Abstract
Background
Hernia repair often involves fascial augmentation using biologic prostheses. Small processing changes during preparation modulate host tissue response, which influence material efficacy and longevity. In this pilot study, a rat model was used to determine the specific influence of tissue origin, decellularisation treatment and 1,6-hexamethylene diisocyanate (HMDI) cross-linking.
Methods
Materials (1 cm2) were implanted subcutaneously into 6-week-old Wistar rats (4 materials per animal, n=6/material per time point) for 2, 5, 7, 14 and 28 days. Histologic processing was carried out after resin infiltration, observing classical histopathology and pathologic indexing. Materials comprised 6 tissue-based grafts covering both experimental and commercial porcine decellularised dermal and small intestinal submucosal materials.
Results
Subcutaneous delivery of biologics demonstrated material-specific inflammatory/host responses. Controlled variations of the Permacol™ manufacturing process showed sodium dodecyl sulfate (SDS) was the most proinflammatory decellularisation reagent, and HMDI cross-linking had no effect on host response. All materials remained recoverable after 28 days, although Surgisis™ had partially resorbed.
Conclusion
Differences in host responses exist between biologic implants for hernia repair in this rat model. It is postulated that these modifications are induced during processing and may have an effect on the clinical outcome of hernia repair.
Introduction
Hernias compromise a diverse class of defects generalised by protrusion of an organ or tissue through the wall of the body cavity that normally contains it. In abdominal wall hernias, this is typified by bowel or omentum protruding through a physically compromised musculo-aponeurotic barrier. This results in fibrosis and encapsulation of offending tissue and pain, bowel obstruction and/or strangulation for the patient. Hernias are usually repaired by augmentation of compromised fascia with a prosthetic material, either synthetic or biologic, to restore mechanical integrity and function. Repair with biologic implants has been advocated in contaminated or infected fields because native tissue approximation alone is associated with high recurrences whilst synthetic meshes have high rates of fistulation and erosion (1). The efficacy of some biologic implants in such clinical circumstances has been demonstrated in systematic reviews (2, 3), but debate remains about the performance of other biologic materials (4).
To successfully remodel soft tissue, an implant must be biocompatible, resistant to physical and physiologic stresses, promote in-growth of host tissue and resist adherence to underlying viscera (5). Tissue-based implants are typically xenogeneic (porcine or bovine) or allogeneic and manufactured by decellularisation and delipidation of a number of primary tissues including dermis, small intestine submucosa (SIS) and pericardium. Furthermore, using a variety of chemistries, these tissues can be post processed to include cross-linking to extend their degradation profile, the persistence of their preoperative mechanical properties and their resistance to bacterial proteases such as collagenase when applied in an infected field. Despite exemplary surgical technique, hernia recurrence and chronic pain rates remain problematic due to inflammation, fibrosis and fistulation elicited by exogenous materials (6-7-8).
A fundamental understanding of host response and inflammatory cellular kinetics elicited by implanted materials is a paramount concern in predicting long-term efficacy, integration and mechanical stability (9). Functional wound healing requires precise migration and coordination of secretory and proliferative responses of circulatory and tissue-resident cells, which include a fine balance of the constructive and destructive roles of leukocytes, ever present at an implant site (6). This drives and controls extracellular matrix production and proliferation of appropriate cells to repopulate and remodel damaged tissue, ideally without sacrifice of mechanical properties like strength and elasticity, associated with tissue scarring (10-11-12).
The foreign body and inflammatory responses dictate clinical performance of any surgical device. As such they are both a benefit and risk; excessive inflammation could result in several chronic surgical complications including seroma, fibrosis, postoperative pain and potentialy accelerated material degradation by leukocyte-secreted proteases and reactive oxygen species (13-14-15). Inadequate inflammation can reduce wound debridement, mechanical stability of the material tissue—interface and increase risk of infection and ultimately unsatisfactory wound healing due to absence or dilution of leukocyte cytokine signals to appropriate stromal cells to proliferate and remodel (16-17-18).
