Abstract
Aim
The purpose of this study was to evaluate the effect of different surface treatments on the bond strength of veneering ceramic to zirconia.
Methods
Square (15 mm x 10 mm x 2 mm) zirconia specimens (n=18) received one of the following surface treatments: Group 1, sandblasting; Group 2, Clearfil ceramic primer application; Group 3, grinding; Group 4, alloy primer application; and Group 5, RelyX ceramic primer application. The zirconia core specimens were layered with a veneering porcelain (5 mm x 3 mm x 3 mm). Mean shear bond strength values (MPa) were calculated. Data were analyzed with one-way ANOVA and Tukey's post hoc pairwise comparisons (α=0.05). Fractured surfaces of the specimens were examined with scanning electron microscope (SEM).
Results
This study showed that bond strengths of the metal primer-treated zirconia specimens (Group 4) were significantly higher than those of the other paired groups. The application of metal primer affected the specimens' failure mode. SEM analysis demonstrated that Group 4 had mainly cohesive fractures, while the other groups showed approximately equal levels of adhesive and mixed fracture types. The mean and SD values for shear bond strengths ranged from 8.90 ± 3.42 MPa (Group 2) to 19.74 ± 4.96 MPa (Group 4).
Conclusions
In conclusion, the application of a metal primer to a zirconia core increased the bond strength of veneering ceramics. The use of chemical agents to improve the strength of the zirconia core's bond to veneering ceramic may have more benefits than the use of mechanical pretreatments.
Introduction
Interest in zirconium oxide (zirconia) ceramics has shown significant growth in recent times and has the potential to have a major impact on the field of prosthodontics. Zirconia has been used as a metal-free alternative because of its mechanical properties, such as high flexural strength (1.0-1.2 GPa) and toughness (7-8 MPa m0.5), biocompatibility, chemical and structural stability, and optical properties (1-2-3-4-5-6-7-8-9-10). In particular, yttrium oxide partially stabilized tetragonal zirconia polycrystalline (Y-TZP) is among the most commonly used all-ceramic core materials, and it can be fabricated using the computer-aided design and computer-aided manufacturing (CAD-CAM) technique (2, 4, 6).
The dental literature contains a wide range of comprehensive systematic reviews focusing on the survival rates of all-ceramic restorations in comparison with metal-ceramic ones (2, 11). In comparison with all-ceramic restorations, which have a 93.3% survival rate, metal-ceramic prostheses have more favorable survival rates (95.6%) after 5 years of clinical use (2). Among the ceramic prosthesis types, zirconia-based prostheses have reportedly shown the best clinical performance and have been shown to be the most reliable all-ceramic system even after 5 years of observation (2, 11). Although other all-ceramic restorations show some framework fractures, zirconia is affected only by cracking or chipping of the veneering ceramic (2, 11, 12). After 3 and 5 years, 13.0% and 15.2% of patients, respectively, were found to have clinical failures in their veneered Y-TZP frameworks (i.e., chipping and/or delamination of veneering ceramic) (8, 13, 14). Hence, sufficient bond strength between the veneering ceramic and the substructure is considered an important factor in the long-term clinical success of zirconia restorations. Fischer et al (10) reported that the bond strength between zirconia and the veneering ceramic is determined by a range of factors, such as chemical bonds, mechanical interlocking, type and concentration of defects at the interface, wetting properties and the degree of compressive stress in the veneering layer because of the significant difference in the exponent factors of thermal expansion between zirconia and the ceramic.
Because the adherence rate of the veneering ceramic to the zirconia substrate has proven to be a key factor in the long-term performance of zirconia restorations, the bond strength gained from in vitro investigations can provide useful information about the behavior and predictability of Y-TZP all-ceramic systems in clinical application (15). Many manufacturers and researchers have attempted to modify the surface properties of zirconia mechanically and chemically to improve its bonding via various surface treatments (6, 8, 10, 16-17-18-19-20-21-22-23-24-25-26-27). Recent studies were summarized in Table I.
