Abstract
Purpose
The aim of this study was to compare the residual monomer and microhardness of poly(methyl methacrylate) (PMMA)–based denture resins processed by using autoclave and conventional water bath techniques.
Methods
To determine the amount of residual methyl methacrylate (MMA) monomer, disk-shaped specimens (n=5) were prepared from 3 different acrylic resins (Meliodent, Paladent and Qc-20). Control groups were polymerized in water bath for 30 minutes at 100°C. The study groups were prepared in an autoclave device for 60°C/30 min followed 130°C/10 min and the other group for 60°C/30 min followed by 130°C/20 min. According to standard calibration curves, ultraviolet spectrophotometry at 230 nm was used to determine the residual monomer. For the Vickers hardness measurements, disk-shaped specimens (n=5) were prepared for each test group. Hardness measurements were performed with a Vickers hardness tester under a 4.91-N press load for a 30 seconds, after immersion in distilled water at 37°C for 48 hours. The data were analyzed by ANOVA and Tukey HSD test (p<0.05).
Results
Autoclave polymerization produced a significant decrease in the amount of residual monomers for all resin groups (p<0.05). This procedure also showed a significant increase in hardness for all resin groups (p<0.05). For the 3 resin groups, no significant differences were found between autoclave polymerization for 10 minutes and for 20 minutes (p>0.05).
Conclusions
The autoclave polymerization technique exhibited significantly lower residual monomer content and greater hardness than conventional heat polymerization.
Introduction
Since the latter half of the 19th century, polymers have dominated the denture fabrication process. Vulcanized rubber, polystyrene, polyvinyl acrylic, polyamides and poly(methyl methacrylate) (PMMA) have been used in denture base fabrication. Although each of these materials displays desirable properties, none has proved superior to PMMA. As a result, PMMA remains the material of choice for denture base fabrication (1-3).
When fabricating a denture base from polymers, certain physical and mechanical properties of the final polymer are important. The cured polymer should be stiff enough to hold the teeth in occlusion during mastication and to minimize uneven loading of the mucosa underlying the denture. Also the cured polymer should be biologically inert and slow to foul when in contact with oral flora. During fabrication of a denture, the physical and mechanical properties mentioned here can be influenced by curing conditions and choice of materials (2-4).
Over the years, curing procedures have been modified with a view to improving the physical and mechanical properties of resin materials. Polymerization of PMMA can be initiated by decomposition of benzoyl peroxide with heat, by chemical activation using material such as dimethyl-p-toluidine, by visible light or microwave energy. Polymerization of PMMA by heat may be affected by a variety of time and temperature parameters, and despite the various polymerization methods, the conversion of monomers to polymers is not complete, and some unreacted monomers, namely residual monomers, are left in the PMMA. All acrylic resins contain variable residual monomer levels, depending on the ambient conditions and the efficiency of heat transfer (3-6). Residual monomer is a well-known plasticizer and affects the physical and mechanical properties of acrylic resins. Also, residual monomer diffused from acrylic resin has been implicated as a primary irritant and a sensitizer that can cause an allergic eczematous reaction on both the skin and the oral mucosa (5-7). Several techniques such as infrared spectroscopy, gas chromatography, gas-liquid chromatography, high-performance liquid chromatography and ultraviolet spectrophotometry have been used to detect the content of residual monomer in acrylic resins (5, 8-11). Ultraviolet-visible spectrophotometry, refers to absorption spectroscopy in the ultraviolet-visible spectrum. This means it uses light in the visible and adjacent (near UV and near infrared) ranges. Ultraviolet-visible spectroscopy is routinely used in analytical chemistry for the quantitative determination of different analytes, such as transition metal ions, highly conjugated organic compounds and biological macromolecules. Determination is usually carried out in solutions (12).
Hardness measurements is an important physical property of acrylic resins, enabling these materials to be used for manufacturing denture bases that resist forces such as those arising from occlusion and mechanical denture cleansing, increasing the longevity of elderly people's dental prostheses (13, 14). The Vickers hardness test has been considered a valid tool for evaluating the hardness, viscoelastic properties and other responses of rigid polymers, and some studies have used the Vickers hardness test to detect changes in hardness of denture base acrylic resins (15-18). Measurements of acrylic hardness may indicate the possibility of polymeric matrix degradation, which will produce a decrease in acrylic hardness, increasing the possibility of fracture and diminishing the longevity of the denture base (14, 19).
