Abstract
Purpose
In this study, osteoblast-like MG-63 cells were cultured on 3 different scaffold types composed of (a) collagen + poly-L-lactic acid (PLLA), (b) collagen + hydroxyapatite (HA; 30°C) or (c) collagen + hydroxyapatite (HA; 37°C) and produced with different porosities.
Methods
Biomechanical properties of the scaffolds were characterized by tensile strength measurements. Properties of the cell-seeded scaffolds were evaluated with scanning electron microscopy (SEM). Cell adhesion and proliferation capacities were evaluated. Alkaline phosphatase (ALP) levels in media were measured. Transmission electron microscopy (TEM) and histological analyses were used to assess morphological characteristics.
Results
Our results showed that collagen-based PLLA and HA scaffolds have good cell biocompatibility. MTT test showed that the scaffolds exhibited no cytotoxicity. According to the force and displacement data, collagen + HA at 37°C showed the highest mechanical strength and displacement.
Conclusion
The results suggest that collagen-based PLLA and HA scaffolds might improve osteoblastic growth in vitro and have biomaterial integration potential in possible therapeutic approaches for future clinical studies.
Introduction
The cell interaction on biomaterials is a highly dynamic process, and it depends on various parameters influencing the cell responses. In adherence-dependent cells, not only the size and shape of cell spreading area but the size, shape and distribution of focal adhesion plaques are decisive for further migratory, proliferative and differentiation behavior of the cells (1, 2).
Scaffolds for bone regeneration have been composed of various materials such as glass beads, ceramics, collagens, tricalcium phosphate, hydroxyapatite and a variety of synthetic polymers (3).
Panzavolta et al investigated the influence of electrospun polymer fibers on the properties of an α-tricalcium phosphate/gelatin biomimetic cement. According to their results, the crystallinity of the apatitic phase was reduced in compact microstructure (4).
Poly-L-lactic acid (PLLA) is one of the well-defined substrates which is in use for osteoblast adhesion. Generally, most of the PLLA substrates for osteoblast cultures are nontoxic, biodegradable materials, widely used as scaffold material in tissue engineering. Type I collagen, the major organic component of bone extracellular matrix, on the other hand, plays an active role in osteoblast phenotype expression and encourages osteogenic differentiation and mineralization in bone (5, 6).
Hydroxyapatite (HA) is the main inorganic component of bone and is recognized as a good bone-filling material, because of its osteoconductivity. However, due to their differences in mechanical and biochemical properties from those of natural bone, HA-based materials, when transplanted, remain in the bone for the patients' lifetime and induce fractures at or around the site of transplantation. Because of its biocompatibility, bioactivity, rapid attachment of osteoblasts and not having any requirement for follow-up surgery, HA is considered a superior choice for drug carrier system and bioactive implant design compared with polymers (7-8-9).
To achieve effective bone remodeling and fracture healing, scaffolds for bone tissue engineering are required to replicate the functionality of the natural bone extracellular matrix, to promote cell attachment and tissue growth, interact with host tissue, and thus have an effective biodegradable, microporous and nanostructure of the bone (10-11-12-13-14).
The objective of this study was to investigate proliferation capacities and osteogenic responses of MG-63 human osteoblast-like cells on 3D collagen-based PLLA and HA scaffolds with different porosities.
Materials and Methods
Scaffold Preparation and Characterization
In this study, 3 different types of scaffolds were assessed: (a) collagen + PLLA, (b) collagen + HA at 30°C and (c) collagen + HA at 37°C.
Collagen and PLLA
PLLA solution (4%) was prepared using PLLA (Purasorb Poly-L-Lactide, molecular weight 800.000 Da. Purac Biochem, Gorinchem, Holland), dissolved in chloroform. Gelfix, a 3D scaffold collagen material (Gelfix® collagen; Euroresearch s.r.l., Italy) was soaked in preprepared PLLA solution, and collagen fibrils were allowed to moisten. Excess solution was then removed, and collagen with PLLA was soaked in absolute alcohol (ethanol). Alcohol was changed regularly to allow PLLA to deposit on collagen fibrils, which was then dried at 50°C in a vacuum.