In several studies of zirconia ceramic bonding, airborne particle abrasion was used to increase the surface roughness and bonding to the veneering ceramic (17, 18, 32); however, the results of these studies are controversial. Teng et al (18) investigated the effects of different surface conditioning techniques on the bond strength and demonstrated that airborne particle abrasion did not create any significant differences in bonding to ceramic compared with other techniques. Kim et al (8) demonstrated that airborne particle abrasion resulted in significantly higher bond strengths than liner application did. Fischer et al (10) showed that increasing the surface roughness of zirconia and applying a liner did not enhance shear strength. Another study by Fischer et al (22) demonstrated that the use of a liner on Y-TZP cores had no significant effect on bond strength. Although Mosharraf et al (21) showed that the type of zirconia did not have any effect on the bond strength between the zirconia core and the veneer ceramic, grinding dramatically decreased the shear bond strength of white zirconia porcelain.
STUDY CHARACTERISTICS AND OUTCOMES OF THE RECENT STUDIES

MDP = 10-methacryloyloxydecryl dihydrogen phosphate.
To mask the framework and to increase the wetting property of the zirconia surface, liners can be applied as an intermediate layer between the zirconia substrate and the veneering ceramic. Contrary to some studies that showed that liner application improves the bond strength between the zirconia and resin cements (7, 16, 28), other reports have claimed that this application has no effect on the bond strength between the zirconia and the veneering ceramic and may even weaken it (8, 17, 22).
In comparison, various adhesive monomers, such as metal primers, have been developed to permit chemical bonding. An alloy primer composed of 10-methacryloyloxydecryl dihydrogen phosphate (MDP) and 6-(4-vinylbenzyl-n-propyl amino)-1,3,5-triazine-2,4-dithione (VBATDT) in acetone improves the bonding to air-abraded oxide ceramics (7, 16, 19). Metal primers usually bond to pure metals and alloys because of their affinity to metal oxides on metal surfaces (29). Some studies have shown that a MDP monomer promotes bonding to air-abraded oxide ceramic (6, 7, 16, 28, 29). Nevertheless, the question of whether MDP monomer—containing primer can advance the bond between ceramic and zirconia remains unresolved.
Other commercially available silane coupling agents exist that contain monomers like 3-methacryloxypropil trimethoxy silane, which are capable of bonding to zirconia. Although a previous study showed that the use of this agent without air abrasion did not promote long-term tensile bond strength (16), recent studies have shown that experimental silane monomer primers can significantly increase the bond strengths between resins and a zirconia surface which was coated with glass powder (30), or treated with tribochemical silica (31).
Although manufacturers recommend airborne particle abrasion or liner application as surface treatments for zirconia, the effects of these methods have not yet to be clarified. The effect of metal and ceramic primers on the bond strength of zirconia to the veneering ceramic has not been demonstrated. Therefore, the aim of this study was to evaluate the effect of different surface treatments on bond strengths between Y-TZP ceramics and the veneering ceramic. The null hypothesis was that surface treatments might improve the shear bond strength between the zirconia and the ceramic.
Materials and Methods
Preparation of Specimens
The materials used in this study are presented in Table II. One type of zirconia-based ceramic (Zirkonzahn; Steger, Ahrntal, Italy), 1 type of veneering ceramic (Vita VM9; Vita Zahnfabrik, Bad Sackingen, Germany) and 3 types of primer (Clearfil ceramic primer; Kuraray, Tokyo, Japan; RelyX ceramic primer; 3M ESPE, St. Paul, MN, USA; and alloy primer; Kuraray, Tokyo, Japan) were selected for this study. Pre-sintered Zirkonzahn zirconium oxide blocks were milled according to the manufacturer's instructions. Sintering was performed at 1500°C in a furnace (Kavo Everest Therm; Kavo Dental GMBH) according to the cycle recommended by the manufacturer. Dense sintered blocks were cut in the green state using a low-speed diamond disc (Izomet 1000; Buehler, Lake Bluff, IL, USA). A total of 90 square zirconia specimens (15×10×2 mm) were produced (n=18). The bonding surfaces of the zirconia core specimens were polished, ground with 1,200-grit silicon carbide abrasives under water cooling, and ultrasonically cleaned in distilled water for 5 minutes.