There have been no studies that have investigated the effect of an alternative polymerization method using an autoclave, on the final properties of acrylic resins. Therefore, the aim of the present study was to investigate the effect of different time durations of autoclave application on the Vickers hardness and residual monomer content of 3 denture base resins. The hypothesis was that the autoclave polymerization technique of the denture bases might affect these properties of the denture base acrylic resins.
Materials and Methods
The 3 acrylic resins used in this study were conventional heat-polymerized resin. These are listed with their manufacturers, chemical compositions and group codes in Table I.
INFORMATION FOR DENTURE BASE ACRYLIC RESINS

To determine the amount of residual methyl methacrylate (MMA) monomer, 5 test specimens were prepared for each test group. A stainless steel mold with dimensions of 20 mm in diameter and 2 mm thickness was prepared to mold disk-shaped specimens from the resins.
For the 3 control groups, conventional polymerization was represented by preparing the control specimens in conventional metal flasks and then polymerizing them in a water bath for 30 minutes at 100°C according to manufacturers' instructions. For autoclave-polymerized (Ar-El Group SAN Kosmos, Greece) specimens, a dough prepared from a powder/liquid mix was packed into the metal mold within flask. Under 3 atmosphere (atm) pressure, the specimens were subjected to 1 of the processing cycles: autoclave-cured for 60°C/30 min followed 130°C/10 min, or autoclave-cured for 60°C/30 min followed by 130°C/20 min. The flasks were allowed to bench cool before opening.
Test specimens were wet ground with silicon carbide grinding papers of 200-, 400- and 600-grit size with an automatic polishing machine (Grin PO 2V grinder-polisher; Metkon A.Ş., Bursa, Turkey). All of the specimens were weighed accurately and placed in a round-bottomed glass flask, and 50 mL of methanol, the extraction solvent, was added. Glass flasks, including specimen and methanol, were connected to the Soxhlet extractor and placed in a water bath, the temperature of which was adjusted to within a range of 60-70°C, taking into account the boiling point of methanol, which is 64.5°C. Glass flasks, including specimen and methanol, were kept in the water bath for 6 hours using a heating setup, to carry out the extraction procedure.
Calibration curves (Fig. 1) were made using standard solutions (acetonitrile, methanol, water) containing known amounts of MMA monomer evaluated in methanol solution (MMA concentrations of 25-100 parts per million [ppm], ml/L, as controls). According to standard calibration curves, ultraviolet spectrophotometry at 230 nm was used to determine the residual monomer content.
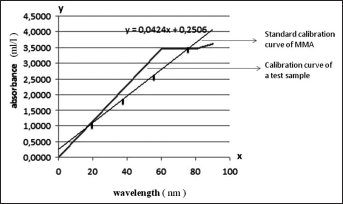
Standard calibration curves: the x-axis of the spectrum is for wavelength, and y-axis of the spectrum is absorbance. The slope of the axis shows the concentration of the residual monomer in the solution. MMA = methyl methacrylate.
Then, extracted solutions of all specimens were read at 230 nm, and according to the standard calibration curve, ppm values were calculated. Weight fractions of residual monomers were calculated for each specimen. Mean values and standard deviations were calculated for all groups of specimens.
For the Vickers hardness measurements, 5 disk-shaped specimens with dimensions of 10 mm in diameter and 2 mm thickness were prepared for each test group as described for the residual monomer test specimens. Hardness measurements were obtained with a Vickers hardness tester (Struers Duramin-1 Microhardness tester), under a 4.91 N press load for a 30-second time period, after immersion in distilled water at 37°C for 48 hours. A total of 3 indentations were made at different points on each specimen, and the means of individual specimens were averaged. For each evaluation interval, the means of individual specimens were averaged. The measurements taken with the device (in millimeters) were transferred to a microprocessor that calculates the hardness values and displays the results. All data were submitted to 1-way ANOVA and Tukey post hoc test with a 95% confidence interval.
