Abstract
Cross-talk between bacteria and mammalian cells is increasingly recognized as an important factor, especially during chronic infections. In particular, the interaction of extracellular bacterial signaling molecules with cells of the innate immune response is of special interest. In this context, we investigated whether the Pseudomonas quinolone signal (PQS) which is a quorum sensing molecule produced by bacteria and participates in biofilm formation and virulence has any influence on polymorphonuclear neutrophils (PMN), the cells of the “first line defense” against bacterial infections. We found that PQS did not enhance the bactericidal activity of PMN and did not induce apoptosis at concentrations up to 100 μM. However, PQS stimulated chemotaxis of PMN in doses of 10-100 μM. This PQS-dependent chemotaxis could be inhibited with SB203580 which blocks MAPkinase p38, suggesting a signaling pathway similar to AHL-12 induction. Using bacterial cell culture supernantants of Pseudomonas aeruginosa wild-type cells and a PQS-deficient mutant strain support the in vivo relevance of these findings. Since PQS is produced in the early phase of biofilm formation, PMN infiltration could be timely enough to eradicate bacteria before biofilm formation is completed, which confers the bacteria with a relative resistance to host defense mechanisms.
Keywords
Introduction
Biofilm formation is the crucial factor in many chronic bacterial infections such as implant-associated osteomyelitis, endocarditis in patients with artificial heart valves, and vascular catheter or ventilator tube infections (1, 2). Moreover, pulmonary biofilm infections are the leading cause of morbidity and mortality in patients with cystic fibrosis due to persistent infections that eventually lead to lung damage and respiratory failure (3). Bacteria grown in biofilms are imbedded in a slimy, self-produced polymeric matrix composed of polysaccharides, nucleic acids, lipids and proteins (4-7). Once established, these sessile communities are protected from the host immune response and are difficult to eradicate due to their high inherent resistance against antibiotic agents (8-12).
Bacterial cell-to-cell communication, also named quorum sensing (QS), is known to be involved in biofilm development and is mediated by a variety of different QS signaling molecules. One major group of QS molecules are the N-acylhomoserine lactones (AHL), which are synthesized and secreted by many Gram-negative bacteria like the opportunistic human pathogen Pseudomonas aeruginosa. In P. aeruginosa, the two AHLs (3-oxododecanoyl)-L- (AHL-12) and N-(butanoyl)-L-homoserine lactone (AHL-4) control the production of extracellular virulence factors and promote biofilm maturation by inducing expression of more than 300 genes (13). Moreover, it has been shown previously that AHL-12 strongly impacts mammalian cells, in particular host immune cells by inhibiting T-cell proliferation and antigen presentation or inducing apoptosis (14, 15). Recently, we and others have shown that AHL-12 also stimulates the host defense by increasing phagocytosis of bacteria by macrophages or neutrophils and induces chemotaxis of neutrophils through induction of p38 and leukocyte specific protein 1 (16-18).
In addition to AHLs, P. aeruginosa uses the non-AHL signal 2-heptyl-3-hydroxy-4-quinolone (the Pseudomonas quinolone signal; PQS, Fig. 1) as an extra QS molecule. PQS is closely linked to the AHL signaling network and possesses numerous regulatory and physiological functions, including control of biofilm development, secondary metabolite and virulence factor production as well as iron acquisition, among others (19). Interestingly, PQS is present at sites of infection, e.g. in sputum samples of cystic fibrosis patients (20, 21), and is capable of modulating the host immune response by modulating activity in T-cell proliferation and cytokine (IL-2) release, suppressing interleukin 12 (IL-12) and tumor necrosis factor alpha (TNF-α) secretion by LPS-stimulated macrophages and repressing HIF-1α protein levels (19, 22-25).
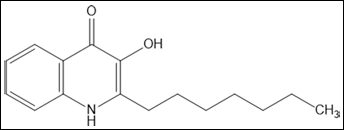
The Pseudomonas quinolone signal (PQS).
In this study we investigated the potential of PQS to activate human neutrophils and analyzed signaling pathways related to migration. We found an induction of chemotaxis and up-regulation of adherence proteins and cellular functions that enhance the local neutrophil response.
