Abstract
Purpose
In this research, the effect of laser properties such as laser power and laser dwell time on the surface morphology and oxidizing of titanium have been investigated in order to enhance the bioactivity of laser textured titanium sheets.
Methods
The Ti samples were irradiated with nanosecond pulses to create the predetermined point patterns on the surface of sample sheets with specific laser parameters. Final bioactivity of the treated samples were evaluated through the use of simulated body fluid (SBF), followed by material characterization techniques such as X-ray diffraction (XRD) and energy dispersive (EDX).
Results
It was observed that by increasing the roughness of the titanium surface samples using a range of dwelling time, and with different powers, titania with higher levels of surface energy in micro/sub-micro scales are produced. The use of laser results in a one-step heat increase and the oxidation of titanium, which results in creation of titania with higher cell adhesion abilities.
Conclusions
It was concluded that the variation of the surface roughness, surface morphology, and oxidation level of the material has a direct effect on the cell adhesion rate to the surface of the titanium. Upon completion of the analysis, it is concluded that using a higher power and a lower dwelling time results in better bioactivity improvement than using higher dwelling times and lower powers.
Introduction
In recent decades, biomedical and tissue engineering have become some of the most steadily growing fields in research and development studies. The significance of these fields has increased upon their contributions to bone and tissue transplants. This treatment is one of the most promising and innovative techniques in medical studies and orthopedic surgeries, because it gives the patients the ability to eliminate problems with donor site scarcity, decrease immune system rejection, and avoid serious organ infections (1, 2). Although the ability to replace a missing organ or limb is a breakthrough achievement, this treatment is still facing essential challenges to the quality of transplants.
Micro- and nano-textured titanium implants are commonly used in scaffold systems in bone and tooth implant applications. The materials used in implants should provide the required structural properties for the implant as well as high bioactivity ability in order to avoid any infections and immune system rejection (2, 3). Bioactivity of the implants affects the healing process as well, by reducing the healing time. Having a reduced healing time is very desirable in implant applications, since the sooner the body accepts the implant organ, the sooner the user can use the function normally (3).
It must be noted that, when implanted in the body, the only portion of the tissue in touch with the cells is the surface of the structure. Hence, altering the surface properties of the materials used in implant applications is an effective method of increasing the bioactivity of those materials (3, 4). One method for altering the materials surface energy is to change its surface topography. Surface topography is generally known as the nature of a surface based on its three main characteristics: lay, roughness, and waviness. The topography and roughness properties of a material surface are very important on the surface energy and deposition rate of bone type apatite to surfaces. Increasing surface energy results in a higher bioactivity and cell adhesion rate of the implant surface. Thus, surface texturing of materials in order to enhance their bioactivity has become an important topic of study in biomedical engineering (5, 6).
There are multiple conventional methods used commercially for surface texturing of materials. The most common methods are sand blasting and machining as the mechanical methods, and acid etching and oxidation as the chemical methods. Although these methods have been used effectively to surface texture and enhance bioactivity of materials, there are major disadvantages associated with them, such as slow production time, complex control processes, and chemical contaminations (7). A method for surface texturing that has been developed in recent decades is laser surface texturing. This new technology enables the creation of patterns in scales as small as micro/nano on the surface of materials such as titanium, its alloys, and other organic and inorganic materials to increase cell adhesion (8). Further, laser surface texturing offers better control and precision, a wide range of selectivity, and more feasibility than conventional methods, and is environmentally friendly (3, 7). In comparison to conventional methods, this laser approach is more flexible and reduces processing time, making it a promising method for custom-scale manufacturing and rapid prototyping over a wide variety of applications, such as those in MEMS/NEMS and nano-biomaterials fabrication. The ideal properties of lasers make this new technology more especially desirable for synthesizing of nano-biomaterials and biomedical devices (9).
