Abstract
Background
Nanoparticles (NPs) are increasingly being used in a number of applications that include biomedicine, biological labeling and cancer marker targeting, and their successful storage is important to preserve their viability. A systematic investigation of the thermal and photo stability of chemically stabilized cadmium telluride (CdTe) quantum dots (QDs) under various storage conditions either in solution or as dried nanoparticles has not been published. Here we report experiments involving chemically synthesized glutathione-capped CdTe QDs whose photoluminescence spectra were examined initially and then periodically during storage times up to 76 days.
Methods
Samples of dried QDs or QDs in solution (water or buffered) were examined under different light conditions including complete darkness, constant 12,000 lux incident light, and under diurnal sunlight; at temperatures ranging from -80°C to room temperature.
Results
Though QDs stored in solution in the dark at -80°C lost only 50% of peak fluorescence (FL510) within 2 weeks, solution-stored QDs exposed to sunlight at room temperature showed FL510 drops of 85% in the first 24 hours. In contrast, QDs precipitated from aqueous solution, dried and stored in time course experiments in the presence of atmospheric oxygen – when resuspended in water – lost an average of only 12% FL510 over 76 days under all conditions tested, even in direct sunlight.
Conclusions
Glutathione-capped CdTe particles can be stored as dried nanoparticles for extended periods of time, enhancing their viability in biomedicine, biological labeling and cancer marker targeting.
Introduction
Nanoparticles (NPs) are being increasingly used in a number of applications that include biomedicine, biological labeling and cancer marker targeting, and their successful storage is important to preserve their viability. To avoid decomposition by hydrolysis or oxidation by atmospheric O2, chemically capping quantum dots (QDs) with primary amines, cysteine and mercaptopropionic acid, among others, has been carried out. Pérez-Donoso et al developed a method to synthesize glutathione-capped CdTe NPs in aqueous solution under mild chemical conditions (1). Though mentioned in passing, reports about storage effects on QDs photoluminescence have involved relatively short storage times – for instance, hours (2), 10 (3) or 15 days (4), or a few weeks (5). To our knowledge, stability experiments involving chemically capped QDs that have been synthesized, precipitated, dried and resuspended after storage at various temperatures have not been reported so far. Recent work by Wang et al (6) does describe experiments regarding photoluminescent stability of CdTe/Cd(OH)2 NPs maintained in solution at room temperature. Li et al report the photochemical stability of QDs maintained in their synthetic solutions for over 50 days; however, these experiments did not involve QDs that were isolated, stored as dried product, or re-solvated and examined (7). We report here a systematic investigation of the thermal and photo stability of chemically stabilized cadmium telluride (CdTe) QDs under various storage conditions either in solution or as dried NPs.
Materials and methods
Glutathione-capped CdTe QD synthesis
QDs were synthesized using the procedure of Pérez-Donoso et al (1). Briefly this involved reducing tellurium oxyanions with glutathione (GSH) in the presence of cadmium chloride in a citrate-borax buffer solution (the same buffer used throughout the study) at pH 9.0. Stock solutions of GSH in deionized water (30 mM GSH in deionized water; Sigma-Aldrich, St. Louis, MO, USA), citrate-borax buffer at pH ∼ 9 (30 mM sodium citrate in deionized water; Fisher Scientific, Fair Lawn, NJ, USA; and 30 mM of sodium borate; Sigma-Aldrich), cadmium chloride (25 mM CdCl2 in deionized water; Sigma-Aldrich) and potassium tellurite (7 mM K2TeO3 in deionized water; Sigma-Aldrich) were prepared. To a test tube, 1.60 mL of 25 mM CdCl2, 3.64 mL of 30 mM citrate-borax buffer and 3.33 mL of 30 mM GSH were added and then vortexed vigorously and subsequently allowed to sit at room temperature for 5 minutes. Below the surface of the vortexed solution, 1.43 mL of 7 mM K2TeO3 was added slowly via micropipette. The final ratio of CdCl2/GSH/K2TeO3 in this synthetic mixture was 4:10:1. The reaction flask was then placed in a 90°C hot water bath to initiate nucleation. Although these QDs can be tuned for maximum fluorescence emission from green to red by increasing the incubation time (1), all QDs used in this study were heated for 2 hours at 90°C. Dynamic light scattering has previously determined the green QDs synthesized in this manner to be approximately 3.3 nm in size on average (1).
