Abstract
Background
Polymethylmethacrylate (PMMA), also known as bone cement, is a commonly used adhesive material to fix implants in Total Hip Arthroplasty (THA). During implantation, bone cement undergoes a polymerization reaction which is an exothermic reaction and results in the release of heat to the surrounding bone tissue, which ultimately leads to thermal necrosis. Necrosis in the bony tissue results in early loosening of the implant, which causes pain and reduces the life of the implant.
Purpose
The main objective of the present study was to understand the thermal isotherms in PMMA and to determine the optimal cement mantle thickness to prevent cell necrosis during THA.
Methods
In this study, the environment in the bony tissue during implantation was simulated by constructing 3D solid models to observe the temperature distribution in the bony tissue at different cement mantle thicknesses (1 mm, 3 mm and 5 mm), by applying the temperature conditions that exist during the surgery. Stems made with Co-Cr-Mo, 316L stainless steel and Ti6Al4V were used, which acted as heat sinks, and a thermal damage equation was used to measure the bone damage. FEA was conducted based on temperature conditions and thermal isotherms at different cement mantle thicknesses were obtained.
Results
Thermal isotherms derived with respect to distance in the bony tissue from the center of the cement mantle, and cell necrosis was determined at different mantle thicknesses. Based on the deduced results, cement mantle thickness of 1-5 mm does not cause thermal damage in the bony tissue.
Conclusion
Considering the long term stability of the implant, cement mantle thickness range from 3 mm-5 mm was found to be optimal in THA to prevent cell necrosis.
Introduction
Polymethylmethacrylate (PMMA) cement is widely used in orthopedic surgeries as an adhesive for fixation of prostheses to human bone. Bone cement helps in immediate fixation of prostheses, which involves an exothermic polymerization reaction (1). Two major problems which are associated with the use of bone cement are thermal necrosis due to high heat generation and chemical necrosis due to unreacted monomer (2). Many studies have pointed out that thermal bone necrosis as a serious problem which may lead to aseptic loosening of cemented prostheses (1, 3). Prosthetic loosening can be defined as an appearance of radiolucent lines between prosthesis and bone (4). There are multiple causes of aseptic loosening, ranging from thermal necrosis to stress shielding.
Temperature distribution plays an important role in the success of the cementation procedure, which affects thermal necrosis and the amount of unreacted monomer (2). An increase in the mold diameter from 0.75 to 2.0 cm results in an increase in temperature from 33°C to 80°C, which shows higher area-to-volume ratios; total volume of cement affects peak temperature of polymerization (5). Peak bone cement temperature starts near bone–cement and moves into the center of the cement mantle over the course of the reaction which shows initiation of polymerization at the bone–cement interface and moves toward the prosthesis (6). In a study by Maffezzoli et al, every point across the cement mantle shows polymerization reaction simultaneously, and a peak temperature of about 50°C is observed at the bone–cement interface, with a temperature of 120°C inside the thicker cement layers (7). During polymerization, the cement material undergoes different rheological changes where a viscous cement is transformed into a solid glass. This process involves the growth of polymer chains by monomer addition reaction, and it continues until a critical viscosity is attained by the reactive mixture based on degree of reaction (7). The point where the reaction mixture reaches critical viscosity shows the onset of gel effect. Bulk polymerization of methacrylate is significantly affected by diffusion at different degrees of conversion. Gel effect or auto-acceleration effect occurs at lower degrees of conversion and causes an increase in the rate of the reaction (8). This effect involves a mobility reduction of the chain-end radicals due to a significant increase in the higher molecular weight molecules, thereby reducing the chance of forming the dead polymers (8). The gel effect is responsible for the fast and uncontrolled heat generation when the reaction has started (7). In one study, the thick cement mantle showed maximum temperature values of approximately 45°C, compared with 33°C for a thin cement mantle (4). The temperature in the bone-marrow region (307 K) is lower than the peak temperature in the cement (337 K) (2). The generation of temperature during the curing of bone cement depends on the balance between the rate of heat production and the rate of heat transfer (9). The higher the initial temperature, the higher rate of heat transfer from cement to the bone (10). The amount of monomer left over from the polymerization is observed to be inversely proportional to the cement mantle thickness (8). The temperature has been found to be highest in the proximal part of femur, decreasing distally (10).