The main objective of this research is to study the effects of laser parameters on enhancing the bioactivity of titanium implants for biomedical applications. The surface topography properties of the titanium sheets are improved by altering a range of nanosecond laser pulse parameters and the effects of power and dwell time on the surface energy and topography are investigated. The bioactivity of the processed samples is then validated by soaking the treated samples in the simulated body fluid (SBF), followed by examining the composition of deposited bone-type apatite by using energy dispersive x-ray (EDX) and x-ray diffraction (XRD).
Methods and materials
Laser system and surface treatment of titanium samples
Thin sheets of titanium were chosen for laser surface treatment in this work. The samples were initially irradiated with nanosecond pulses to create the predetermined point patterns on the surface of sample sheets with specific laser parameters. Experiments were completed using a pulse repetition rate (frequency) of 20 kHz, while the dwelling time for each row of points was set to 0.1 ms, 0.5 ms, 1 ms, and 2 ms, using the EZCAD© software. The laser power throughout the experiment was measured to be 8 W and 10 W, which was calculated using a power meter prior to processing of the samples. Table I displays the experiment design parameters.
Laser process parameters used in surface treatment of titanium samples
Samples soaking in SBF for in vitro assessment
The next phase of the study required the preparation of SBF. The SBF was conceived to evaluate the bioactivity of the textures created on the surface of the samples. To prepare SBF, a strict step-by-step method was followed. Since SBF is supersaturated with respect to apatite, failing to follow the preparation procedure accurately could lead to precipitation of apatite in the solution (10).
After dissolving the exact amount of the given chemicals in order, using Tris and hydrochloric acid, the pH level of the solution was controlled to be in the range of 7.40-7.45 at a temperature of 36.5°C, exactly. It is essential to ensure that the pH of the solution does not increase beyond 7.45 at any point during the preparation. The solution was finalized once the pH level of the solution was adjusted on 7.40 at a temperature of 36.5°C exactly (10).
Upon conceiving the SBF, processed samples were placed in the fluid for a set period of time to evaluate their bioactivity. The obtained results are presented and discussed in the following sections.
Results and discussion
The topography of point patterns upon the surface of the samples
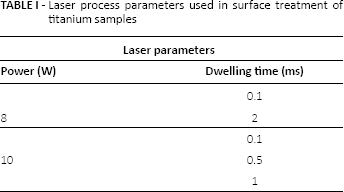
Surface topography and morphology play an essential role in altering material apatite-inducing ability. Laser parameters such as pulse power, dwell time and frequency can influence these material properties significantly. Generally, laser treatment of the materials affects the surface structure and topography of that material by increasing the roughness and oxidation of the material. The exposed surface area of the laser-treated implant is more readily available upon the increase of surface coarseness, which in turn increases the surface energy and wettability and finally, improves the cell adhesion rate and apatite-inducing ability of the treated samples (11). This is because a larger surface area is exposed for apatite inducing to take place. Essentially, the imposed bullet patterns on the surface of the samples in this work, as shown in Figure 1, increase the deviation of the material's surface; therefore, the average arithmetic deviation of the surface increases, which results in a higher average surface roughness and realizes the essential aim of this experiment (6, 8, 12). Furthermore, surface laser processing of the titanium samples increases the surface temperature of the samples up to oxidation temperature, and hence results in creation of thin layers of titania upon the surface of the samples (11). Titania generally exists in two crystallographic structures of anatase and rutile, which are commonly detected by XRD in 2θ = 25.28° and 2θ = 27.4° peaks. Increasing the oxidation rate of the treated surfaces has a direct effect on increasing the wettability of laser-treated samples, which consequently leads to an increase in the apatite inducing ability of the Ti implants and significantly enhances their bioactivity (11).
Point patterns overview (
Figure 1 displays an overall overview of the bullet patterns obtained in this study.