MATERIAL AND METHODS
Bacterials strains and materials
Pseudomonas aeruginosa PA14 wild-type and the corresponding pqsA transposon insertion mutant ID23621 (mutant) were obtained from the P. aeruginosa PA14 mutant library (26). Cells of wild-type and mutant strain were grown for 20 h in 30 mL BHI medium at 37°C under shaking conditions. The culture medium was centrifuged at 5800×g for 10 min and the supernatant was subsequently sterilized by filtration and used for functional assays (see below). A FITC-labeled Escherichia coli was used in the phagocytosis assay. PQS, calcein AM and f-MLP were purchased from Sigma-Aldrich (Seelze, Germany) and SB 203580 from Calbiochem (La Jolla, USA).
PQS measurement by liquid chromatography/tandem mass spectrometry (LC-MS/MS)
Sterilized cell-free supernatants of bacterial overnight cultures were mixed with two volumes of dichloromethane and the lower organic layer containing PQS was transferred into a new reaction tube following evaporation under nitrogen gas at room temperature. The pellet was resuspended in pure methanol following quantification by LC-MS/MS using a Zorbax Eclipse XCB-C8 5 μm, 150×3.6 mm HPLC column (Agilent, Santa Clara, CA, USA) and a mobile phase consisting of acetonitrile - water 80:20 (v/v) with 100 μM EDTA and 0.1 % acetic acid at a flow rate of 0.40 ml/min. Electron spray ionization (ESI)-MS was performed on an API 365 triple quadrupole mass spectrometer (PE Sciex, Toronto, Canada) using a turbo ion spray interface used in positive mode. Single MS experiments (Q1 scan), MS/MS experiments (product ion scan, PIC), and multiple reaction monitoring (MRM) were performed using nitrogen as curtain gas, nebulizer gas, heater gas and collision gas. Instrumental parameters were optimized by infusion experiments with PQS standard solution (10 μg/ml; Sigma-Aldrich, Seelze, Germany) infused into the mass spectrometer using a syringe pump (Harvard Apparatus, South Natick, MA, USA) at a flow rate of 10 μl/min. To quantify PQS with high selectivity and sensitivity, MRM experiments were performed using the transitions from precursor ion to fragment ion: 260/175 (quantifier), 260/146, 260/147 and 260/188 (qualifier). An external calibration was performed using PQS standard solutions with concentrations ranging from 10 ng/ml up to 1000 ng/ml.
Isolation of PMN from peripheral blood
Heparin blood from volunteers (mainly laboratory personnel and students) was layered on PolymorphPrep (Nycomed, Oslo, Norway) after receiving approval for the procedure from the local ethics committee. After centrifugation, the PMN fraction was harvested and washed repeatedly in phosphate-buffered saline (PBS, pH 7.4). For the analyses of chemotaxis, the cells were suspended in Hank's balanced salt solution (HBSS) containing 0.5% bovine serum albumin (BSA), at a final concentration of 1 × 10 6 cells per milliliter HBSS. Because of the wide intraindividual variation of the cell response, all assays were performed with cells of at least three different donors.
Determination of cell death and apoptosis
Intercalation of propidium iodide into DNA was measured by cytofluorometry as described by Belloc et al (1994) (27). In parallel, binding of Annexin V FITC which binds to the membrane phospholipid phosphatidylserine (PS) in the presence of Ca2+ was determined. Isolated PMN were incubated for 30 min with PQS in doses ranging from 1 μM to 100 μM; formalydehyde (3%) was used as a “death” control.
Phagocytosis
A commercially available kit (Glycotope Biotechnology, Heidelberg, Germany) was used. In brief, FITC-labeled Escherichia coli were incubated with whole heparinized blood at either 4°C or 37°C for 15 min. Uptake into PMN of bacteria was quantified by cytofluorometry.
Production of oxygen radicals
To determine the production of oxygen radicals, we used the method described by Babior et al (28). In brief, PMN (1×106) were suspended in cytochrome c solution (Sigma-Aldrich, Seelze, Germany, 1 mg/ml HBSS,) and stimulated with phorbol ester (PMA) (Sigma-Aldrich), or with PQS in the concentrations as indicated in the text. Reduction of cytochrome c was measured photometrically after 30 min and calculated as ΔOD (550 nm).