MATERIALS USED IN THIS STUDY
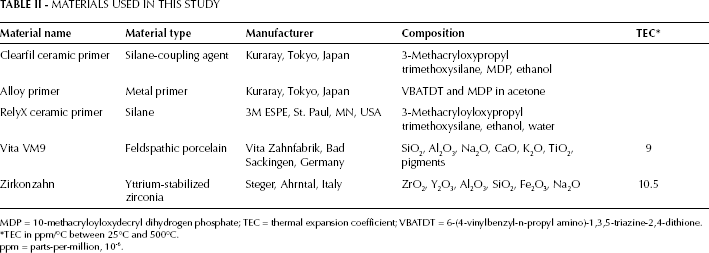
MDP = 10-methacryloyloxydecryl dihydrogen phosphate; TEC = thermal expansion coefficient; VBATDT = 6-(4-vinylbenzyl-n-propyl amino)-1,3,5-triazine-2,4-dithione.
TEC in ppm/°C between 25°C and 500°C.
ppm = parts-per-million, 10-6.
Surface Conditioning and the Bonding Procedure
The specimens were randomly divided into 5 groups of 18 specimens each (n=18), which received the following treatments for the bonding surfaces:
Group 1: The zirconia was sandblasted with 120-μm Al2O3 particles at 3.5 bar pressure for 10 seconds from a distance of 10 mm perpendicular to the surface.
Group 2: The zirconia was treated with a single-component adhesive primer (Clearfil ceramic primer).
Group 3: The zirconia was ground with a diamond bur with a 100-μm grain size (Bredent, Senden, Germany) at a speed of 20,000 rpm under water cooling. The bars were replaced after the grinding of every fifth specimen to maintain a consistent amount of grit.
Group 4: The zirconia was treated with a metal primer (alloy primer).
Group 5: The zirconia was treated with a prehydrolyzed silane-based primer (RelyX ceramic primer).
Each primer was applied to the bonding surface according to the manufacturers' recommendations. The prepared zirconia specimens were placed in a separable stainless steel mold with a 5-mm clearance in the diameter and a 3-mm height above the core material to condense the veneer ceramic. The veneering procedure was performed using the manual layering technique. The intaglio surface of the mold was isolated (Ceramic Separating Stick; Ivoclar Vivadent AG, Schaan, Liechtenstein) to avoid the adhesion of ceramic powder to the mold during layering. The ceramic powder was mixed on a glass slab with an appropriate amount of the respective liquid, as is commonly done in a dental laboratory, and condensed into the mold. Only the dentin porcelain was layered, not the enamel porcelain. The veneering ceramic was fired according to the firing program of the manufacturer (Vita Vacumat 4000 Premium T; Vita Zahnfabrik, Bad Säckingen, Germany). After the first layer was fired, a second layer was applied. To compensate for firing shrinkage, the previous step was repeated to produce a final veneer thickness of 3 mm and a diameter of 5 mm. The final dimensions were measured with a digital micrometer (Mitutoyo Digimatic Caliper; Mitutoyo Corp., Kawasaki, Japan).
Shear bond Strength Test
Using acrylic resin (Meliodent; Heraeus Kulzer GmbH, Hanau, Germany), each specimen was embedded at the center of a metal ring holder 13 mm in height and 15 mm in diameter with the core—veneer interface positioned at the top level of the holder. These metal holders were then mounted in the jig of a universal testing machine (Model 4202; Instron Corp., Norwood, MA, USA). All of the specimens were tightened and stabilized to ensure that the edge of the shearing rod was positioned as close to the core—ceramic interface as possible. The load was applied parallel to the long axis of the specimen through a wedge at the core—veneer interface at a crosshead speed of 0.5 mm/min until the veneering ceramic was delaminated. The shear bond strength (S) values (expressed in MPa) were calculated using the formula S=L/A, where L is the load at failure (in N) and A is the adhesive area (in mm2) measured with a digital micrometer. Shear load at failure was recorded. The fractured specimens were examined for surface topography and further failure mode analysis using a scanning electron microscope (SEM; EVO L10; Carl Zeiss, Oberkochen, Germany). Digital pictures of these specimens were taken at various magnifications to evaluate the fracture surfaces and to verify the modes of failure. A failure between the zirconium and veneering ceramic was defined as an adhesive failure. A failure within either the framework or the veneering ceramic material was defined as a cohesive failure. The term mixed failure was used to describe the combination of these 2 failure types. Specific specimens were selected for the chemical surface structure of zirconia using energy-dispersive X-ray spectroscopy (EDS) analysis. Statistical analyses were performed with SPSS for Windows 15.0 (SPSS Inc., Chicago, IL, USA). The bond strength data obtained from the 5 groups were analyzed with a 1-way analysis of variance (ANOVA). Tukey's post hoc test was used to detect pairwise differences among the groups at the confidence interval of 95%.