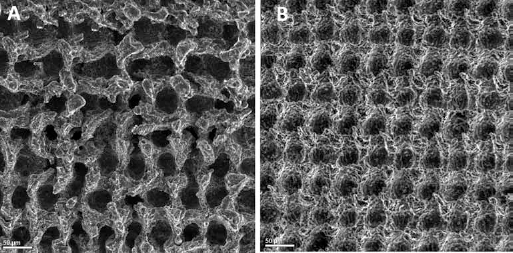
In order to fully examine the effect of roughness on the apatite precipitation of the samples, the pattern-imposed surface of the sample is compared to the original surface of the sample, as displayed in Figure 2. Although the unprocessed section still shows some minor apatite-inducing ability on the surface, the processed section clearly has a thicker layer of apatite induced upon its surface. The processed surface of the sample is much rougher than the unprocessed surface, and therefore has a larger exposed area for cell migration to take place. The higher apatite precipitation rate results in a better bioactivity of the surface titanium. This is in agreement with the expectations discussed in previous sections, and displays the effects of laser surface texturing explicitly. The roughness and oxidation level of the samples could easily be controlled and achieved with a simple one-step process, using lasers. As shown in this figure, the apatite particles are distinctly more on top of the irradiated pattern in comparison to the regular surface of the titanium. It should be noted that, upon laser irradiation on the surface of the samples, the molten material pours around the indicated path, and therefore increases the roughness of the surrounding surfaces. As a result, the small depositions can be seen surrounding the points (10, 12). Another key difference between the treated and untreated surface of the sample is the oxidation level of the two surfaces. The treated portion of the surface has been exposed to controlled laser irradiation, which throughout the delivery of continuous and concentrated energy upon the surface of the sample increases the temperature of the surface. This temperature rise can be controlled to achieve the oxidation level temperature, and results in the formation of titania (titanium oxide) across the surface. Having the thin layer of titanium oxide on the surface provides a cushion-type layer on the surface to which the cells can attach, resulting in a higher apatite perception on the nanosecond laser pulses treated titanium sample (13). As can be seen in Figure 2, the bone-type apatite across the laser-irradiated surface appears as larger spheres compared to the deposition across the unprocessed surface.
Treated and untreated surfaces of titanium sample soaked in simulated body fluid. Power 8 W, 2 ms.
This observation confirms the fact that laser surface texturing of the titanium samples decreases the smoothness and increases the oxidizing level of the Ti samples, and hence increases the cell attachment rate to the surface of the material.
Treated samples soaked in SBF
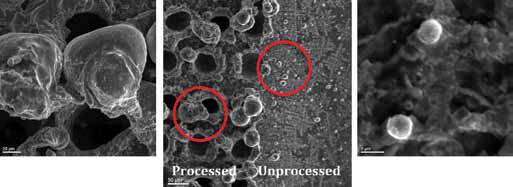
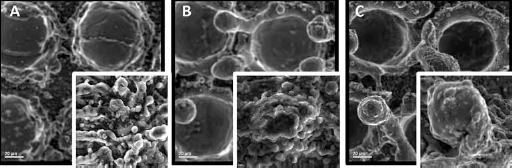
Scanning electron microscope (SEM) images of the apatite-inducing ability of Ti sheets prepared by a nanosecond laser power of 8 W after soaking in SBF are examined. Figures 3 and 4 display the results of these samples, which were soaked for three and seven days, respectively.

Power 8 W; (
Power 8 W; (
The samples displayed in Figure 3 have dwelling times of 0.1 ms and 2 ms, as indicated. In this experiment, the dwelling time is defined as the time given to the creation of each bullet point. The point patterns created on the Ti surface induced by nanosecond laser pulses have increased the surface roughness, and as a result a thicker bone type apatite layer has been deposited on the implant surface. The thicker the bone-like apatite layer deposition is, the higher the surface energy, and thus, the higher the bioactivity of the Ti samples. Similar to the case displayed in Figure 3, Figure 4 illustrates the samples with same conditions soaked in SBF for seven days. Comparing pictures (a) and (b) in Figure 4, it is clearly shown that picture (b) with dwelling time of 2 ms and picture (c) with dwelling time of 0.1 ms has a much better cell adhesion rate, since it has a thicker apatite precipitation across it. When comparing Figure 4 to Figure 3, it is clearly illustrated that a thicker amount of apatite precipitation has covered the surface of the samples in seven days. This is in close agreement with the expectation that higher exposure time to SBF results in better cell adhesion and better bioactivity.