QDs synthesized in aqueous solution were precipitated with 2 volumes of ethanol and centrifuged for 20 minutes (18,500 g). Precipitated QDs were dried for 24 hours in a desiccator over CaCl2. For absorbance and fluorescence determinations, dried QDs were dissolved in borax-citrate buffer (1), checked in a UV transilluminator (UVP, Upland, CA, USA), and analyzed using a fluorescence spectrometer (1 cm cuvette; model F-4500; Hitachi, Schaumburg, IL, USA). Dried-storage QDs were treated in a slightly different manner and were resuspended in deionized water (see Storage).
Storage
QDs for long-term thermal and photo stability studies were initially collected and dried as described, then either replicate masses (2.5 ± 0.2 mg solid) were weighed into individual cuvettes and stored as dried solids (shaken periodically) or resuspended in deionized H2O or in buffer (2.5 ± 0.2 mg QDs in 2.5 mL H2O or buffer), and stored in that solution. No effort was made to exclude atmospheric O2 from QDs stored as solids or in solution. All dried-storage QDs were resuspended in water before examination. Freshly prepared QDs for the initial analysis were precipitated, dried and resuspended in buffer (see Glutathione-capped CdTe QD synthesis). Storage containers were disposable cuvettes (polypropylene; 3 mL; Brand GmbH, Wertheim, Germany). For fluorescence analyses, solids were resuspended in deionized water and analyzed once using the fluorescence spectrometer and a 1-cm cuvette with water as the solvent. Stored solutions were inverted repeatedly before fluorescence analysis and photography.
Thermal and photo stability experiments
Each sample type (dried, in H2O or in buffer) was stored in capped (but not sealed) cuvettes under 3 different temperature conditions and under various light conditions: from complete darkness to sunlight. Temperature conditions consisted of -80°C, 4°C and room temperature (∼25°C). Incident light intensity was measured periodically with a light meter (model 3252; Control Company, Friendswood, TX, USA). Light conditions in storage included complete darkness, constant refrigerator light (100 lux), work-day cycling laboratory light (diurnally varying, periodically up to 400 lux), fluorescence bulb light (constant 1,200 lux) and sunlight (diurnally cycling up to 70,000 lux through a standard glass, laboratory window). QD samples were examined for absorbance and fluorescence intensities periodically, beginning 1 day after synthesis and for up to 76 days.
Sulfur content
QDs (in H2O) were analyzed for sulfur content (i) 24 hours after synthesis, centrifugation, drying and resuspension and then (ii) after exposure to sunlight through the laboratory window for 96 hours (4 days and nights). Sulfur was determined using inductively coupled plasma spectrometry (ICP; model Spectro Ciros Vision ICP-OES; Spectro, Mahwah, NJ, USA) in 10% HNO3 using the 180.731-nm sulfur line.
Results and discussion
Freshly prepared, green GSH-capped CdTe QDs had a peak absorbance at 475 nm and peak fluorescence emission at 510 nm (FL510) confirming earlier work by Pérez-Donoso et al (1). Time course experiments of dried-, water- and buffer-stored samples, however, showed large differences in QD colors under room light and transilluminator excitation. Digital photographs (auto setting; model DSC-H70; Sony) in room light and fluorescence emission on a transilluminator (ex: 302 nm) for the 3 sample groups were taken periodically (Figs. 1-2-3).

Quantum dots (QDs) under visible and UV light after being stored in deionized water and photographically imaged periodically for 76 days. LW = stored at room temperature in the lab; SW = room temperature in sunlight; BW = room temperature under bulb at 1,200 lumens; 4dW = 4°C in dark; 4LW = 4°C in refrigerator in light; -80dW = -80°C in refrigerator in dark; LdW = room temperature in dark.