Preparation of Artificial Body Fluid (ABF)
Artificial body fluid was prepared as in Tab. I. The list of chemicals used in preparation of ABF was as follows: (i) CaCl2.2H2O, (ii) MgCl2.6H2O, (iii) KCl, (iv) NaCl, (v) Na2HPO4.2H2O, (vi) Na2SO4, (vii) NaHCO3, (viii) lactate and (ix) lactic acid. In ABF × 2.5; Ca+2 and PO4−3 concentrations were 2.5 times higher compared with the concentrations in Tab. I. To prepare ABF, the chemicals numbered 3-8 were added in a container with 900 mL of distilled H2O, and then the pH was adjusted to 8.05 while the solution was stirred on a heated magnetic mixer. Seven milliliters of lactic acid (1 M) was quickly added, the temperature was increased to 37°C quickly and pH was adjusted to 6.95. Salts with numbers 1 and 2 were dissolved in 10 mL of distilled H2O and added to the first solution quickly, and pH was adjusted to 7.37. The final volume was completed to 1 L.
COMPARATIVE ION CONCENTRATIONS OF Tris-ABF AND Lac-ABF USED IN THIS STUDY
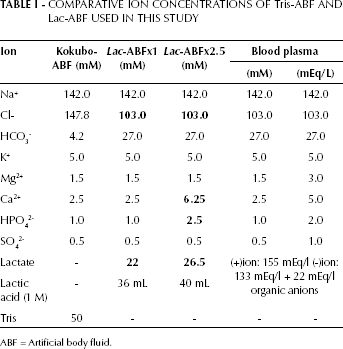
ABF = Artificial body fluid.
In the literature, an incubation period of 1-2 months has been suggested for the preparation of collagen + HA using artificial body fluid (ABF). When ABF is used as 1 × blood plasma product, this duration is decreased to 3 weeks. In our study, by using ABF × 2.5 and by increasing the HPO and Ca concentrations 2.5 times, we achieved 6.5 mM and 2.5 mM final concentrations for Ca and HPO, respectively, which was enough to have HA deposition in 24 hours. This is one of the novel findings of our study (Tab. I).
Preparation of Collagen and HA at 30°C, using ABF
Gelfix collagen scaffold (5×5×1 cm) was soaked in the prepared ABF solution at 30°C in a closed container. After 24 hours, the pH was measured as 7.04. No change was observed in the size of the collagen scaffold after 24 hours at this temperature. The scaffold was then washed with distilled water and dried at room temperature.
Preparation of Collagen and HA at 37°C, using ABF
Gelfix collagen scaffold (5×5×1 cm) was soaked in ABF solution and kept at 37°C for 24 hours. This procedure caused a decrease in size of the scaffold to 2.5×2.5×0.5 cm. The scaffold was then washed with distilled H2O and dried at 40°C.
All scaffolds were sterilized at 90°C with ethylene oxide prior to use in tissue cultures.
Cell Culture
MG-63 Osteosarcoma Human Cell Line, obtained from American Type Culture Collection (ATCC; Manassas, VA, USA) (ATCC-CRL 1427 Lot number: 57840088) was used. The cells were seeded in a culture flask (25 cm2) with MEM-Eagle (MEM, 03-025-1A; Biological Industries) medium which contained 10% fetal calf serum. L-glutamine (200 mM, G7513; Sigma) and Pen-Strep-Ampho (03-033-1B; Biological Industries) and was cultured at 37°C in a humidified 5% CO2 atmosphere. The medium was changed on alternate days. After that, the cells (5×105 cells/μL) were seeded on scaffolds (size: 8×6×3 mm; L×W×H; n = 3).
Scanning Electron Microscopy
Scaffolds were analyzed by scanning electron microscopy (SEM) prior to cell seeding to evaluate their porosity sizes. When a confluent monolayer was achieved, cells were trypsinized and centrifuged, and supernatant was discarded. After 21-day cultures on scaffolds, the scaffolds were washed with phosphate-buffered saline (PBS). Subsequently, the cells were fixed with 5% glutaraldehyde (pH 7.2), 7% sucrose and 2% osmium tetroxide in sodium cacodylate buffer (0.1 M). The specimens were dehydrated using graded ethanol changes and gold splattered in vacuum (Polaran SC7620) at 10 kV, and examined using SEM (JEOL JSM—6060).
Transmission Electron Microscopy
The scaffolds were fixed on day 21 with Karnovsky solution (2.5% buffered glutaraldehyde + 2% paraformaldehyde in 0.1 M sodium phosphate buffer [Sorensen buffer]). The following day, scaffolds were postfixed with 0.1-M sodium cacodylate buffer (pH 7.4). After that, the scaffolds were dehydrated in acetone serial. The samples were then embedded in Epon solution. Sections were cut with an ultramicrotome, set to 50- to 100-nm section thickness and examined with an EFTEM (Carl Zeiss Libra 120, Germany) at 60 kV.