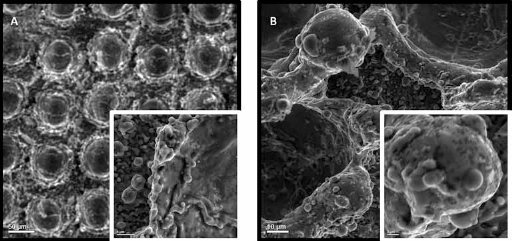
Similar to this case, the samples prepared using power of 10 W and soaked in SBF for three and seven days, respectively, are examined. As was concluded previously, with a longer dwelling time, better apatite-inducing ability is achieved. Similarly, comparing the samples displayed in Figure 5 (3 days SBF soaked time) to Figure 6 (7 days SBF soaked time), it is clear that a thicker deposition layer is visible across the surface of the samples.
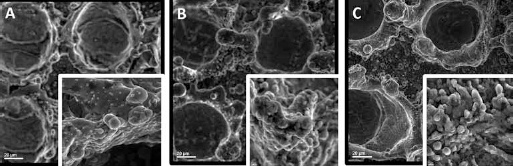
Power 10 W; (
Power 10 W; (
When comparing Figures 6 and 4, it is clear that samples with a higher processed power have a thicker deposition than the samples with lower power. With a higher power, more heat is transferred to the sample, a higher accumulated heat is obtained, and therefore a deeper penetration can be created. However, it must be noted that a maximum power limit must be set to avoid creation of grooves and ablation of the material. A deeper penetration means a higher roughness, which results in more cell adhesion. When comparing the effects of power and dwelling time on the adhesion rate of the samples, it is shown in Figure 5C that higher power results in more apatite-inducing ability than a higher dwelling time does. Overall, the higher the power is, the better the apatite-inducing ability and bioactivity results.
A key factor to consider in these two figures is the difference in the dwelling times between each sample. As indicated, the dwelling times used vary throughout the samples. Comparing the two figures, it can be seen that samples with higher power have more and clearer apatite precipitation layers across their surfaces. This occurs because power has a direct effect on the surface roughness and in turn on the bioactivity of the surface materials. The higher power gives more energy to the creation of the pattern, and thus a higher amount of heat would be transferred to the material. This extra amount of heat results in deeper (thus rougher) penetrations, and eventually increases the cell adhesion rate of the surface of the material. In other words, higher power increases the average roughness of the surface by creating larger irregularities upon melting the Ti surface, and is considered more effective in comparison to dwelling time (14-15-16).
Also, at higher laser power, more energy is transferred to the surface of the Ti target; therefore the temperature of irradiated surface can reach the oxidation temperature. This consequentially will turn the laser treated zone into titanium oxide with increased wettability. Wettability of a material is closely associated with the biological interactions between the biomaterial surface and the actual tissue or organ. These interactions rely heavily upon the surface energy of the Ti implant, which increases with an increase of the wettability of the material. Therefore, when wettability of a material is increased, a higher surface energy exists on the surface of the implant materials, and thus a better meditation of cell adhesion can be attained. Essentially, higher cell adhesion provides a better apatite-inducing ability. Therefore, using higher powers, the bioactivity of the titanium samples is more enhanced than when using longer dwelling times.
EDX and XRD analysis of soaked samples
The EDX analysis of the Ti samples soaked in the simulated buddy fluid shows the presence of titanium, calcium, phosphorous, oxygen, and aluminum; while the non-soaked sample only indicates the existence of titanium, aluminum, and oxygen. The deposition of bone-type apatite on the surface of the SBF-soaked Ti sheets is hence shown via these elements. Figure 7 compares an EDX analysis of the sample before and after it is inserted in SBF and Figure 8 displays an XRD analysis of the samples.