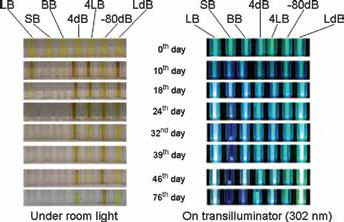
Quantum dots (QDs) under visible and UV light after being stored in citrate-borax buffer and photographically imaged periodically for 76 days. LB = stored at room temperature in the lab; SB = room temperature in sunlight; BB = room temperature under bulb at 1,200 lumens; 4dB = 4°C in dark; 4LB = 4°C in refrigerator in light; -80dB = -80°C in refrigerator in dark; LdB = room temperature in dark.
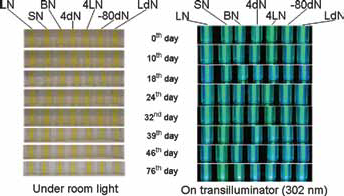
Quantum dots (QDs) under visible and UV light after being stored as dried solids and photographically imaged periodically for 76 days. Dried QDs were resuspended in water immediately before examination. LN = stored at room temperature in the lab; SN = room temperature in sunlight; BN = room temperature under bulb at 1,200 lumens; 4dN = 4°C in dark; 4LN = 4°C in refrigerator in light; -80dN = -80°C in refrigerator in dark; LdN = room temperature in dark.
Peak fluorescence at 510 nm (ex: 375 nm) examined periodically over the same time course (Fig. 4) highlighted significant differences between the solutions and dried storage conditions. The 375-nm excitation wavelength was used for assessing degradation instead of the 475-nm absorbance peak to avoid disposable cuvette light scattering, an artifact not present in quartz cuvettes.
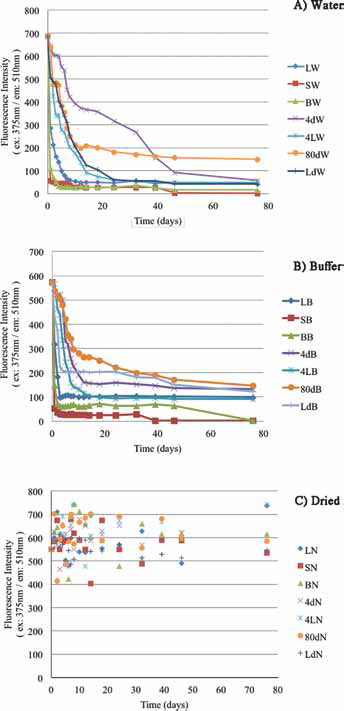
Fluorescence intensity vs. time for quantum dots (QDs) stored in water
Photographic and FL510 data along the 76-day time course showed large differences for water- or buffer-stored samples. QDs stored in either solution degraded quickly, as measured by FL510 drops, and this process was accelerated at warmer temperatures. For example, either sunlight-exposed solution sample (25°C in water or buffer) was visually bleached by the 10th day with FL510 drops of over 85% in the first 24 hours; by the 76th day, solution-stored sunlight-exposed QDs FL510 had dropped by 99.9%. Buffer-stored QDs were more stable than those stored in deionized water but only marginally. Solution-stored samples did much better in the dark at -80C°, losing only 50% of initial FL510 intensity in 2 weeks.
These results for stored QD solutions can be sharply contrasted with the thermal and photo stability of QDs stored as dried solids (Figs. 3 and 4c). Dried QDs stored in capped cuvettes, shaken regularly to expose these solids to O2, atmospheric moisture and light, showed a FL510 range of ± 24% RSD (relative standard deviation) for all 126 dried QD samples over the entire 76-day time course. Taking the average FL510 on the first day for all 7 storage conditions and FL510 average on the 76th day, the difference was 12%. And most interesting, it appears that FL510 variations over the entire dried QD storage time course were uncorrelated with either light source or storage temperature. In fact, it is most reasonable to suggest that the greatest single influence on the FL510 measurements for the 126 dried QD samples stemmed from the uncertainty in the initial mass of dried QDs weighed into each of the separate cuvettes at the experiment's start (2.5 mg). Even room temperature, sunlight-exposed dried QDs showed no obvious degradation trend over the entire 76-day time course: compare FL510 for those replicates at days 1, 14, 18, 32 and 76. The high relative humidity of our laboratory (30°42’ N; 95°32’ W) also apparently did not contribute to degradation via surface hydrolysis reactions of even the longest stored GSH-capped QDs.
