Abstract
Purpose
Nerve gap injuries may be associated with lesions in other structures, like tendons or bones; in these cases, it is common to plan a second surgery to improve functional recovery. Since macroscopic observations of nerve regeneration in humans are rare, we exploited these second surgeries for the purpose of studying nerve regeneration in humans.
Methods
We assessed the clinical outcomes of 50 implants of collagen-based nerve guides in the upper limb. We performed a second look at 20, assessing macroscopically both nerve regeneration and collagen degradation.
Results and Conclusions
Pain was never recorded in these patients. An adequate sensory recovery took place whenever nerve regeneration was found inside the guide. Motor recovery seemed to occur only when the gap lesion was shorter than 10 mm. The degree of degradation appeared to be variable and was not directly correlated with time; we hypothesize that it could be associated with the site of implantation. Such a large number of second looks in humans has never been previously reported in the literature.
Introduction
It is more than 20 years since artificial nerve guides (or nerve conduits, or nerve cuffs) have been introduced into clinical practice, as an alternative to nerve autografts in short-gap lesions (1). A nerve autograft is a self-donation of another nerve of the patient, used for the repair of the nerve gap injury. It is important to outline that the patient loses the function of the donor nerve after the harvesting of the autograft. Therefore, donor nerves are selected among small sensitive nerves which innervate body parts which are reputed to be of less functional importance, such as the calf.
The nerve guides used in clinical practice are basically cylindrical conduits inside which a regenerating nerve stump may find protection and guidance (2). Early guides were made of silicone and were not biodegradable (3). They were considered responsible for the stenosis of the regenerate, and eventually removed (4, 5). Degradable guides were soon developed, using polyglycolic acid (6). Subsequently, other biodegradable polymers and treated bovine collagen (7) were exploited. An overview of the clinical literature shows that nerve guides in humans perform at least as well as autografts in gaps not greater than 20 mm, bringing the significant advantage of avoiding donor site sacrifice and morbidity (8, 9).
In clinical papers it is more common to encounter the term nerve conduit instead of nerve guide, since the devices used in humans are cylindrical. However, in biomaterials papers we can find already more complex geometries, so we prefer to apply the term nerve guide also in this clinical setting (10).
Clinical results on nerve guides in humans have been collected in the literature, following various clinical protocols (reviews of clinical trials can be found in (11-12-13)). The main difference between clinical studies and in vivo studies in animals (14), lies in the fact that in humans, it is not possible to retrieve the nerve regenerate for a microscopic analysis (15). Even if it is not possible to ethically justify in clinical studies the retrieval and analysis of a successfully regenerated nerve, there are particular instances in which we can have a macroscopic second look at nerve regeneration. In fact, the nerve gap injury may be associated with lesions in other structures, like tendons or bones. In these cases, it is common to plan a second surgery (e.g., in the surgical removal of fibrotic adhesions) to improve the functional recovery. We were often able to perform this second look surgery in our series, because our patients often presented complex multiple lesions. We did this in 20 out of 50 early implants. Such a large number of second look cases in humans has never been reported previously in the literature (11-12-13).
Materials and Methods
We implanted nerve guides in patients with gap injuries up to 30 mm. Site of lesions ranged from the elbow to the fingers. Most of the patients were treated in emergency, that means within 24 hours from trauma, and presented with associated lesions. We proposed a collagen implant (NeuroMatrix; Stryker, Mahwah, NJ, USA; or Neuragen; Integra, Plainsborough, NJ, USA) as an alternative option to autologous grafting or neurectomy. We informed the patients that a second operation could be undertaken when needed: e.g., a tenolysis (tendon debridement) in cases of multiple tendon lesions. During these successive surgeries, we had the chance to have a second look at nerve guides and to assess macroscopically their stage of degradation and their inner content.
In our early 50 implants, we treated 15 digital, 11 metacarpal, 10 median, 7 ulnar and 6 superficial radial nerves. We assessed the clinical recovery — namely, the restoration of protective sensation and of motor function — by clinical examination.
We performed 20 second looks: 6 in digital, 3 in metacarpal, 5 in median, 4 in ulnar and 2 in superficial radial implants. Of the remaining implants, in 22 we found no indication for a second surgery, while the remaining 8 were in patients who did not present with associated lesions.