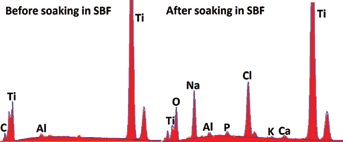
Energy dispersive x-ray analysis of titanium sheets. Before and after simulated body fluid insertion
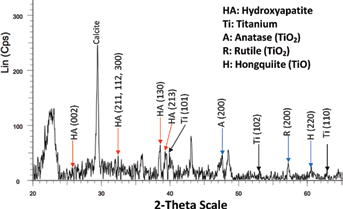
X-ray diffraction analysis of sample soaked in simulated body fluid.
Energy dispersive X-ray (EDX) results of the composition of the deposited apatite layer on the Ti samples (soaked in SBF) indicate the presence of titanium, oxygen, calcium, and phosphorous, which corresponds to hydroxyapatite (HA) (Ca5H2(PO4)3-OH) as shown in Figure 9. HA has a composition similar to the mineral phase of bone, and it has been demonstrated that bone-like HA possesses good osteo-conductivity and has a high affinity for living bone cells.
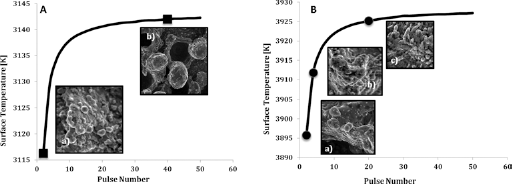
Average surface temperature; (
The existence of the bone-like apatite layer is further investigated by analyzing the samples using XRD; as indicated in Figure 8, upon obtaining XRD analysis, traces of HA were detected on the surface of the processed titanium samples after soaking in SBF, which is in close agreement with the expectations.
Overall, the most optimal laser surface texturing for biomedical applications is the pattern that produces the most roughness on the surface of the material, and the parameters that result in the highest level of oxidation, are able to increase the cell attachment rate to the surface of the samples. Based on the analysis presented, having a high processing power and a high dwelling time results in the most effective pattern for enhancing the bioactivity of the materials. However, power is proven to be more influential in increasing the apatite-inducing ability of the material.
Temperature profile analysis of the laser treated Ti samples
In order to validate the obtained experimental results, a detailed theoretical analysis and computed calculations of the effects of laser parameters on the surface temperature during the laser processing have been conducted.
In order to simplifying the calculation process, we assumed that the laser pulses energy is absorbed in a very thin layer on the surface of irradiated area compared to the heat wave penetration depth; thus, the one-dimensional heat conduction equation can be applied as (12, 17):

where,

Where
In order to simplify the heat-conduction profile, the profile of laser pulse is assumed to be rectangular with the step-like behavior. Thus, the surface temperature for each laser pulse is calculated by:
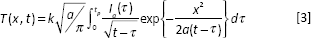
The maximum surface temperature,
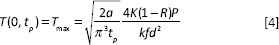
Where,
In this study, the reflection coefficient is 0.56 for laser beam at the wavelength of 1064 nm, the residual energy coefficient is 0.45, and laser spot diameter is 30 µm. Finally, the average temperature across the surface of the samples after
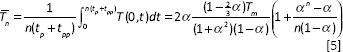
Here, the maximum temperature at the end of each pulse is
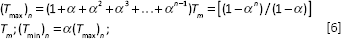
Upon completion of the computed calculations, the average surface temperature for both cases with 8 W and 10 W powers, and a pulse number range of 2 to 50 pulses is attained. The analytical results obtained in this research are associated with possible inexactness due to some assumptions made throughout the simplifying the calculation process. However, the computed results are in a good agreement with our experimental results as shown in Figure 9.
Figure 9 displays the average surface temperatures for the Ti sheets irradiated by nanosecond laser pulses at 8 W and 10 W. As shown, the temperature rises with an increase in the dwell time (number of pulses irradiated on the Ti samples). The higher temperature at higher power and laser pulse number results in more ablation on the surface of the Ti sheets, which in return alters the surface morphology and oxidizes the Ti surface in the irradiated area. Looking at Figure 9A, the two points indicated on the graph are the two samples prepared using dwelling time of 0.1 ms and 2 ms (a) 2, (b) 40, respectively. As illustrated, with a higher dwelling time, a higher pulse number (40) is obtained, and a better apatite-inducing ability occurs on the surface (B) (21).