Cytofluorometry
To determine the expression of surface receptors on PMN, a FITC-labeled antibody to CD66b (Coulter Immunotech, Marseille, France) was used to identify PMN, and phycoerythrin (PE)-labeled antibodies to either anti-CD11b (BD Biosciences, Heidelberg, Germany) or anti-CD18 (Serotec GmbH, Düsseldorf, Germany). One hundred microliters of whole blood was incubated with anti-CD66b-FITC and the respective PE-labeled antibodies (0.1-5 μg) for comparison with mouse IgG (either PE- or FITC labelled; Beckman Coulter Marseille, France) for 20 min. Then the erythrocytes were lysed using BD Facs Lysing solution (BD Biosciences) and cells were subjected to cytofluorometry using FACS-Calibur and CellQuest Pro software (Becton Dickinson, Heidelberg, Germany). Ten thousand events were counted, and expression of CD11b or CD18 on CD66b positive cells was shown as a histogram. Expression of CD11b or CD18 was quantified as mean fluorescence intensity (MFI).
Chemotaxis
Chemotaxis was determined using the BD Falcon HTS Fluoroblok 96-Multiwell Insert System with a polyethylene terephthalate (PET) membrane insert plate with 3 μm pore size. Isolated PMN were labeled with calcein (10 6 cells/ml were incubated for 15 min at 37°C with calcein (1 μg/ml), then washed three times with HBSS and seeded in a concentration of 2×10 5 /50 μL into the insert plate. PQS in concentrations of 1 μM, 10 μM, 100 μM, and for comparison HBSS or fMLP (10−7 M) was added to lower wells. After 2 h at 37°C the insert plate was removed and fluorescence was determined by a PerkinElmer VIC-TOR2™ microplate reader using Wallac Workstation software (Waltham, MA, USA).
Results and Discussion
PQS does not induce apoptosis or bactericidal activity of PMN
Polymorphonuclear neutrophils were isolated from at least three different donors and subsequently incubated with PQS to identify any toxic effect. After 30 min of incubation, PQS did not show any cytotoxicity towards PMN when used in doses up to 100 μM (Fig. 2). To answer the question whether PQS would modulate PMN functions, in a first set of experiments an impact on the phagocytosis of bacteria was studied. PMN efficiently took up opsonized, FITC-labeled E. coli demonstrating that PQS did not enhance phagocytosis when used in doses of 1 μM to 100 μM (example in Fig. 3). In this regard, PQS exhibited functional differences in comparison to the AHL-12 quorum sensing molecule, which was shown to significantly enhance phagocytosis (17, 29) and induce apoptosis (14).
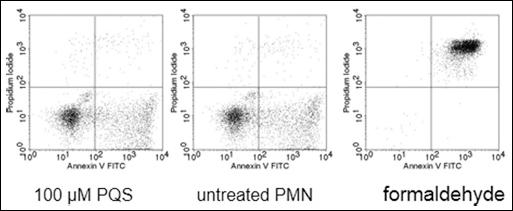
PQS does not induce apoptosis of PMN. PMN were incubated with (left panel) or without PQS (100 μM) for 30 min. Apoptosis and cell death were determined by staining with Annexin-V FITC and intercalation of propidium iodide, respectively. On the right panel, the “death control” is shown.
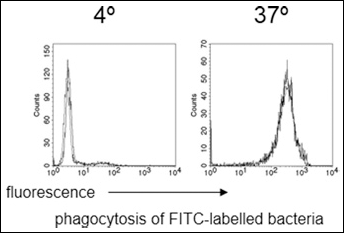
PQS does not affect phagocytosis of bacteria by PMN. PMN in whole blood were incubated either with PQS (100 μM; thick line) or without (thin line) with opsonized, FITC-labeled E. coli at either 4°C or 37°C for 15 min. Thereafter, green fluorescence associated with the cells was determined by cytofluorometry.
In a further set of experiments, the possible effect of PQS on the production of reactive oxygen species was tested. In concentrations of 1 to 100 μM, PQS did not induce reactive oxygen species and did not modulate the reactive oxygen production induced by f-MLP (10 7 or 10 8 M) (data not shown). In that respect, PQS does not directly affect the bactericidal activity of PMN.
Furthermore, the effect of PQS on the constitutively expressed adhesion proteins CD11b/CD18 was analyzed. As demonstrated in the example shown in Figure 4, PQS only marginally enhanced their expression; on average an increase of 22% for CD11b and 17% for CD18 (mean of three experiments with cells of different donors) was detectable.