Immediately after surgery, stress signals known as damage-associated molecular patterns (DAMPS) which are released by compromised stroma recruit neutrophils. Although the innate role of these cells is to rapidly decontaminate an area from invading organisms, these nonspecific granulocytes can potentially degrade a material before it commences its integrative phase, causing nondiscriminate damage to healthy surrounding tissue (19-20-21). As the inflammatory cascade progresses, neutrophils are accompanied by various classes of leukocyte including monocyte/macrophage cells and professional antigen-presenting dendritic cells (22, 23). As these cells begin wound debridement, their secretome and physical migration make the decision whether to progress the response to a chronic phase and drive production of a specific cellular armoury in the form of T and B lymphocytes (14, 15, 24). Therefore driving inflammation into a constructive wound remodelling rather than destructive tissue degeneration mode is a prerequisite to successful material-mediated surgical augmentation (25).
It is fundamentally acknowledged that, implanted material composition, stiffness, hydrophobicity, porosity and precise anatomical source (tissue-based implants) modify host response. To compound this complex milieu of inflammatory contributors, specific chemistries used up-stream of the implant to decellularise, modify mechanical strength and resist biodegradation by cross-linking native collagen (26-27-28), may also impact foreign body properties of a material. It is therefore vitally important to implant materials with defined and reproducible cellular reactions to maximise the potential for successful surgical intervention without undesirable host vs. graft, or foreign body style complications (4, 29). It has been suggested previously that subtle changes in fabrication chemistries and other preparative steps can influence down-stream wound-healing (30, 31). Although studies have been performed which aim to elucidate the effects of manufacturing variables on inflammatory processes, typically these compare materials broadly across manufacturers, based on disclosed parameters, such as the presence or absence of cross-links, for example. Although valuable from a whole material perspective, it is difficult to deduce from these studies the specific influence of particular fabrication steps. Manufacturers will incorporate several additional proprietary processes into material production, undisclosed to the researchers, which may also modify tissue responses, making it difficult to isolate the role of any single fabrication parameter in influencing tissue physiology (32, 33).
The aim of this study was to evaluate the initial inflammatory and host tissue response toward a number of biologic surgical prostheses. Using commercially available materials in conjunction with defined single-reagent changes to the Permacol™ proprietary processes, we were able to deduce the influence of decellularisation chemistry and cross-linking using HMDI on host responses to porcine tissue-based implants in a subcutaneous animal model with a level of process control rarely published. The animal model was designed to evaluate the initial inflammatory cell interrogation, colonisation and integration of the material over 28 days, to precisely characterise the pathologic response to the grafts without the compounding variable of requiring the prostheses to physically bridge and heal a wound. Additionally, biologic prostheses are often used to repair a diverse variety of tissue areas, therefore it is valuable both scientifically and surgically to not restrict their evaluation to a specific style of defect. For these reasons, a broad inflammatory model was designed.
Materials and Methods
Material Sourcing and Fabrication
Six different implant materials were investigated. Four implants were commercially available and were derived from porcine SIS (Surgisis™ Biodesign™) and porcine dermis (Strattice™ Firm, Strattice™ Pliable and Permacol™). Two implants were experimental and derived from porcine dermis using controlled modifications to the proprietary Permacol™ process. The first implant (implant A) utilised SDS as the decellularizing agent, but the remainder of the process was identical to that for Permacol™. The second implant (implant D) was processed identically to Permacol™ but did not undergo additional cross-linking. The SDS-treated implants were prepared by slicing 1.5-mm-thick sheets of dermal collagen from porcine hides, which were then decellularised. Following decellularisation, samples were dehydrated using acetone and cross-linked with HMDI. Implants were rehydrated in saline, and sterilised by irradiation in foil pouches. The non-cross-linked implants were prepared by slicing 1.5-mm-thick sheets of dermal collagen from porcine hides. Solvent was used to extract lipids and nonfibrous proteins, before decellularisation using trypsin. Implants were rehydrated in saline, and sterilised by irradiation in foil pouches. A summary of the processing techniques used for the prostheses is given in Table I.