Biomechanical Properties of Scaffolds
The biomechanical tests were carried out by using a tensile-testing machine (AG-I 10 kN; Shimadzu, Japan). The tensile test was applied to all 3 types of scaffolds (n = 6) with a loading speed of 1 mm/min. The dimensions of scaffolds were 24×4×6 mm. Statistical analyses were completed.
Alkaline Phosphatase Activity Assay
To investigate the osteoblastic activity, the osteoblastic marker, alkaline phosphatase (ALP) activity was measured according to manufacturer's recommendations for 7 days in culture medium using a commercially available kit (Enzyline PAL Optimise; Biomerieux, France) by spectrophotometer (Cary 50 UV-Vis).
MTT Assay
Cellular growth of MG-63 cells on collagen-based PLLA and HA scaffolds was verified by 3-(4,5-dimethylthiazol-2yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay using a commercially available kit according to manufacturer's recommendations in 4-day cultures.
Histological Analysis
The cultures were cultivated for 21 days on scaffolds for histological observations (n = 3). Scaffolds were stained with hematoxylin and eosin (H&E), and semi-thin sections of 1.5 μm in thickness were cut and stained with 1% toluidine blue for transmission electron microscopy (TEM) fixation.
Statistical Analysis
The data of biomechanical tests of force data and MTT assay were analyzed with a nonparametric Kruskal-Wallis test. The statistical analysis was carried out using “SPSS”, version 15 (SPSS Inc., Chicago, IL, USA). A p value of <0.05 was considered statistically significant.
Results
SEM Observations and Cell Morphology
MG-63 cells showed comparable adhesion characteristics on dense and porous scaffolds by SEM analysis. Porosity range of the scaffolds varied between 50 and 200 μm.
Figure 1 shows the morphology of osteoblast cells attached on the various surfaces of collagen-based PLLA scaffolds. Fig. 1a-b-c are the images of scaffolds with no seeded cells, and Fig. 1d-e-f are the respective materials with cells seeded on. Fig. 1b shows the SEM photographs of the collagen-based HA at 30°C scaffold prior to cell seeding. High amounts of calcium precipitates were observed on some scaffolds prior to the cultures (Fig. 1c). Cells proliferated on scaffolds making confluent layers, while fusiform-shaped cells were observed to attach to the pore walls of the scaffold with extracellular matrix after 21 days (Fig. 1a, c).

Scanning electron microscopy (SEM) morphology of collagen-based scaffolds without and with MG-63 cells, respectively, collagen + PLLA (a, d), collagen + HA at 30°C (b, e), collagen + HA at 37°C (c, f) at day 21 of culture (scale bar = 10 μm; ×1,000 magnification).
Transmission Electron Microscopy
TEM investigations were carried out on cells in 3D scaffolds on day 21 (Fig. 2). Multilayer cells with large nuclei could be seen in scaffolds along with collagen fibers, visible in the scaffold structure.

MG-63 cells seeded on scaffolds: collagen + PLLA (a-b), collagen + HA at 30°C° (c-d) and collagen + HA at 37°C (e-f) for 21 days were assessed with transmission electron microscopy (TEM). N = nucleus; D = dark cell; C = clear cell. Scale bar = 5 μm (a-b), scale bar = 2 μm (c), scale bar = 5 μm (d), scale bar = 5 μm (e-f).
Biomechanical Properties of Scaffolds
Results of the tensile strength of force data are shown in Fig. 3. Collagen + HA at 37°C showed the highest mechanical strength, whereas collagen + PLLA showed the minimum. The differences between the mean mechanical strength values of the study groups were statistically significant (collagen + PLLA vs. collagen + HA at 37°C, P = 0.005; collagen + HA at 30°C vs. collagen + HA at 37°C, P = 0.025). In terms of displacement data, collagen + HA at 37°C scaffolds had the highest, whereas the collagen + HA at 30°C scaffolds had the lowest, mean values. Statistically, mean values of all 3 groups were significantly different from each other (collagen + PLLA vs. collagen + HA at 30°C, P = 0.004; collagen + PLLA vs. collagen + HA at 37°C, P = 0.008; collagen + HA at 30°C vs. collagen + HA at 37°C, P = 0.016).