Results
The calculated means and standard deviations of the residual monomer of test specimens are given in Table II. The statistical analysis showed that the autoclave polymerization produced a significant decrease in the amount of residual monomers for all resin groups (p<0.05). But the difference between the mean values of residual monomer content for autoclave polymerization for 10 minutes and for 20 minutes was not statistically significant for the 3 resin groups (p>0.05).
RESIDUAL METHYL METHACRYLATE MONOMER (%WT) FOR DENTURE BASE RESINS
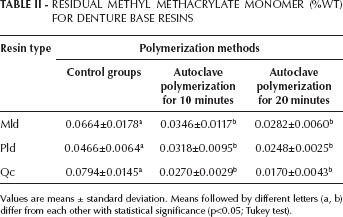
Values are means ± standard deviation. Means followed by different letters (a, b) differ from each other with statistical significance (p<0.05; Tukey test).
The results of the analysis of Vickers hardness data (Tab. III) revealed that autoclave polymerization resulted in a significant increase in hardness for all resin groups (p<0.05) according to the 1-way ANOVA and Tukey post hoc test. The Qc resin resulted in significantly higher Vickers values (p<0.05) compared with the Paladent at baseline. No significant differences were found between Mld and Pld (p>0.05).
VICKERS HARDNESS FOR DENTURE BASE RESINS
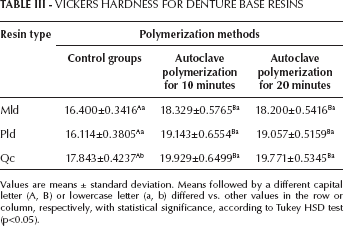
Values are means ± standard deviation. Means followed by a different capital letter (A, B) or lowercase letter (a, b) differed vs. other values in the row or column, respectively, with statistical significance, according to Tukey HSD test (p<0.05).
For the 3 resin types, no significant differences were found between autoclave polymerization for 10 minutes and for 20 minutes (p>0.05). Significantly higher Vickers values were observed at 10 and 20 minutes compared with the baseline values, in the Pld group (p<0.05). Significantly higher Vickers values were observed at 10 and 20 minutes compared with the baseline values, in the Mld group (p<0.05).
Discussion
Based on the results, the hypothesis that recently introduced autoclave polymerization techniques have lower residual monomer and higher hardness than conventional heat polymerization was confirmed. Conventional heat polymerization for PMMA denture base resins dominate laboratory procedures. New polymerization techniques must equal the approved conventional technique and also offer improved properties and advantages to be competitive. It is important to determine the residual monomer content and hardness of the tested materials and polymerization methods, as these properties influence the clinical performance of these materials.
An autoclave is an instrument used to sterilize equipment and supplies by subjecting them to high-pressure saturated steam at 121°C for around 15-20 minutes depending on the size of the load and the contents. It was invented by Charles Chamberland in 1879. Autoclaves are widely used in microbiology, medicine, veterinary science, mycology, dentistry and prosthetic fabrication (20-22).
Da Silva et al (23) analyzed the microhardness and diametral tensile strength of 2 hybrid resin composites which were polymerized with 6 laboratory photo-curing units and the results compared with an alternative polymerization method using a conventional halogen light source in conjunction with an additional polymerization in an autoclave (15 min/100°C). This resulted in a significant increase in hardness and tensile strength. The satisfactory mechanical properties obtained indicated that the autoclave was a feasible alternative for confecting indirect restorations with similar functionality but with reduced final treatment cost. Also, in our previous study (3), it was concluded that autoclave polymerization was a potential method for increasing the degree of monomer conversion and enhancing the mechanical properties of the material.
According to the results obtained in the present study, autoclave polymerization produced a significant decrease in terms of residual monomer values for all resin groups and a similar increase in hardness values, because hardness is sensitive to the residual monomer (24). Wear of dental materials is related to hardness and can be considered to be as important a parameter as the residual monomer content in clinical use. Therefore, hardness evaluation has frequently been used to predict dental material wear. Thus, the Vickers microhardness test is considered to be a valid method to evaluate rigid polymers by their ability to resist the penetration of a specific load. This is a simple and effective way to evaluate the degree of conversion during the resin polymerization reaction (13, 15, 25, 26).