A rise in temperature may cause tissue injury but reduces the amount of unreacted monomer by attaining onset temperature resulting in maximum degree of reaction (7). When the level of thermal exposure by the bony tissue gets higher, the time in which the cell undergoes necrosis shortens (11). An increase in cement thickness affects the exposure time at the highest temperature rather than the maximum value of the temperature at the bone–cement interface (7). The higher the starting temperature, the higher the maximum temperature reached and the greater the degree of monomer conversion (8). The thinner the cement layer, the smaller the necrotic area, but the implant fixation strength is compromised (12). The thermal properties of the prosthesis are important to consider because a metal prosthesis acts as a heat sink in cemented hip implants (6). The initial temperatures of bone, cement and prosthesis are important to consider for analysis because they can affect the success of the hip replacement surgery (13).
Preheating the prosthesis stem before implantation decreases voids near the cement–prosthesis interface, but it leads to a weaker bone–cement interface due to reversal in the polymerization direction (13). Precooling the cement–prosthesis leads to a longer setting time for bone cement and an increase in the amount of unreacted monomer concentration (13). The setting temperature of bone cement not only depends on the heat generated during polymerization but also on the heat conducted to the surrounding area (14). In a study by Borzacchiello et al, the maximum temperature was below 50°C even with the highest starting temperature and the thickest cement mantle, and the degree of conversion was about 90% with the lowest starting temperature and thinner cement mantle (8). While curing, lower temperature increment was seen in case of PMMA with either BaSO4 or MgO nanoparticles when compared with pure PMMA (15). The parameters that control the temperature at the bone–cement interface are still under research. The main objective of the present study was to understand the thermal isotherms in PMMA and to determine the optimal cement mantle thickness to prevent thermal damage in bony tissue during total hip arthroplasty (THA). The present work showed the thermal isotherms in a hip model by simulating the conditions that exist during THA, and the percentage of cell necrosis was determined using a damage function at different cement mantle thicknesses.
Materials and Methods
Human femur bone
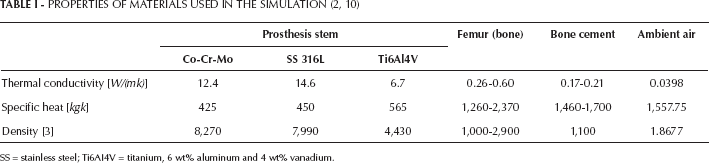
During THA, the proximal part of the human femur is removed and replaced with a metal stem. Cortical bone or compact bone is 70% to 90% mineralized. It is present in the shaft of a human bone surrounding the marrow cavity. Cancellous bone is usually found internal to the compact/cortical bone, and it is also called spongy bone. The thermal conductivity of the femur bone lies between 0.26-0.60 [W/(mk)].
Cement
PMMA bone cement preparation involves mixing the solid component, powder, with the liquid component in the operating room. The ratio is usually 2 g of powder to 1 mL of liquid; however, this composition can vary depending on the type of cement being used. Usually the solid part is the polymer, PMMA, plus the initiator, benzoyl peroxide, and the liquid part is the pure monomer, Methyl methacrylate (MMA), plus the activator. During the polymerization process, monomer MMA is converted into PMMA, which involves an exothermic reaction as shown in Figure 1. The thermal conductivity of PMMA lies in the range 0.17-0.21 [W/(mk)].
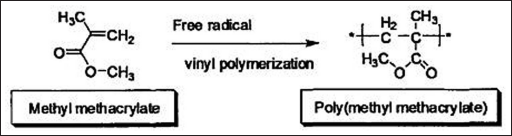
Polymerization reaction of methylmethacrylate (reproduced with permission from 16).
Device
The hip implant device was modeled after actual devices used in patients. They are usually made up of titanium alloys, stainless steel (316L) or cobalt chromium alloys. For the purposes of this analysis, Co-Cr-Mo, 316L stainless steel (SS 316L) and Ti6Al4V (titanium, 6 wt% aluminum and 4 wt% vanadium) devices were used. Co-Cr-Mo alloys are widely used in knee implants, hip joints and dental prosthetics. The thermal conductivity of Co-Cr-Mo alloy is 12.4 [W/(mk)]. SS 316L is composed of different elements like chromium, nickel, molybdenum etc. The thermal conductivity of SS 316L is 14.6 [W/(mk)]. Ti6Al4V is used in joint replacement components because of its superior corrosion resistance properties compared with stainless steel and Co-Cr-Mo alloys. When compared with stainless steel and Co-Cr-Mo alloys, Ti6Al4V provides less stress shielding because of its axial and torsional stiffness which are close to the stiffness properties of bone (17). The thermal conductivity of Ti6Al4V is 6.7 [W/(mk)]. The cement layer was in very close proximity to the femur cortical bone layer and the hip model, as shown in Figure 2a. The cortical bone took most of the thermal damage during the device implantation, causing loosening and other damage to the femur bone. In addition to thermal necrosis, the metal debris generated due to wear of the prosthetic surface causes bone resorption which resulted in aseptic loosening of the implant also observed in cemented TPA (18).