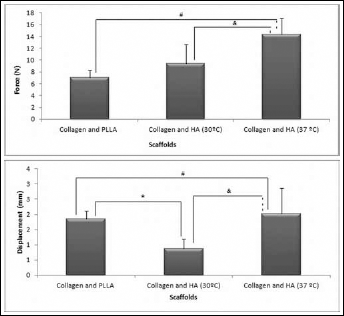
Tensile strength of force and displacement data were significantly different between groups: #P=0.005, collagen + PLLA vs. collagen + HA at 37°C; &P = 0.025, collagen + HA at 30°C vs. collagen + HA at 37°C. Statistically, mean values of displacement data in all 3 groups were significantly different from each other (*P = 0.004, collagen + PLLA vs. collagen + HA at 30°C; #P=0.008, collagen + PLLA vs. collagen + HA at 37°C; &P=0.016, collagen + HA at 30°C vs. collagen + HA at 37°C).
ALP Activity
Osteoblastic activity of MG-63 cells seeded on scaffolds and groups were determined by measuring ALP activity in culture media for 7 days. ALP activity of osteoblasts increased in all groups in a time-dependent manner (Fig. 4).
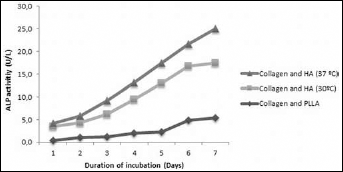
Alkaline phosphatase (ALP) activity of cells seeded on scaffolds after 7 days in culture on 3 types of scaffolds. ALP activity was expressed as U/L.
MTT Test
The biocompatibility of scaffold types on MG-63 cells and the proliferation state of the cells were tested by MTT assay. MG-63 cells continued to proliferate on all 3 types of scaffolds, suggesting no toxic effects of scaffolds on cells. Although MTT levels of cells on the collagen + HA at 37°C scaffold were slightly lower compared with the other groups at days 2 and 3, this difference between scaffold groups disappeared with a steady increase in collagen + HA at 37°C group in the following days (Fig. 5). The comparison of the groups among each other were statistically significant: collagen + PLLA vs. collagen + HA at 30°C; collagen + PLLA vs. collagen + HA at 37°C; and collagen + HA at 30°C vs. collagen + HA at 37°C (P<0.05). The comparison of the MTT levels of cells on each day were shown to be significantly different P<0.05 except collagen + HA at 37°C (comparison between 1 and 2 days).
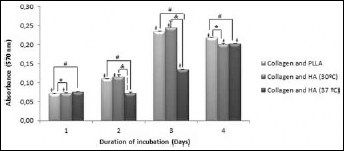
Viability of MG-63 cells seeded on scaffolds measured by MTT assay for 4 consecutive days. Means ± SD (error bars) for n=3. Note: comparing the groups among each other: *P<0.05, collagen + PLLA vs. collagen + HA at 30°C; #P<0.05, collagen + PLLA vs. collagen + HA at 37°C; & P<0.05, collagen + HA at 30°C vs. collagen + HA at 37°C. The comparison of the MTT levels of cells on each day showed significant differences (+P<0.05) except collagen + HA at 37°C (comparison between 1 and 2 days).
Histological Results
Semi-thin (1.5 μm in thickness) histological sections of cell-seeded scaffolds were prepared and stained with toluidine blue fixation at the end of 21 days of culture period (Fig. 6). Thin layers of MG-63 cells were present on the scaffold's outer surface. In the collagen + PLLA group, more cells were present at the outer surface of scaffold compared with the collagen + HA at 30°C and collagen + HA at 37°C groups.
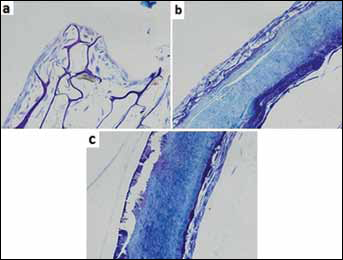
MG-63 cells seeded on collagen + PLLA (a), collagen + HA at 30°C (b), collagen + HA at 37°C (c); scaffolds were stained with toluidine blue at day 21 (×20 magnification). MG-63 cells mostly seen at outer surface of the scaffolds.
Discussion
At present, cell therapy and tissue engineering are a promising alternative to artificial permanent implants for repair of tissue lesions. As a newly developing discipline in the biological and medical sciences, bone tissue engineering achieves successful results in growth of human bone tissue. In this study, the behavior and osteogenic activity of MG-63 cells seeded on different collagen-based PLLA and HA scaffolds were investigated. We observed that MG-63 cells securely attached to the material surfaces and maintained good attachment. Li et al suggested that HA/collagen could improve the interactions between osteoblast cells and the polymeric scaffold (15). In agreement with Li et al, levels of ALP activity of MG-63 cells and SEM analysis suggested that collagen-based PLLA and HA scaffolds have good interactions with osteoblasts and thus are very good candidates for bone regeneration models in vivo.
