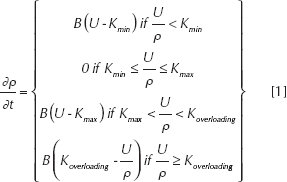Abstract
Purpose
The aim of the study was to assess some possible factors influencing the bone stresses and remodeling process after the placement of an implant in a postextraction socket.
Methods
A finite element model of a portion of the maxilla was used. Three osseointegrated titanium dental implants with different thread profiles were placed into the extraction socket. Three different lengths, thus with different depth into the bone, were considered. Bone remodeling was simulated by using the strain energy density as the remodeling stimulus. A load with magnitude 200 N and inclination of 45° to the longitudinal axis in the lateral direction was applied directly to the implant.
Results
Both implant length and thread profile had an influence on the bone stresses, growth and loss after implantation. Longer implants reduced both the size of the bone loss area and the total bone mass loss.
Conclusions
It is not easy to establish the relationship of the long-term success of implantation and bone turnover activity via clinical trials. After a proper clinical validation, the protocol developed using computational modeling may become a viable option to predict the clinical outcome, even on a patient-specific basis, regarding its noninvasive and time-efficient nature.
Introduction
Dental reconstruction is often employed for the replacement or filling of missing dental structures resulting from caries or other pathologies and traumas (1-4). Two approaches are commonly employed for dental reconstruction. In direct approaches, a soft material, usually a polymer (1), is inserted into the empty space to be filled. The material then becomes hard and acquires the definitive shape. In indirect restorations (5), ceramic or gold structures built based on dental impressions and replacing the missing parts are attached to the native anatomical structures with dental cement.
Despite the wide use and success of such techniques, in some cases teeth need to be removed completely and are replaced with a dental implant following extraction (6). These implants are inserted into the jawbones to support a prosthesis and are retained because of the intimacy of bone growth onto their surface (7). Several clinical papers, but only a few randomized controlled trials, suggest that placing implants immediately after tooth extraction may provide some advantages, such as limiting postextraction ridge alterations and reducing surgical procedures and treatment time (8, 9). Indeed, this point is debated, since a series of recent clinical trials and animal experiments demonstrated that ridge alterations following tooth loss occurred in a similar manner in implanted and in nonimplanted socket sites (10-12). A meta-analysis of prospective trials (13) concluded that implant-supported single-tooth replacement in the aesthetic zone with adjacent natural teeth can lead to short-term, favorable outcomes with regard to implant survival and marginal bone level change with early/immediate or conventional surgical placement and loading strategies.
The higher rate of implant failure associated with implant therapy in the maxilla has been related to the biomechanical complications (such as overloading) that lead to excessive marginal bone loss (14-16). The stress/strain distribution on the surrounding bone may be influenced by various parameters such as the implant size, shape, position and angulation, implant-abutment connection and the magnitude and direction of the occlusal load (17-19). Although it is not well established how mechanical loading affects the osteoblastic and osteoclastic activities, variables like the stress, strain or strain energy density have been proposed as effective mechanical stimuli for bone remodeling (20, 21). The effect of these variables can be evaluated using specifically developed mathematical algorithms and can be combined with finite element (FE) analysis to put forward quantitative numerical models for the prediction of bone remodeling (20, 22-25). Phenomenologically, there are certain similarities of remodeling mechanism and algorithms between long bones and jaw bones, and it is realistic to simulate jaw bone remodeling by using the procedure established in long bones (19). Taking into consideration the fact that the results from FE analyses, especially for bone remodeling, should be carefully interpreted (26, 27), they could be useful indicators for the development of human in vivo tests.
The aim of this computational study was to assess some possible factors influencing the bone remodeling process, the interfacial stress and the survival of an immediate loaded implant in a postextraction socket – namely, the depth of the implantation and the thread profile.
Materials and Methods
A simplified FE model of a portion of the maxilla, with average thickness 5.2 mm, was created from computed tomography (CT) images of an adult male directly following a dental extraction. A point cloud was generated with the 3-dimensional reconstruction software Amira 4.1 (Visage Imaging GmbH, Berlin, Germany) and imported in FE software Ansys 12.0 (Ansys Inc., Canonsburg, PA, USA), which was used to create and mesh the solid models (Fig. 1). The CT images showed a cortical layer around the extraction socket having an average thickness of 1.2 mm, which was considered in the present model.