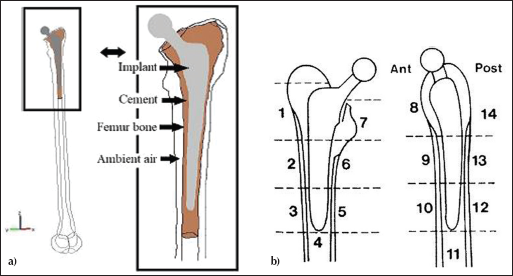
a) Different layers present in the hip model (reproduced with permission from 10), and b) Gruen zones in anteroposterior and lateral planes (reproduced with permission from 19).
Solid models
Different cement mantle thicknesses were considered ranging from 1 to 5 mm to observe the necrosis of bone tissue due to the generation of heat. The generation of heat varies with cement mantle thickness, which was observed in this study in different Gruen zones as shown in Figure 2b.
Stems were designed based on the parameters used by Patel and Goswami (20), and each stem is inserted into a solid model of PMMA hollow cylinder with mantle of thickness ranging from 1 to 5 mm, using Solidworks 2010 ×64 as shown in Figure 3. The designed Solidworks stem models were then imported into ABAQUS as a “.step” file (STEP AP203) for analysis.

Femur models with different cement mantle thicknesses 1, 3 and 5 mm (from left to right).
Loading conditions
Peak temperatures at the bone–cement interface were considered based on the study by Vallo (9), with initial temperature of 25°C for bone, cement and metal stem for analysis. A layer of ambient air at a temperature of 298 K separates the femur bone and bone cement. The implant was assumed to be at a room temperature of 298 K upon placement into the femur, because the surgical field had been exposed to room temperature (297 K - 301 K) during the implantation (9). The cement–bone interface was at 278 K which was warmer when compared with the cement–stem interface, while the outer surface of the bone was at 310 K (21). The temperatures that existed at the bone–cement interface were also assumed at the stem–cement interface. The difference in temperature produces a gradient that allows for convection to occur as the cement cures. During convection, the bone tissue encounters a maximum temperature of exposure. This maximum exposure for 1 second can cause necrosis to occur.
Finite element analysis
Finite element analysis was performed using ABAQUS to analyze the thermal efflux produced from the cement mantle into the femoral cavity by doing heat transfer in the STEP module. The thermal properties and density of different components of the hip model are shown in Table I.
SS = stainless steel; Ti6AI4V = titanium, 6 wt% aluminum and 4 wt% vanadium.
Using the thermal film coefficients provided in Table II and sink temperatures, heat transfer analysis was implemented. The assembly was then meshed using a seed size of 3 with tetrahedral elements. Thermal analysis was then performed by creating the nodes at areas of interest. Output graphics show the maximum temperature reached at each section of the femur during the heat transfer process with respect to above-mentioned temperature conditions (corresponding data is shown in the results section).
VALUES OF INTERFACE CONDUCTIVITIES/CONVECTION FILM COEFFICIENTS IN [(2 K)] (10)

Necrosis damage function
After the thermal analysis, the temperatures were measured at different regions of the assembly using nodal points. The temperatures that were generated in the femur bone due to heat efflux from the cement mantle were measured and substituted in the damage function given by Fukushima et al (12) to identify the amount of damage. The relationship between the temperature and the percentage of bone necrosis was calculated through the following equation.
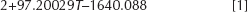
Where D(T) represents the damage function, while T represents the specific temperature of the bone. The equation shows the necrosis damage function with respect to temperature (12).
Results
By applying the boundary conditions mentioned in the previous section, the heat transfer across the hip model was measured. The temperatures at the center of the cement mantle for different thicknesses are shown in Table III, and it can be observed that with an increase in the cement mantle thickness, the core temperature of the mantle also increased.
TEMPERATURE VALUES OBSERVED IN THA AT THE CENTER OF CEMENT FOR DIFFERENT MANTLE THICKNESS

THA = total hip arthroplasty.