Shifts in maximum QD absorbance wavelengths (data not shown) and fluorescence wavelengths (see Figs. 1 and 2) occurred for samples stored in solution. The reason for this is difficult to determine but can reasonably be explained this way: For QDs stored in solution, the particle size apparently changed due to nanoparticle coagulation or partial aggregation during the storage period. Initially, as the particle size increased, the excitation and emission wavelengths shifted to longer wavelength (1); however, sometimes this process led to precipitation – even in solutions shaken immediately before imaging – and as a result, we sometimes observed large black-colored particles at the bottom of the cuvettes (Figs. 1 and 2).
Aldana et al proposed 3 distinguishable processes in photodegradation of thiol-capped QDs in solution: photocatalytic oxidation of surface caps, photooxidation of the QDs themselves and finally precipitation of degraded QDs (8). As Figures 1 and 2 show, the third step in this process, QD precipitation, is obviously visible for solution-stored QDs and most clearly in those samples exposed to sunlight (all samples were shaken for a few minutes before being photographed). While initially the precipitated QDs still fluoresced after settling at the bottom of the cuvette, by the 76th day, FL510 had dropped almost to zero (Fig. 4a, b) in solution-stored samples.
In an effort to follow sulfur incorporated in or on these QDs, total sulfur content of freshly-prepared QDs was compared with total sulfur present in the aqueous supernatant and precipitated phase of QDs exposed to sunlight for 96 hours at room temperature (Tab. I).
Inductively coupled plasma spectrometry analysis for sulfur before and after 96 hours of photodegradation in sunlight
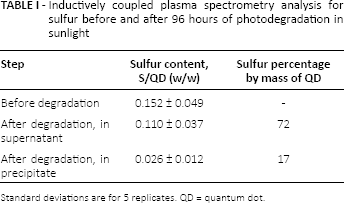
Standard deviations are for 5 replicates. QD = quantum dot
This experiment most closely mimics the work of Aldana et al, because they used direct UV light on QDs resuspended in water to measure degradation (8). The results for 5 photodegraded replicates showed that the supernatant on average contained 72% of the initial QD sulfur content and the precipitate only 17%. The 89% recovery of the average initial sulfur is almost certainly low due to the loss of precipitate in the drying and reweighing step where dried samples had an average mass of 10 mg (n = 5). And these losses would contribute to sulfur collected in the spun-down fraction. This means that clearly, over 70% of sulfur initially in or on GSH-capped CdTe QDs was lost to solution during their photodegradation. Again, this confirms the proposed mode of degradation suggested by others but until now never directly examined: hydrolysis or oxidation and dissolution of sulfide capping agents for QD samples in solution (2-3-4, 8). The long-term storage stability of dried, GSH-capped CdTe QDs also reinforces the importance of solution interactions in the proposed degradation processes because the lack of solvent in experiments reported here – involving dried QDs – helped confer thermal and photo stability in the presence of O2, room temperature and even sunlight.
Conclusions
QDs precipitated from aqueous solution, dried and stored in time course experiments in the presence of atmospheric oxygen – when resuspended in water – had only lost an average of 12% FL510 over 76 days under all conditions studied and even in direct sunlight. Photodegradation in aqueous solution in sunlight over 96 hours released over 70% of sulfur initially contained in freshly prepared NPs into solution, suggesting hydrolysis or oxidation and dissolution of sulfide capping agents for QD samples in solution.
Footnotes
Financial Support: P.T.W. and T.G.C. received financial support from the Robert A. Welch Foundation (grant X-011), Houston, Texas, USA. J.M.P.-D. received financial support from the Chilean Antarctic Institute, Punta Arenas, Chile (INACH T-19_11) and Chilean National Commission for Scientific and Technological Research (Fondecyt 11110077 and Anillo ACT 1107), Santiago, Chile. W.A.D.-V. and C.C.V. received financial support from Fondecyt Regular grant 1130362.
Conflict of interest: The authors declare that they have no conflict of interest.