SUMMARY OF PROCESSING TECHNIQUES USED FOR MESHES STUDIED
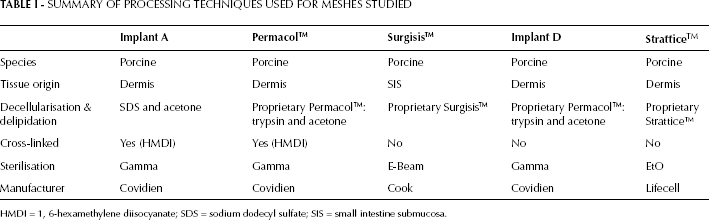
HMDI = 1, 6-hexamethylene diisocyanate; SDS = sodium dodecyl sulfate; SIS = small intestine submucosa.
Animals and Surgery
Implants were cut into 1-cm2 pieces from stock material in a sterile field (class II cabinet) using standard aseptic technique, rehydrated if required (Surgisis™ Biodesign™) and maintained in sterile saline until implanted.
Subjects (male Wistar rats, 6 weeks old, 250-270 g) were anaesthetised using an equal ratio of O2/N2O supplemented with isoflurane at 2% (v/v). Once the subject was sedate (confirmed by absence of a pedal withdraw reflex), the incisional area was shaved and sterilised using surgical iodine and pain relief (buprenorphine) and prophylactic antibiotic (Baytril) administered by subcutaneous injection.
A 1.5-cm-long cutaneous incision was made in the centre of the subject's dorsal side, through which 4 subcutaneous channels were generated using blunt dissection to dissociate attachment of cutaneous tissues to underlying musculature, terminating above each shoulder and hip. Into these channels, materials were inserted using a randomised delivery approach. External wounds were closed using 3 interrupted sutures Polysorb™, Covidien. During in vivo incubation, animals were housed singly and checked daily for defective sutures. Sacrifices were performed at days 2, 5, 7, 14 and 28, which encompassed a suitable time scale to permit host cell interrogation, colonisation and tissue integration to give a representation of the foreign body reaction elicited by the materials and processes in a rapid throughput model. Implants and associated tissue was excised for subsequent processing and analysis.
Animals were euthanised using procedures approved under Schedule 1 of the UK Animals (Scientific Procedures) Act 1986. This was achieved by overdose of an inhalation agent appropriate for the size and species of animal. In this instance, animals were subject to a rising concentration of CO2, taking care that animals were only subject to 100% CO2 once consciousness had been lost, therefore avoiding any visible symptoms of dyspnoea. Death was confirmed by mechanical destruction of the vertebral column.
Animal welfare adhered to UK Home Office use of animals in scientific procedures guidelines and ISO 10993-2, and were approved by the Institutional Review Board of the University of Liverpool.
Histopathology
Fixation and antigen presentation were performed as previously described (34). Briefly, samples were placed directly into periodate-lysine-paraformaldehyde fixative on a roller for 7 days at 4°C, during which fixative was refreshed 3 times. Fixative was removed from samples and replaced with a cold washing solution at 4°C for 24 hours. After this time had elapsed, cold washing solution was replaced by ice-cold acetone to remove phosphate crystals and dehydrate the specimens, for 48 hours at 4°C. Once samples were suitably dehydrated, they were immersed in Technovit infiltration solution for 24 hours at 4°C. Specimens were then cast in place in an appropriately sized mould, orientated for optimum cutting and submersed in Technovit embedding solution (TAAB, UK). The pre-resin surface was covered with mineral oil to prevent oxidation and evaporation, sealed with aluminium foil and placed at —55°C to infiltrate for 4 days, followed by –20°C for 2 days to complete polymerisation. Tissue blocks were sectioned (5 μm) using a polycut microtome (Reichert-Jung, USA) and placed onto microscope slides coated with 3-(aminopropyl)triethoxysilane. Unless stated otherwise, all reagents were purchased from Sigma-Aldrich, UK.
Haematoxylin and Eosin Staining
Samples were heat fixed, rehydrated using a 100%>90%>70% ethanol gradient and submersed into Harris' haematoxylin for 5 minutes, followed by 5 minutes under running tap water to allow colour development. Samples were then differentiated using 1% acid alcohol (1% HCl/EtOH) for 5 seconds, and washed under running water for 1 minute. Samples were then bathed in 1% eosin for 3 minutes and finally washed under running tap water for 1 minute. Specimens were dehydrated using a gradient of ethanol (70%>90%>100%), cleared with xylene and mounted immediately using DPX.