To define the magnitude of collagen degradation, we applied a simple 3-grade degradation score. In grade 1, the guide was nearly completely degraded; no remnants or sparse remnants remained, but they never encircled the nerve regenerate; there was nothing to cut or peel off of what remained of the guide. In grade 2, the guide was degraded, but its remnants appeared to encircle part of the nerve regenerate; the consistency of the guide allowed it to be cut by microscissors but not peeled off with pinchers. In grade 3, the guide was mildly degraded; remnants encircled the nerve regenerate; the consistency of the guide allowed it to be cut and peeled off.
Mathematical correlations were analyzed using standard software (Microsoft Excel and Microsoft PowerPoint).
Results
Before reporting our results, we would like to point out that all patients who received the second look operation declared a further improvement in their clinical condition, and they were satisfied because of that.
Clinical Outcomes in the Complete Series
Implants in digital nerves showed a good sensory recovery and absence of painful neuroma. Guides which were implanted in proximity to a joint did not add any discomfort to the patient and at second look did not show any collapse. Implants in metacarpal nerves showed an absence of any painful neuroma; however, sensory recovery was obtained only in 4 out of 11 cases. In implants in median nerves, there was an absence of any painful neuroma, and there was sensory recovery in all cases; however, motor recovery was not obtained in 6 out 10 of cases. In implants in ulnar nerves, there was an absence of any painful neuroma, but sensory recovery was present in only 2 short-gap lesions and motor recovery in only 1, out of 7 cases. In all implants in superficial radial nerves, there was, notably, an absence of any painful neuroma; sensory recovery was present in 5 out of 6 cases.
Macroscopic examination of the nerve regenerate at second look
Nerve Regeneration
In digital implants, a nerve regenerate was found inside all guides, which were often mildly degraded (Fig. 1).
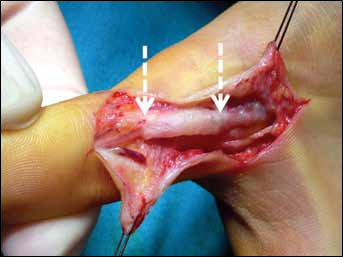
A mildly degraded 3-mm (diameter) nerve guide in a 19-year-old boy. This was used to bridge a 10-mm gap in a digital nerve of the thumb and was implanted 15 days after trauma. Second look after 3 months.
In metacarpal implants, we found a nerve regenerate in a case associated with sensory recovery but no regenerate in 2 cases without recovery. In median implants, we always found a nerve regenerate, and this was associated with the sensory recovery of the patient (Fig. 2).
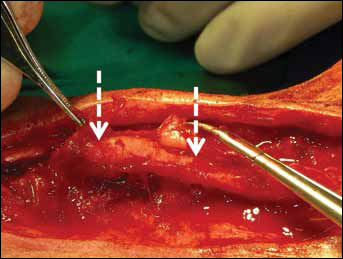
Median nerve regeneration inside a 6-mm (diameter) nerve guide in a 43-year-old man. This was used to bridge a 25-mm gap and was implanted 51 days after trauma. Second look after 8 months.
However, the diameter of the regenerate was variable, and in 2 cases, it was less than one third of that of the healthy nerve. In one of these cases, it was possible to associate an electrophysiological test which suggested that an alternative route was working (a Martin-Gruber anastomosis with the ulnar nerve). In ulnar implants, a nerve regenerate was found in those patients in whom a clinical recovery had occurred, while an empty guide was found in association with an absent recovery (Fig. 3).
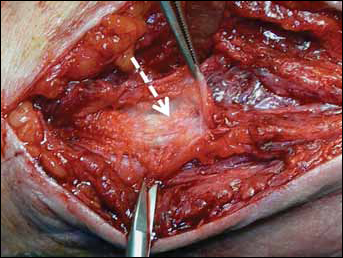
Fibrous streaks without any ulnar nerve regeneration inside a 6-mm (diameter) nerve guide in a 43-year-old man. This was used to bridge a 25-mm gap and was implanted 76 days after trauma. Second look after 8 months.
In superficial radial implants, we found a regenerate always associated with highly degraded guides (Fig. 4).