The ultraviolet spectrophotometry technique, which was utilized in the present study, is a precise and simple method to determine the residual MMA monomer content. There has not been any study evaluating the residual MMA monomer content of heat polymerized denture base materials by ultraviolet spectrophotometry reported in the current literature, so it was not possible to compare the residual monomer content results with previous studies.
The studies of Hayden (27) and Farina et al (13) demonstrated that due to the incomplete monomer conversion as a result of the autopolymerization process, residual monomer acts as a plasticizer and reduces the resistance of the material. Durkan et al (28) found that the autoclave postpolymerization method increased the fracture toughness of autopolymerized acrylic resins. This increase may be related to the decrease of residual monomer content and thus elimination of the plasticizing effect.
Residual monomer is known to have a plasticizing effect which reduces the polymer interchain forces so that deformation occurs more easily under load during hardness tests. The residual monomer content can be reduced by 2 temperature-dependent mechanisms: further polymerization reaction at the sites of active radicals and diffusion of the unreacted molecules out of the resin (4, 29-31). One possible explanation for the reduction of the residual monomer content in the present study may be the diffusion of the unreacted molecules out of the polymer matrix by the pressure that was applied during autoclave polymerization.
Braun et al (32) demonstrated that the leaching of residual monomer from denture base materials contributed to the higher hardness values observed after storage in water. It is possible that the increase in hardness was related to further polymerization and residual monomer release mechanisms, which probably overcame the plasticizing effect of water uptake (32). In a previous study by Ali et al (33), the Vickers hardness number for Meliodent resin was reported (17.000) to be similar to our results in the control group (16.400). According to our results, the Vickers hardness values of the acrylic resin after autoclave polymerization were higher (for 10 minutes 18.329 and for 20 minutes 18.200) than the hardness values of the samples which were polymerized by other conventional techniques.
Some authors have suggested that to improve the physical properties of autopolymerized acrylic resins, they should be kept under pressure for at least 2.5 hours during the polymerization process, so that the resin polymerization is completed in an oxygen-reduced environment. It is suggested that resin exposure to oxygen during polymerization might compromise this process (10).
Mowery et al (34) mention that dental prosthesis polymerized by microwave energy presents a higher fit than conventionally polymerized dental prosthesis. This increased fit might be a result of immediate transformation of microwave energy into heat, after its absorption by the acrylic resin. The activation mode of polymerization significantly influences the degree of conversion of monomer to polymer (6). Bartoloni et al (5) investigated the effect of different polymerization techniques (traditional heat cure, microwave cure and rapid boil out) on the monomer conversion by Fourier transform infrared (FTIR) spectroscopy and reported that the polymerization technique used had affected the conversion values. A potential method for decreasing the residual monomer of heat-polymerized denture base resins is autoclave polymerization, and this can be easily performed in laboratory conditions.
The main purpose of decreasing residual monomer and increasing hardness of an acrylic resin used to fabricate dental prostheses for elderly people is to avoid loss of smoothness, reduce plaque retention, decrease cytotoxicity pigmentation and aesthetic problems, leading to a longer period serviceability of the dental prostheses (13, 35).
A limitation of this study was the restricted generalization of our results to other types of denture base resins – e.g., resins polymerized by different techniques. Also, we tested 2 autoclave polymerization protocols and heated the specimens for 2 different time periods, but the resin hardness and residual monomer after autoclave heating for longer periods and with other protocols are unknown. Further studies should investigate different autoclave polymerization protocols on different materials to assess if the polymerization increase effect of autoclave heating is resin-specific.
Conclusion
Within the limitations of this study, the following conclusions were drawn: There were significant differences in hardness and residual monomer content before and after autoclave polymerization in all resin groups. The autoclave polymerization method produced a significant decrease for all resin groups when compared with the conventional heat-polymerization method. Autoclave polymerization can be easily performed in laboratory conditions and can be routinely used in dental practice.