Evaluation
Specimens were evaluated using a pathologic index considering 20 biologic features and cell populations associated with wound healing, infection and foreign body response taking into consideration a number of the pathological features suggested in ISO 10993-6. These parameters were specifically scored — for example: cells (quantity; 0 none — 4 cells abundant), colonisation (host cells in the implant; 0 none — 4 cells throughout), encapsulation/fibroplasia (neo matrix; 0 none — 4 dense matrix deposition) and degeneration (or degradation; implant remaining; 0 none — 4 implant intact).
Each animal received 4 different implants. At each time point (2, 5, 7, 14 and 28 days), 6 samples of each material were delivered across the animals using a semirandomised approach which ensured that each animal never received a particular material more than once. For the purpose of statistical analyses, scorings from all time points from all available samples were combined to create the largest and therefore most statistically significant data set.
Results
After 2 days in vivo, implant A, SDS decellularised porcine dermis, demonstrated a large degree of cellular encapsulation with no notable infiltration into the bulk of the material. The predominant cell population were neutrophils. Surgisis™ showed delamination at this time period with significant cell infiltration. Permacol™, implant D (non-cross-linked dermal implant) and Strattice™ Firm all demonstrated similar histopathologies after 48 hours; with low levels of infiltration of mixed cell populations comprising macrophages, fibroblasts and polymorphonuclear cells not extending beyond the first few micrometres of the material—tissue interface. Strattice™ Pliable, however, showed larger degrees of cellular infiltrate of the same composition as the previous 3 materials but in greater numbers.
At the 5-day end point, implant A had continued to recruit large numbers of polymorphonuclear cells around its periphery without noticeable infiltration into the material bulk. Surgisis™ was completely surrounded with neofibrous tissue in addition to a continued neutrophil presence at the host—tissue interface. Permacol™, implant D and Strattice™ Firm continued to demonstrate similar cellular responses, with small numbers of infiltrating cells from the surrounding connective tissue layers, without significant penetration into the material. In contrast, Strattice™ Pliable demonstrated large amounts of cellular penetration throughout.
After 1 week in vivo, Surgisis™ was almost completely infiltrated by host cells. The material delaminated creating a large, macroporous structure through which host cells could infiltrate. Implant A continued to be surrounded by large numbers of neutrophils without significant infiltration of cells from the interface. The pathologic similarity of Permacol™, implant D and Strattice™ Firm continued with materials demonstrating peripheral fibroplasia; the same was observed for Strattice™ Pliable at this time point (Fig. 1).
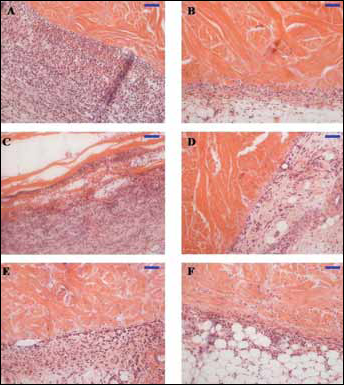
Haematoxylin-and-eosin stained explanted abdominal wall repair patches after 7 days of subcutaneous incubation in vivo. A) Implant A; B) Permacol™; C) Surgisis™; D) Implant D; E) Strattice™ Pliable; F) Strattice™ Firm. Bar represents 100 μm.
Fourteen days postimplantation, the polymorphonuclear cells from the periphery of implant A had infiltrated to form a mixed population of cells which included fibroblasts and macrophages, resulting in a reduction in capsule density as cells migrated into the material. After 14 days, it was not possible to determine differences between the 2 Strattice™ materials, with both Firm and Pliable showing macrophage and fibroblast infiltration and similar interfacial tissue composition. Permacol™ and the non-cross-linked equivalent, implant D, continued to display identical pathology, while Surgisis™ had partially resorbed.