The heat transfer across the bone was measured from the center of the cement mantle in all of the Gruen zones, using the models submitted to heat transfer analysis, and the corresponding results were plotted in Figures 4-7 by selecting individual nodes across the layers present in the hip model in which the temperature varied at different cement mantle thicknesses. Figures 4-7 show a drop in temperature from the center of the cement, which had a maximum temperature (i.e., 345 K at 5-mm cement mantle thickness), toward the bone–cement interface (i.e., 325 K at 5-mm cement mantle thickness). It decreased gradually to 298 K in the bone domain, and no significant drop in temperature was observed for 1-mm and 3-mm cement mantle thicknesses.
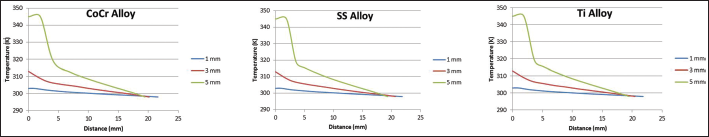
Distribution of temperatures at different cement mantle thicknesses with different implant stem alloys in Gruen zone 7.
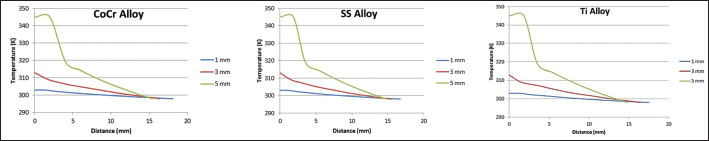
Distribution of temperatures at different cement mantle thicknesses with different implant stem alloys in Gruen zones 2, 6, 9 and 13.
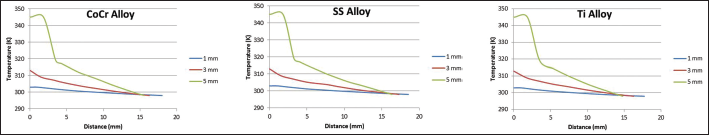
Distribution of temperatures at different cement mantle thicknesses with different implant stem alloys in Gruen zones 3, 5, 10 and 12.
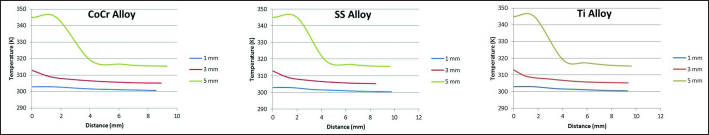
Distribution of temperatures at different cement mantle thicknesses with different implant stem alloys in Gruen zones 4 and 11.
Gruen zones 4 and 11 showed the distribution of the temperature at the base of the implant. At a distance of 6 mm from the bone–cement interface, temperatures of approximately 301 K, 306 K and 319 K were measured for cement mantle thicknesses of 1 mm, 3 mm and 5 mm, respectively, as shown in Figure 7.
Temperature distribution from Figures 8-10 varied from 298 K to 340 K across the different layers of the models. It can be observed from Figures 8-10 that the bone–cement interface temperatures never exceeded 320 K (47°C), and a temperature of 320 K was observed at the bone–cement interface for the cement mantle thickness of 5 mm.
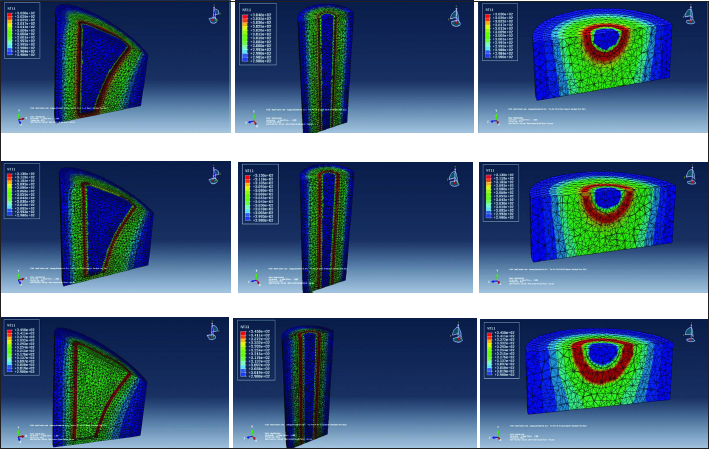
Temperature distributions in the bone domain at neck (top row), stem (middle row) and base (bottom row) of the Co-Cr-Mo stem at different cement mantle thicknesses (column wise).
