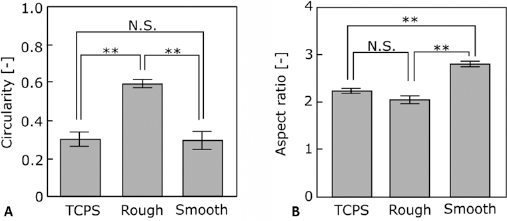
Cell shape analysis. Circularity (
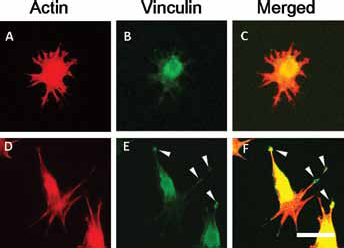
Fluorescent microscopic images. The cells cultured on the r-Ti (
To determine proliferation activity, cells were cultured on r-Ti, s-Ti, and TCPS for 1, 3, 5, and 7 days (Fig. 6). Cells cultured on TCPS showed higher proliferation activity than those on r-Ti and s-Ti. The number of cells observed on s-Ti was significantly higher than that on r-Ti from day 1 to 7 days.
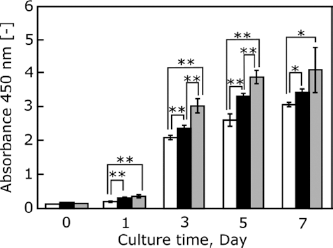
Cell proliferation analysis. Proliferation activity of cells cultured on the r-Ti (open bar), s-Ti (closed bar), and TCPS (gray bar) after 0, 1, 3, 5, and 7 days of incubation. Data are mean ± SD (n = 5). ** p<0.01, * p<0.05.
Discussion
The purpose of this study was to analyze the effects of the surface roughness of titanium on cell attachment activity. In many previous studies, preparation of a roughened surface was accompanied by a change in surface chemical composition (19-20-21). In this study, we used a silicon compound alone to adjust surface roughness, and the prepared r-Ti and s-Ti had similar chemical compositions (data not shown). By using this procedure, problems related to changing surface chemical composition were avoided and we could focus on analyzing the effects of surface roughness.
We found that the cell-spreading area markedly decreased on r-Ti in comparison to s-Ti, and analysis of cell circularity and aspect ratio indicated that cells with a round shape formed on r-Ti. Proliferation of cells cultured on the s-Ti was similar to that on TCPS. In the first few days of culture, these substrates induced higher proliferation activity than r-Ti. However, over time, the difference in proliferation activity between s-Ti and r-Ti decreased. These results suggest that a 100 nm scale surface roughness affects the initial adhesion process of cells. Vinculin, which is a component protein of focal adhesion clusters, was expressed at the tip of the pseudopodia of cells cultured on s-Ti but not in cells cultured on r-Ti. These results suggest that r-Ti does not accommodate mature cell adhesion for at least 4 hours following seeding.
Consistent with our results, osteoblasts on silicon wafers (with nanotopography produced by silica particles) previously showed decreasing proliferation activity on surfaces with 190 nm particle spacing (24). Additionally, in a study of cell adhesion on polyethylene glycol grafted surfaces with cell adhesive peptide-modified gold nanodots, Arnold et al found that a critical separation length of ≥73 nm between the adhesive dots dramatically reduced cell function because of restricted integrin clustering (25). Huang et al also reported similar results (26). Furthermore, Cai et al revealed that 1 nm-scale roughness provides no effects on biocompatibility of titanium among substrates with the same chemical composition (27). Cell attachment activity on titanium alloy, which has surface roughness of approximately 20 nm, was not significantly different from that on TCPS (28). The Ra value of the rough substrate used in this work was approximately 100 nm, and there were many undulations on the substrate's surface. Undulations of this size may inhibit cell adhesion and lead to a reduction of cellular function.
Footnotes
Financial support: This work was supported by JSPS KAKENHI Grant-in-Aid for Young Scientists (B) Grant Number 25820397.
Conflict of interest: The authors declare no conflict of interest associated with this manuscript.