The microstructure of scaffolds and surface morphology is another important factor for adhesion, and tissues grew into the pores of osteoblast cells, thus demonstrating the tissue biocompatibility of the scaffold. In our study, porosity range of the scaffolds varied between 50-200 μm. SEM analysis of cells seeded on scaffolds showed that cells were mostly attached to pores and walls of collagen + PLLA scaffolds at day 21, where they formed extracellular matrix around them (Fig. 1). The cells also demonstrated good attachment to the surface of both collagen + HA at 30°C and HA at 37°C scaffolds. The cells proliferated more on collagen + HA at 37°C than on the collagen + HA at 30°C scaffolds. Furthermore, better calcium phosphate deposition was observed on collagen + HA at 37°C scaffolds throughout the culture period. We believe that this effect was the cause of higher Ca ratio of collagen + HA at 37°C scaffolds compared with HA at 30°C.
Analysis of TEM images is an important factor for understanding the morphology of multilayer osteoblast cells around scaffold. In our study, cells had large nuclei showing with granule and cell contacts on all 3 types of scaffolds (Fig. 2). Furthermore, they tended to attach to all 3 types of scaffolds, mostly around the collagen fibrils. Cytoplasmic vesicles, fragments, large cell nuclei and scaffold material were observed. The results of TEM and SEM data indicated that both collagen-based PLLA and HA scaffolds had good biocompatibility, biodegradation and osteointegration in in vitro cell cultures. According to our results, all 3 bioresorbable scaffolds showed sufficient space for cell adhesion, distribution and ingrowths into deeper layers of its microstructure. Similar results were reported by Prabhakaran et al showing biocomposite PLLA/collagen/HA scaffolds to be potential substrates for the proliferation and mineralization of osteoblasts, enhancing bone regeneration (16).
Collagen + HA at 37°C showed the highest mechanical strength, whereas collagen + PLLA showed the minimum, in biomechanical testing. An interesting observation was that collagen + PLLA with the minimum mechanical strength was more breakable than the other scaffolds. We think this finding may be due to the higher porosity ratio of collagen + PLLA scaffold, as it is known that increased porosity causes a decrease in mechanical strength. However, this characteristic makes collagen + PLLA type scaffolds better candidates for bone tissue engineering, as cells tend to attach more to scaffolds with increased porosities.
In our study, the highest ALP levels were observed in cells on collagen + HA at 37°C scaffolds at the end of the 7-day culture period. ALP activity of MG-63 cells seeded on collagen + HA at 30°C scaffolds was also higher compared with that on cells on collagen + PLLA scaffolds, by the end of 7 day cultures, suggesting collagen + HA scaffolds to be suitable for osteoblast seeding and growth (Fig. 4).
Based on the MTT assay, collagen-based scaffolds are nontoxic and biocompatible (Fig. 5) The MG-63 cells continued to proliferate on all 3 types of scaffolds, while being higher on collagen + PLLA ones. In summary, the scaffolds are nontoxic and suitable for the proliferation of MG-63 cells. The histological analyses also confirmed that MG-63 cells were well attached to and proliferated on the scaffolds. The histological observations indicated that cells preferred to attach on the dense as well as porous membranes. Collagen-based PLLA and HA scaffolds were found to have dense morphologies (Fig. 6). In our study, porosity ranges of the scaffolds varied between 50 and 200 μm. We think that the microarchitecture of the pore structure of the scaffolds plays a crucial role in defining cell infiltration efficiency. Despite the porous structures of collagen + HA scaffolds, longer culture periods might have caused shrinkage in scaffolds, decreasing the porosity and causing fewer cells to infiltrate into scaffolds' inner sections.
Collagen-based PLLA and HA scaffolds have a high potential for applications in the field of bone tissue engineering. In the present study, our results suggest that using collagen-based scaffolds which promote osteoblast phenotype, it may be possible to develop techniques for bone regeneration and biological prostheses and diminish the risk of complications caused by currently available bone prostheses. Our results also suggest that scaffold surface morphology might be an important factor for adhesion and growth of osteoblast cells.
In summary, our results indicate that collagen + PLLA scaffold showed a higher porosity ratio for adherence and proliferation, but better calcium phosphate deposition was observed on collagen + HA at 37°C scaffold. Our findings also propose that the collagen-based PLLA and HA scaffolds, since they have a good affinity to cells, may be suitable for the regeneration or replacement of bone defects. However, further studies are needed to examine how collagen-based PLLA and HA scaffolds can enhance the bone regeneration efficacy of osteogenic cell transplantation in vivo.