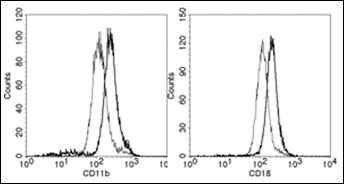
PQS upregulates expression of CD11b/CD18. PMN in whole blood were cultivated with (thick line) or without (thin line) PQS (100 μM) for 30 min at 37°C. Shown is the surface expression on PMN (identified as CD66b positive cells).
PQS induces chemotaxis of PMN
In addition, we investigated the chemotaxis of PMN in response to different PQS concentrations and observed that PQS was chemotactic for isolated PMN. In doses of 10 μM to 100 μM PQS significantly induced chemotaxis (examples and summary of data are shown in Figs. 5A and B). Compared to bona fide chemokines such as f-MLP or IL-8, the molar concentrations required for induction of chemotaxis were much higher, a phenomenon that was also observed for AHL-12, and which raises the question of the in vivo relevance. To this end, we investigated the effect of bacterial supernatants from P. aeruginosa PA14 wild-type cells as well as the corresponding PQS-deficient mutant strain ID23621 (26) which is not able to synthesize PQS due to a transposon mutation in the PQS synthesis gene pqsA. Bacteria were grown for 20 h at 37°C in BHI medium under shaking conditions and supernatants of the wild-type and mutant strain were sterilized and subsequently analyzed for PQS using LC–MS/MS. The amount of PQS in supernatants was quantified according the settings given above using the specific transitions from m/z 298 (molecule ion) to m/z 197 and m/z 102. As expected, PQS was only detectable in the wild-type supernatant (1240 ng/ml supernatant). Using these supernatants in our chemotaxis assay, we observed that supernatants of P. aeruginosa induced a chemotactic response, which was in accordance to our previous studies on AHL-12 (16). Culture supernatant of the mutant strain induced a lower response in comparison to the wild-type, which, however, could be reconstituted by PQS (Fig. 5C). These results indicate that sufficient PQS is produced by wild-type bacterial cells to induce chemotaxis of PMN. It is not the only chemokine, however, a likely candidate for an additional molecule is AHL-12. This notion is supported by our previous observation that AHL-12-deficient supernatants still induced chemotaxis of PMN, though to a lesser extent (16).
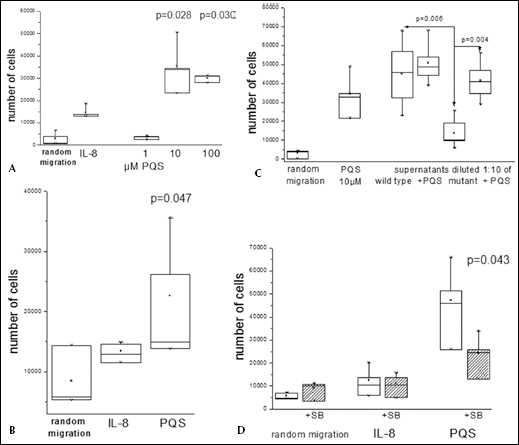
PQS induces chemotaxis of PMN.
PQS-induced chemotaxis was inhibited when cells were pre-incubated with SB203580, which blocks the MAPkinase p38 (Fig. 5D). This enzyme is required for the induction of cellular migration by so-called “end-point” chemokines, and suggests a signaling pathway similar to AHL-12. How PQS is recognized by PMN is not yet clear. As a lipid, it may enter the cell freely; chemotaxis, however, supposedly requires a polarization of the cells, which is accomplished by a clustering of appropriate surface receptors, a phenomenon that needs further investigation.
Conclusions
The results of our studies demonstrate that the Pseudomonas quinolone signal does not exhibit cytotoxic effects on PMN nor does it have an impact on apoptosis. However, we found that PQS stimulates chemotaxis of PMN, likely via a signaling pathway similar to AHL-12. Regardless of the activation mechanisms, the fact that PQS induces chemotaxis might have an implication for the local host defense. Since PQS is produced in a similar way to AHL-12, before and in the early state of biofilm formation, PMN as a first-line defense could be recruited in a phase of infection where bacteria might still be vulnerable and can be eradicated (30, 31). On the other hand, infiltration of PMN into sites of infection can also enhance the local inflammatory response, thus leading to a progression of tissue damage. A better understanding of the dynamics of host-pathogen interactions, in particular in cell signaling events is increasingly needed for the development of new and more efficient antibiotic treatment options for biofilm infections.
Footnotes
ACKNOWLEDGEMENTS
We thank Anke Neidig and Michael Nusser for technical assistance.