Results
Shear Bond Strength
The mean and standard deviation (SD) shear bond strength values of the test specimens are shown in Table III.
SHEAR BOND STRENGTH VALUES (IN MPa) AND FAILURE MODES, EXPRESSED AS PERCENTAGES
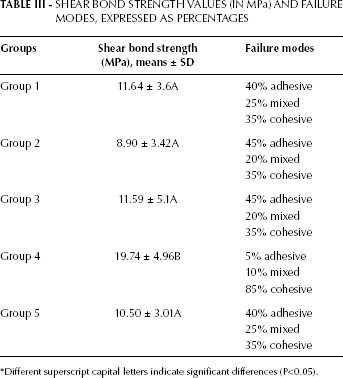
Different superscript capital letters indicate significant differences (P<0.05).
The mean and SD values of the shear bond strengths ranged from 8.90 ± 3.42 MPa (group 2) to 19.74 ± 4.96 MPa (group 4). One-way ANOVA showed significant differences among the 5 groups (P<0.05). Tukey's post hoc test revealed that group 4 had a significantly higher bond strength than did the other groups (P<0.001), while no significant differences were found among groups 1, 2, 3 and 5 (P>0.05). The shear bond strength values of the tested groups were as follows: group 4> group 1> group 3> group 5> group 2. The application of a metal primer also resulted in an increase in the bond strength (P<0.001).
SEM Analysis for the Failure Mode
Table III summarizes the failure modes of the specimens. All of the groups showed a combination of cohesive, adhesive and mixed failure modes. Cohesive fracture was the principal failure mode of the metal primer—applied group (group 4), while in the other groups, the failures were divided approximately equally between adhesive, cohesive and mixed modes. Mixed fractures were observed much less frequently than the other fracture modes in all groups. A thin layer of the veneering ceramic covering the zirconia surface confirmed cohesive failure within the veneering ceramic. The veneering material remaining on the zirconia surface was clearly visible (Fig. 1). The EDS spectra analysis showed the presence of ceramic (identified by distinguishing the compounds of silicium into silica, calcium, sodium, aluminum and potassium on the zirconia fracture surface; Fig. 2). This observation confirmed that the failure was of a cohesive nature in group 4. SEM images of ground or ceramic primer-treated zirconia specimens demonstrated adhesive fractures on the surfaces (Fig. 3a, b). SEM analysis of the zirconia-veneer surface revealed that structural defects can interrupt the contact between the zirconia and the ceramic (Fig. 4). Representative SEM micrographs of the fractured specimens demonstrated the mixed failure mode of the air-abraded and silane coupling agent—treated groups (Fig. 5a, b).

SEM image of the fractured specimen in group 4. The veneering ceramic remaining on the zirconia surface is clearly visible (magnification ×50).
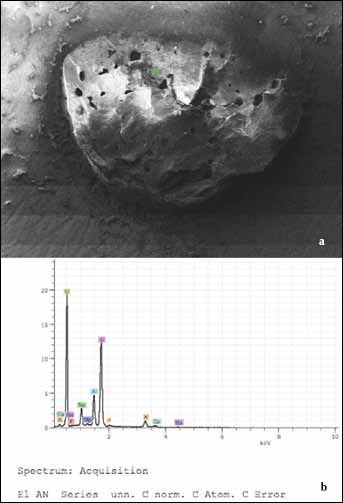
SEM (a) and energy-dispersive X-ray spectroscopy (EDS) (b) micrographs of the zirconia specimen showing the cohesive failure in group 4 (magnification ×50). EDS spectrum (green point) illustrating the presence of silica, calcium, sodium, barium, aluminum and potassium components of the ceramics on the fractured surface.

Adhesive failure with ceramic residue on the zirconia surface around the fractured area was found in group 2 (a) and group 3 (b) (magnification ×50).