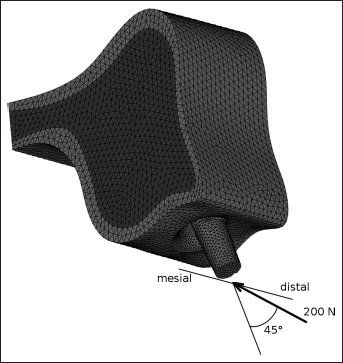
Finite element model of a portion of the maxilla including an implant in the postextraction socket. The direction of the load applied in the simulations is shown.
Three osseointegrated titanium dental implants, differing in thread profile, were subsequently placed into the extraction socket (Fig. 2). For 1 of the 3 shapes (model 1), implants with 3 different lengths, thus with different depth into the bone, were considered (Fig. 3). Implant and abutment were modeled as a single component; the internal fixation screw was neglected.
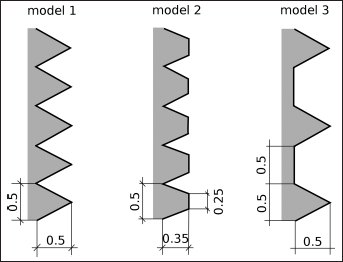
Profile of the thread for models 1, 2 and 3.
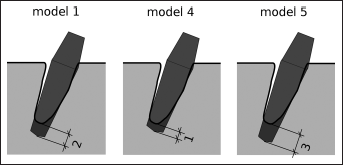
Schematic view of the models with implants of different lengths (models 1, 4 and 5).
Quadratic tetrahedral elements were used to mesh all components. In all models, a load with magnitude 200 N and inclination of 45° to the longitudinal axis in the lateral direction was applied directly to the abutment (Fig. 1). The exterior distal and mesial surfaces were fixed in all simulations. A surface-based contact with a friction coefficient of 0.5 (19) was employed to simulate the interaction between implant and bone in the extraction socket.
Bone remodeling was simulated by using the strain energy density (SED) as the remodeling stimulus (20), by employing a program written in the scripting language provided by Ansys, APDL. The bone apparent density ρ was assumed to locally vary following the equations (19):
where U is the element-based SED, Kmin Kmax and Koverloading are the 3 threshold values for bone resorption, growth and overloading respectively, and B is a coefficient determining the remodeling velocity. Bone loss is achieved for both low
MATERIAL PROPERTIES AND THRESHOLD VALUES OF REMODELING ALGORITHM (14)

as described by Mellal et al (28). One hundred iterations were performed for all models. For each time step, the total bone loss and growth in terms of mass were computed.
Prior to the simulations including bone remodeling, a mesh sensitivity analysis was carried out for all models considering the initial material properties, in order to exclude any dependency of the results on the employed mesh.
Results
The stresses acting in the bone surrounding the implant after the first iteration, thus with the initial material properties of the bone, are shown in Figures 4 and 5. Higher stresses with respect to values reported in a previous study were found (28), both tensile and compressive. The extension of the area subjected to high stress was comparable and not clearly dependent on the thread profile or the length of the implant.
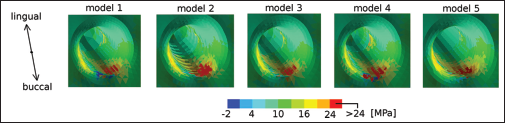
First principal stress around the implantation socket for the different models.
Third principal stress around the implantation socket for the different models.
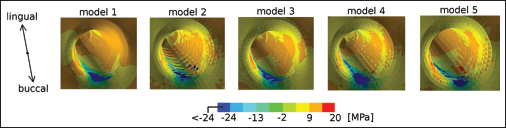
The calculated bone density distributions after 100 iterations showed a slight dependency on the different implant configurations (Fig. 6). Bone growth was found for all models on the buccal side of the implantation socket, with a comparable extension of the remodeling area. A small growth area was calculated for 2 models on the lingual side, in correspondence with the cortical shell surrounding the implant.

Section (first row) and 3-dimensional (second row) views of the local bone density predicted for the 5 models after 100 iterations.
Localized marginal bone loss was computed on the buccal side for 4 models. This loss was particularly pronounced for model 1 and model 4, which were the implants with the standard thread profile and with the standard and the shorter lengths. The implant with the greater length (model 5) induced a markedly smaller resorption area. The implantation of the models with the different thread profiles resulted in a small (model 2) and totally absent (model 3) bone loss area. Bone resorption due to overloading was also observed near the tip of the implant, for all configurations.