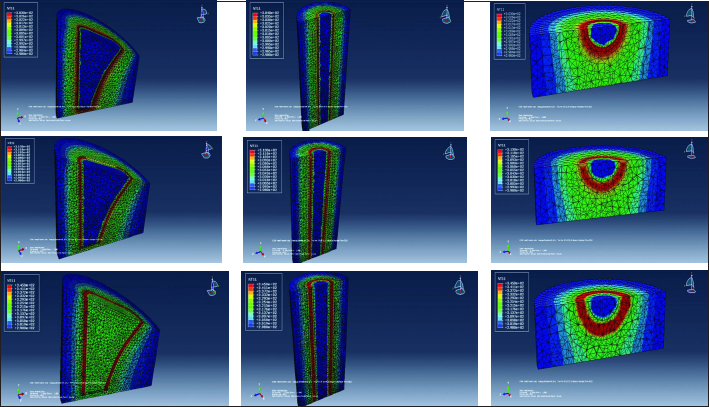
Temperature distributions in the bone domain at neck (top row), stem (middle row) and base (bottom row) of the SS 316L stem at different cement mantle thicknesses (column wise).
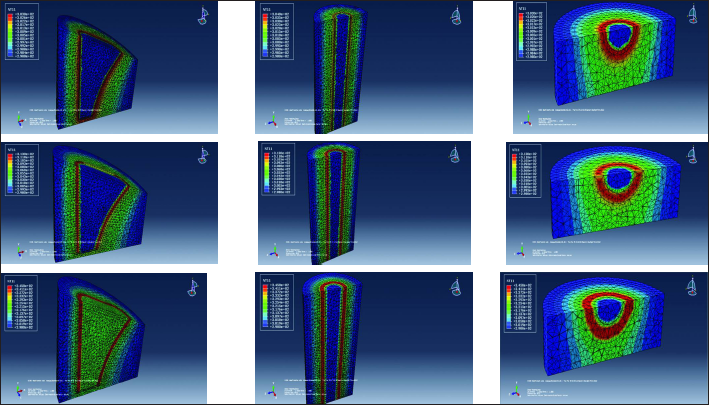
Temperature distributions in the bone domain at neck (top row), stem (middle row) and base (bottom row) of the Ti6Al4V stem at different cement mantle thicknesses (column wise).
In Gruen zones 1 and 7, as shown in the Figures 8-10, it can be observed that for 1-mm and 3-mm cement mantle thickness, the average temperatures across the bone and prosthesis regions of the hip model were the same, and also there was a significant difference in these values between the two regions for the 5-mm cement mantle thickness.
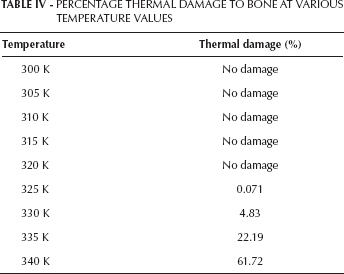
The percentage of thermal damage of the bone was calculated by substituting temperature values ranging from 300 K to 340 K in Equation 1, and the resultant values showed 0% to 61.72% cell damage occurring at different temperatures. These values are tabulated in Table IV.
PERCENTAGE THERMAL DAMAGE TO BONE AT VARIOUS TEMPERATURE VALUES
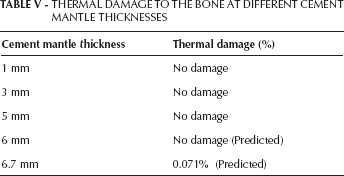
Temperatures existing in the bone region for different cement mantle thicknesses were substituted into Equation 1, and the corresponding results showed no damage in the surrounding bone tissue due to thermal necrosis at the boundary conditions mentioned for different cement mantle thicknesses. These values are provided in Table V.
THERMAL DAMAGE TO THE BONE AT DIFFERENT CEMENT MANTLE THICKNESSES
The temperature at a certain distance from the bone–cement interface can be measured using the following regression equations 2, 3 and 4 generated for different cement mantle thicknesses. These equations were derived based on the resultant temperatures existing across the bony tissue for a particular cement mantle thickness.
For 1-mm thickness:
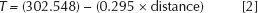
For 3-mm thickness:
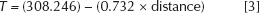
For 5-mm thickness:
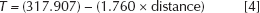
These regression equations could help in determining the range of temperatures that exists in the bone region for the cement mantle thicknesses values. Based on the obtained results, a regression equation was generated relating the temperature at the bone–cement interface with the cement mantle thickness. The following equation helps in determining the temperature at the bone–cement interface to predict the occurrence of thermal necrosis for different cement mantle thicknesses.