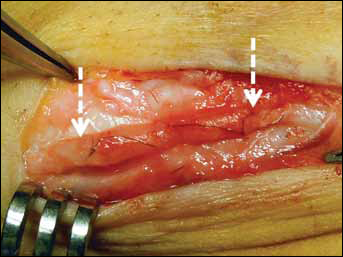
Superficial radial nerve regeneration inside a 2-mm (diameter) highly degraded nerve guide in a 33-year-old man. This was used to bridge a 15-mm gap and was implanted 13 days after trauma. Second look after 13 months.
Nerve-guide Degradation
In Figure 5, we plot the degradation score of the macroscopic assessment of nerve guide degradation versus months at which the second look was performed. Data are grouped according to the site of implantation.
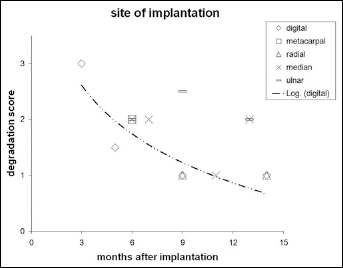
Degradation score versus months at second look: data are grouped according to the site of implantation. A logarithmic regression can be traced for digital implants.
In Figure 6, data are grouped according to the inner diameter (lumen) of the guide.
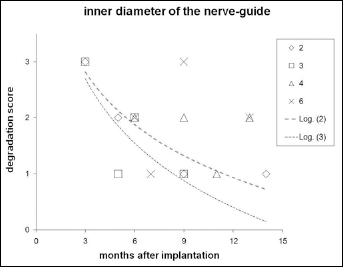
Degradation score versus months at second look: data are grouped according to the inner diameter of the guide (in millimeters, see key). A logarithmic regression can be traced for 2-mm (diamonds) and 3-mm (squares) diameter nerve guides.
Discussion
Macroscopic observations of nerve regeneration in humans are rare — in contrast to the wealth of data collected from animal experiments (16-17-18). Despite the cases that have been episodically described in conferences and in case reports (3, 19), we found no evidence in the literature for a systematic approach at second looks after nerve guide implantation (11-12-13). We pursued such a systematic approach in having a surgical second look whenever possible, so we were able to gather a number of patients that has never been reported previously in the literature. Our data are in accord with what is known from animal studies, but in our opinion, they are more relevant because they have been collected in humans.
Most of the cases dealt with purely sensitive nerves. We found that the recovery of protective sensation followed a territorial pattern, stemming from the site of the lesion. This suggests that true regeneration stems from the proximal stump, instead of an overlapping innervation from a side branch. A nerve regenerate was found in association with clinical recovery (a “true positive” outcome). However, we judge as relevant also the fact that the absence of regenerate was associated with no clinical recovery (a “true negative” outcome).
Before discussing data on degradation, we think that it is important to underline that the clinical meaning of the degradation of an artifact is not purely measured in time (namely, the time needed for the in vivo degradation process to occur). It should instead be correlated with the duration of the healing process in which the artifact is employed (20). So, if we use degradable devices from material that has a nominal degradation time (which, in the case of our collagen nerve guides is variably disclaimed between 8 and 36 months (13)) and the process of nerve regeneration needs a shorter time to be completed, then the device will perform clinically as a permanent device. This also means that nerve regeneration occurred, and was clinically effective, inside nerve guides whose morphological integrity was preserved.
Plotting our data for degradation versus time, we obtained poor correlations. This was due to the limited number of data for each subgroup and to the several variables involved. However, we looked for a possible trend, and we found that data from implants with smaller lumen (inner diameter) provided a coherent plot when a logarithmic correlation was applied. In data grouped according to the site of injury, we found a coherent trend only in digital implants. These 2 plots were interrelated, since small diameter guides are mostly used in digital nerves.
It can also be pointed out that patients who received the second-look operation declared a further improvement in their clinical condition. We found that this result was notable but could not be used to support the conclusion that a second look may be a necessary step in treating patients with nerve guides. As a matter of fact, in our series, the majority of patients were not selected to be included in the group which received second-look surgery, because there was no need for it. More clinical cases will be necessary in future to clarify this point further.
Conclusions
We summarize our results as follows: (i) pain was never recorded in any of the patients in this series; (ii) an adequate sensory recovery took place whenever a nerve regenerate was found inside the guide; (iii) motor recovery seemed to occur only when the gap lesion was shorter than 10 mm; (iv) the degree of degradation appeared to be variable and not directly correlated with time; we hypothesize that it could be associated with the site of implantation.