A quantitative analysis of the total bone loss and growth is shown in Figure 7. In general, the curves show that 100 iterations were not sufficient to obtain a steady state solution, in which all elements are in the zone for which no density alteration occurs. The shape of the thread (models 1, 2 and 3) determined a critical difference in the bone remodeling process. Model 2, exhibiting a thread without a sharp tip, induced a total bone loss 2 orders of magnitude lower than the other models, but the predicted bone growth was in the same range as the others. Model 3, which has a flat portion in the thread, determined a significantly lower bone loss with respect to model 1 (-65%); again, the calculated bone growth showed a weaker dependency on the shape of the thread (-33% with respect to model 1).
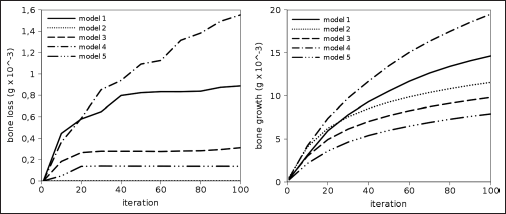
Total bone loss (left) and growth (right) versus iteration number predicted for the 5 models.
The length of the implant and the corresponding implantation depth also played a role in governing the bone remodeling process. The implant with the shortest length (model 4) induced markedly higher bone loss (+74%) and growth (+33%) with respect to model 1. The opposite behavior was observed for the implant with greatest length (model 5; −84% bone loss and −46% growth with respect to model 1).
Discussion
The simulation of the implantation of different osseointegrated dental implants in an extraction socket in the maxilla was presented. This clinical condition is generally characterized by a layer of cortical bone surrounding the socket, which may induce a different postimplantation behavior of the bone close to the implant with respect to the implantation in the edentulous maxilla. This difference may be augmented by the fact that the implant is only partially engaged in the bone, due to the generally bigger size of the socket if compared with the implant. Thus, higher stresses and remodeling stimuli may be expected with respect to the implantation in solid bone. The results generally confirmed these qualitative predictions. As a matter of fact, both tensile and compressive peak stresses were markedly higher than those in the literature data (28). This result may confirm that an immediate implantation in the fresh extraction socket should be able to reduce postoperative bone loss (29). However, in the previous study (28) a lower load (130 N) was applied, thus limiting the relevance of this finding.
Generally speaking, both implant length and thread profile had an influence on the bone growth and loss behavior after implantation. Longer implants reduced both the size of the bone loss area and the total bone mass loss. Shorter implants, providing a higher stimulus to bone remodeling, increased both the bone growth and loss, in the respective areas. The thread profile also had a major influence on the results. The smoother profile, which induced stress values under the overload threshold, drastically limited the bone resorption. However, since none of the thread shapes considered refer to any specific commercially available products, which generally feature rounding radii to some extent, a clinical interpretation for these results cannot be drawn directly.
Some limitations can be identified for the present models. The use of SED as the remodeling stimulus was found to be coherent with in vivo measurements (28), and has been widely adopted to simulate bone remodeling around dental implants (19, 28, 30). However, a comprehensive validation of this approach, including the bone resorption due to overloading, have never been presented in the literature. The time step size was chosen to avoid instabilities in the solution, which may be present due to the explicit time integration in the remodeling algorithm. However, the number of iterations was arbitrarily chosen, to keep the computation time manageable. The results showed that the total simulated time was not sufficient to reach a steady state solution, in which all elements are in the lazy zone. However, this limitation does not prevent the evaluation of the results in a comparative way, which was the focus of the present paper.
A single load type was used in all of the computations. An axial load combined with a lateral force component was found to induce a higher axial SED if compared with a pure axial load (19), thus enhancing the marginal bone loss and representing well a worst-case scenario. However, considering more load types may allow for the prediction of the bone remodeling in a more accurate way. Other limitations may concern the absence of a fixation screw between the implant and the abutment and the consideration of an immediate loading only, thus neglecting a partially or completely osseointegrated condition, and the general simplification of the geometry of the implants with respect to commercial products.
It is not easy to establish the relationship of the long-term success of implantation and bone turnover activity (31) via clinical trials, because such a time-consuming exercise would require that patients to receive considerable radiation dosages from regular CT or cone beam CT examination over the healing period (32). Therefore, after a proper clinical validation, the protocol developed using computational modeling may become a viable option, even on a patient-specific basis, for its noninvasive and time-efficient nature. However, results from FE analyses should be carefully interpreted, and an experimental and clinical validation should be performed whenever possible.
Footnotes
Financial support: None.
Conflict of interest: None.