SEM image of the zirconia surface showed defects and pores in the ceramic area in group 3 (magnification ×500).

SEM images of the mixed failure type in group 1 (a) and group 5 (b) (magnification ×50).
Discussion
The present study demonstrated that the application of a metal primer provided higher bond strengths compared with other surface treatment methods. Furthermore, sandblasting with 120-μm Al2O3 particles, grinding with a diamond bur or applying ceramic primers (Clearfil or RelyX ceramic primers) did not improve the ceramic's bond strength to zirconia. Therefore, the null hypothesis stating that sandblasting, grinding, ceramic or metal primer application has significant effects on the shear bond strength between zirconia and porcelain was partially rejected.
Core—veneer bond strength has a noteworthy effect on the success of ceramic restorations. The strength of the bond is usually determined by its weakest component. Previously, different approaches and techniques have been examined (1, 3-4-5-6-7-8-9-10, 17, 18, 20, 21). The mechanical interlocking of the bonding mechanism between the zirconia and the veneering porcelains seems to be essential (18), even though this mechanism is not yet fully understood. However, Fischer et al (10) have stated that zirconia and the veneering ceramic bond chemically. Moreover, some studies have reported that surface treatment of the zirconia, including mechanical interlocking (e.g., airborne particle abrasion), grinding or chemical bonds (e.g., the application of a liner), had significant effects on the bond strength (3, 5, 8, 17, 18, 21). Still, the particular factors of the bonding mechanism between the zirconia and the veneering ceramic have not been presented.
Because the air abrasion technique could increase the surface energy, surface area for bonding and wettability, it is widely used on zirconia to increase surface roughness. In the present study, airborne particle abrasion with 120-μm Al2O3 particles did not increase the bond strength between the zirconia and the veneering ceramic. This result is in agreement with previous studies (10, 18, 22). Similar to our study, Guess et al (4) and Tada et al (33) reported that surface roughening with alumina abrasion had no significant effects on the bond strength between the core and the veneer. Although some studies have recommended using air abrasion to increase the bond strength between the zirconia and the porcelain (8, 32), others have reported that air abrasion also results in a phase transition that transforms the zirconia crystal structure from tetragonal to monoclinic (7, 18, 24, 34). These crystal structures exhibit changes in the coefficient of thermal expansion (CTE) of zirconia (7, 18, 24, 34). When porcelain is fused to the bulk substructure material, attention must be paid to ensure that the CTE values of the core materials match those of the veneering porcelain. A large difference in the CTE between the porcelain veneer and core materials can cause residual tensile stresses, resulting in veneer chipping (35, 36). Moreover, this monoclinic phase layer created by air abrasion may contain microcracks, flaws and surface lifts (24). Such surface characteristics, even at microscopic levels, can decrease the core—veneer bond strength (3, 7, 32). Therefore, changes to the CTE of the zirconia surface likely hampered the recovery of the bond strength of the zirconia framework to the veneering porcelain, despite an increase in surface roughness.
The tetragonal-monoclinic transformation of zirconia may also occur during grinding (33). Grinding, especially with coarse diamonds, causes some tetragonal-monoclinic transformation on the surface of the zirconia, thereby decreasing the reliability and strength of the zirconia material with increased surface roughness (30, 37, 39). The reduced strength and the content of the monoclinic phase might be the possible results of grinding that caused the local development of temperature changes (24). This not only affects the strength of the structure but also may affect the core—veneer bond strength (36). In contrast to the assumption that rougher surfaces provide higher bond strengths by creating a larger surface area for bonding, the results of the present study showed that surface grinding did not affect bond strength. Grinding has been reported to produce high stress and uneven distributions of large defects and microcracks, all of which can reduce bond strength (24). A previous study showed that grinding significantly decreased the bond strength of the zirconia and the veneering ceramic (21). According to the results of the present study, such mechanical roughening methods as grinding and air abrasion, failed to produce improvements in the bond strengths between the zirconia substructure and the veneering porcelain.