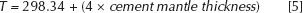
The above equation is used to determine the minimal cement mantle thickness that causes thermal necrosis in the bony tissue, and the predicted values are provided in Table V.
Discussion
Many studies have demonstrated that polymerization of cement causes thermal necrosis in the surrounding bone tissue, which leads to earlier failure of the implant. The amount of polymerization depends on the thickness of the cement mantle around the implant and the initial temperatures of the components in the THA. So there is a need to optimize the process to prevent necrosis in bony tissue by considering the factors that play a major role. Three different cement mantle thickness scenarios for hip implants made of 3 different metals (Co-Cr-Mo, SS 316L and Ti6Al4V) were investigated for thermal necrosis. In this study, the environment present during a THA was simulated, and the temperature distribution in the surrounding bone tissue was observed. Based on the temperature at the cement–bone interface and the temperature in the center of the cement mantle, these models were analyzed for thermal necrosis based on the above-mentioned conditions.
From the results, it was observed that with an increase in the cement mantle thickness, the temperature at the bone–cement interface also increased, and the temperature distribution in the surrounding bone tissue varied. There was a significant difference between the different cement mantles used in this study in terms of the maximum temperatures at the cement–bone interface and also at the center of the cement mantle. Use of different metal stem materials in this study did not show any significant difference in temperature distribution in any Gruen zones, and these observations are supported by the results of a study done by Kaorapapong et al (10) with similar materials. The implant materials used in that study showed very less effect on the heat transfer process. So based on these observations, we can conclude that the stem materials that are commonly used in THA do not show any significant effect on the thermal isotherms. No significant drop in temperature was observed in the hip models for 1-mm and 3-mm cement mantle thicknesses, because the temperature at the bone–cement interface was very close to the temperature at the center of the cement. Based on the thermal contours in Gruen zone 1 and 7, we can observe that the prostheses exhibited their heat sink properties significantly at higher polymerization temperatures or at higher cement mantle thickness. From Table IV, we can observe that a temperature around 325 K (52°C) caused thermal necrosis in the surrounding bone tissue. Equations 2, 3 and 4 showed degradation of temperature, (a factor of distance from the bone-cement interface) increases with increase in the cement mantle thickness. The temperature distribution measured at Gruen zones 4 and 11 showed a significant distribution of temperatures over long distances in the bony tissue, which resulted from the presence of greater amounts of bone–cement at the base of the metal stem during implant fixation.
A study done by Vallo (9) deduced that a temperature around 323 K (50°C) causes necrosis in the surrounding bone tissue at a cement mantle thickness of 6 mm. In another study, a temperature higher than 50°C was determined for a cement thickness of 7 mm (22). The regression equation generated in this present study predicted a temperature of 325 K at the bone–cement interface for a cement mantle thickness of 6.7 mm, and the results obtained closely matched the results from the previous studies mentioned above. The small difference between the results could be due to some variance in the thermal damage equation where it predicted the minimal thermal damage at 325 K or due to different boundary conditions considered for the study. So based on the results obtained at greater cement mantle thicknesses – i.e., at 6 mm or more – there will be significant thermal necrosis in all of the Gruen zones due to any generation of temperatures higher than 52°C at the bone–cement interface.
Cement mantle thickness has been directly correlated with loosening of the implant both from mechanical and thermal perspectives. Several biomechanical studies have deduced that cement mantle thickness of 3-5 mm was the optimal range for better implant stability by preventing femoral component loosening (23, 24). The practice of precooling and preheating the prosthesis before implantation has certain disadvantages (13). Based on the results obtained here, this study has deduced that a cement mantle thickness of 1-5 mm does not cause thermal damage in the bony tissue. Taking the stability of the implant into consideration, a cement mantle thickness of 3-5 mm is the optimal thickness range that helps in preventing thermal necrosis through a compensating precooling/preheating procedure and also provides long-term stability to the implant.
Conclusions
Analysis shows that with an increase in the cement mantle thickness, thermal necrosis in the surrounding bone tissue increases. The material type did not play a significant role in affecting the temperature fields. The initial temperature conditions of individual components in THA may affect the polymerization reaction and temperature distribution in both metal components and in the surrounding bone tissue. In this study, the maximum temperature was generated at a mantle thickness of 5 mm.
A cement mantle thickness range from 3 mm to 5 mm was found to be optimal in THA. Any increase in the cement mantle thickness causes an increase in bone loss. There was no thermal damage at the optimal cement mantle thickness range of 1-5 mm investigated in this study. For long-term stability of the implant, a cement mantle thickness of 3-5 mm is desired.