Similarly, looking at Figure 9B, a similar surface temperature profile is generated. With higher dwelling times (0.1 ms, 0.5 ms, and 1 ms), a higher pulse number is obtained (a) 2, (b) 4, (c) 20, respectively, and hence a higher average surface temperature results. Similar to previous case, the adhesion rate of the samples increases at higher temperatures. In both cases, the surface temperature increases above the melting point of titanium; hence, some ablation occurs that in return increases the surface roughness of the material (21). This is in agreement with the experimental observations.
When comparing the specified points on Figures 9A and B, it is clearly shown that the sample prepared with power of 10 W and dwelling time of 1 ms (picture c), has the best apatite deposition layer across its surface, even though it has a shorter dwelling time compared to case (b) in Figure 9A. This is explained by looking at Figure 9B: the average surface temperature of Figure 9 A (picture c), is much higher than Figure 9B, (picture b) due to its high power. Hence, more ablation takes place across the surface of picture c), which results in a higher surface roughness (21, 22). Also, a larger amount of titania is created around this point, which affects the apatite-inducing ability (11, 13) Therefore, having a higher power is more influential on the roughness of the surface, compared to the dwelling time. This is in very close agreement with the experimental results.
It is important to note some key factors with regard to use of laser for surface texturing. Based on the presented results, it is clear that the parameters influential on the bioactivity of a material are easily controlled through the use of a laser. The environment at which the implant devices would have to function is drastically different from environments in which regular engineering devices function. Implants must be designed so that they can function in human body temperature at 37°C, aqueous and saline natural environments, with water as the dominant chemical species, in often-aggressive conditions, which would eventually give rise to both corrosion- and wear-related problems. Therefore, one of the most important characteristics of the materials used in the implant systems are their chloride content, dissolved oxygen level and pH level (23). Hence, the oxide level of the materials used in designing the scaffolding system is of utmost importance, in order to prepare them for body conditions.
From this point of view, laser surface texturing offers a huge advantage in comparison to more conventional methods. Through the use of these methods, the properties of the irradiated spot change both in roughness and oxidation level of the material. The transferred heat to the surface of the material increases its temperature to the oxidation point, and hence adds to the oxidation level of the material. Therefore, by using a laser, not only are the final results easily and accurately controlled, but also both aspects of the material in terms of its bioactivity are enhanced in one single step. This is an important contribution of lasers in the biomedical field, which surpasses all the conventional methods commonly used in industry.
Conclusion
In this study, the effects of laser power and laser beam dwell time on the apatite-inducing ability (bioactivity) of titanium implants were investigated. This was done by using different power and dwelling times in preparation of bullet point patterns. By increasing the surface roughness of the samples, the apatite precipitation across the surface of the titanium was increased; hence its bioactivity was made more applicable in bone and tooth implant fabrication.
After pre-defined point patterns were generated on their surfaces, the samples were soaked in SBF for three days and seven days, respectively, before being examined for their bioactivity properties. The SBF soaked Ti samples were then analyzed using SEM, EDX, and XRD to evaluate the bone-type apatite-inducing ability on nanosecond laser-treated samples. Also, the effects of power and laser beam dwell time on the surface roughness and oxidation level of the material were compared. Upon completion of the analysis, it is concluded that using higher power and longer dwelling time results in a higher surface temperature at the laser irradiated spot. Therefore, a larger roughness and oxidation level is obtained. This leads to better cell adhesion rate on the surface of the titanium implants. In conclusion, power was found to be more influential on surface topography and morphology properties of the material in comparison to dwelling time.
Footnotes
Financial support: This research is funded by the New Brunswick Innovation Foundation (NBIF) and the McCain Foundation.
Conflict of interest: The authors declare that there is no conflict of interests regarding the publication of this paper.