Because the aforementioned surface treatment methods alone are not capable of producing reliable bond strengths between the zirconia and the porcelain, chemical modification of the zirconia surface has also been recommended to encourage a strong bond (7). Although previous studies have shown that metal primers influence the strengths of dental ceramics' bonds to resin cements (6, 16, 19), available information regarding such primers' effects on the bond strength between zirconia and porcelain is limited. In the present study, the bond strength of the veneering ceramic to the zirconia core was significantly increased when the alloy primer was applied. The adhesive functional monomer of the alloy primer (VBATDT) was previously shown to promote a strong and durable bond to both noble alloys (40). The ability of these adhesive monomers to form chemical bonds with metal oxides (6, 16, 19), secondary forces (such as van der Walls forces) or hydrogen bonds at the interface warrants consideration. These interfacial forces improve the surface wettability of the zirconia ceramic and the ceramic bonding. A possible explanation of increased bond strength between the zirconia and the veneering ceramic is that the chemical characterization of the zirconia surface is similar to that of metal surfaces. MDP monomer bonds directly to metal oxides. There may be reactions between the hydroxyl groups in the MDP monomer and the hydroxyl groups on the zirconia ceramic surface. The 2 materials fuse together, and certain elements may diffuse across the interface (37). These occurrences may cause a chemical alteration in the glass layer adjacent to the core. An elemental analysis was performed for silica, calcium, sodium, aluminum and potassium, showing that residual porcelain was present on the core surfaces (Fig. 2). This may indicate that the veneering porcelain is chemically bonded to the metal primer.
In the current study, veneering ceramic pores and defects were identified on the SEM images of the sectioned areas (Fig. 1), which also might be the primary reason for bonding failures at the interface. Adhesive failure never occurs in the presence of a good bond between a compatible ceramic core and the veneer material (8, 25). In the present study, 85% of the failures were cohesive in group 4. SEM analysis also demonstrated that the fracture of the metal primer—treated zirconia specimens occurred within the veneering porcelain, leaving a thin layer of veneering porcelain on the zirconia surface. This finding indicated that the weak link in the metal primer—treated zirconia specimens (group 4) was in the veneering ceramic. Although failure type may not always correspond to bond strength values, the predominance of cohesive failures in group 4 may be associated with the fact that this group had the highest bond strength among the experimental groups. In contrast, groups 1, 2, 3 and 5 exhibited all failure types (i.e., adhesive, cohesive, and mixed). Thus, it could be suggested that the bond strength between the zirconia and the veneering ceramic in group 4 was higher than the bond strength in the other groups.
A silane coupling agent was subsequently applied to the roughened surface with hydrofluoric acid, which dissolved the glassy components of the silica-based ceramics and produced a porous, irregular surface that increased the bond strength of resin cement. Silane forms a siloxane network with SiO2-based ceramics, the chemical reaction of which is inapplicable to zirconia-based ceramics because it lacks a silica phase (31, 41). According to previous studies, silanes alone do not provide reliable chemical covalent bonding with zirconia, because of zirconia's inertness (31, 41). In the present study, the application of a silane coupling agent did not improve the strength of the ceramic's bond to the zirconia. The fact that poor wetting and silane were used alone without surface roughening for the chemical bonding to zirconia may explain this finding.
In the present study, the groups that underwent zirconia surface treatment (grinding or airborne particle abrasion) did not show any significant differences when compared with the groups that received ceramic primer application; however, metal primer application improved the shear bond strength. Zirconia-veneering ceramic bonding presumably was not influenced by surface treatments but by other factors, such as wetting properties, the chemical composition of the liner and the degree of compressive stress in the veneering layer (8, 10, 22).
Consequently, surface roughness created by sandblasting or grinding are not necessary to enhance the bond strength, as the present study shows. Chemical bonding seems to be a more reliable method if a suitable agent is used. Future studies should continue to focus on surface treatments that will enhance the bond strength of zirconia to the veneering porcelain. Such tests should consider various types of veneering porcelain and zirconia core materials and different techniques involving the application of the veneering porcelain to the core material, aging conditions and the effects of such factors.
Conclusions
Within the limitations of the present study, the following conclusions were drawn:
Chemical agents designed to improve the bond strength of the zirconia core to the veneering ceramic may have more benefits than mechanical pretreatments do.
The application of a ceramic primer to the zirconia core before ceramic firing does not have any advantages, nor do grinding and surface abrading.
The application of a metal primer to the zirconia core increased the bond strength of the veneering ceramic.