Permacol™ and implant D retained their identical pathology after 1 month in vivo. Implant A demonstrated a continuation of the neutrophil reaction. Similarly to the observations recorded at 14 days, the pathology of the 2 Strattice™ materials had largely normalised after the subtle differences observed throughout the first week of implantation, with similar amounts of cellular infiltrate and interfacial tissue composition (Fig. 2). Surgisis™ had continued to resorb, although material remained recoverable.
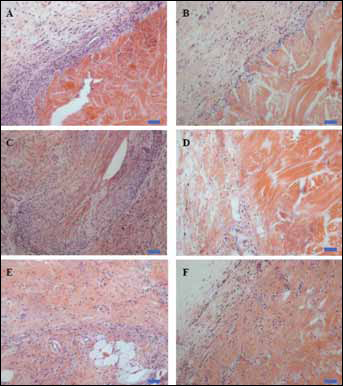
Haematoxylin-and-eosin stained explanted abdominal wall repair patches after 28 days of subcutaneous incubation in vivo. A) Implant A; B) Permacol™; C) Surgisis™; D) Implant D; E) Strattice™ Pliable; F) Strattice™ Firm. Bar represents 100 μm.
Pathologic indexing scores combined for days 2, 5, 7, 14 and 28 are presented in Table II. Surgisis™ had more marked cellular infiltration as well as degeneration. It was not possible to deduce any significant host response differences in response to cross-linking the materials using HMDI, based on direct comparison between Permacol™ and implant D. Implant A, however, the SDS-decellularised Permacol™ equivalent, appeared to display a marked early acute cell response and a retarded cellular colonisation.
PATHOLOGIC INDEXING OF INFLAMMATORY CELLS AND PHYSIOLOGY OF 6 TISSUE-BASED PROSTHESES IMPLANTED SC IN A RAT MODEL
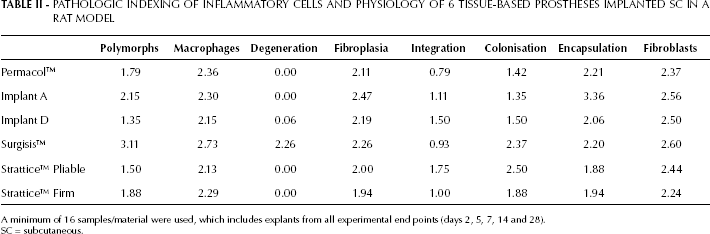
A minimum of 16 samples/material were used, which includes explants from all experimental end points (days 2, 5, 7, 14 and 28).
SC = subcutaneous.
Discussion
Durability of biologic implants used in hernia repair has been postulated to be the key factor in preventing recurrence (4). Biologic implants that degrade/resorb before mature collagen type 1 to scar tissue type 3 ratios are achieved in the healing wound to introduce optimal mechanical compliance to the remodelled tissue may be susceptible to higher recurrence rates. In response to this, inclusion of biochemical cross-linking is often considered, which is generally accepted to extend the longevity of a tissue-based prosthetic, by resisting or retarding degeneration by collagenase and other secreted proteases, favouring this class of materials particularly in infected fields. Preparation of tissue-based implantable materials involves a multitude of processing steps with the goal of removing the maximum amount of original cellular remnants without negatively impacting the mechanical properties of the tissue matrix. These fabrication variables may influence down-stream wound healing and inflammatory responses due to a number of factors: primarily retention of unwanted cellular artefacts within the biomaterial inducing inflammation through leukocyte pattern recognition style pathways. In this preliminary work, we considered key fabrication variables in enzymatic vs. SDS decellularisation and HMDI cross-linking, to elucidate their effects on host response in an appropriate subcutaneous inflammatory animal model. This is a well-established model used in the characterisation of the inflammatory properties of implantable materials. Previous work has demonstrated that significant populations of leukocytes will interrogate a device in this niche as rapidly as the initial hours postdelivery (35). We designed the experiment to extend beyond this to a maximum of 28 days, to also allow us to formulate conclusions with respect to the integration of the materials with surrounding connective tissues. This model, combined with appropriate experimental end points, enabled the precise pathologic responses toward particular fabrication parameters to be elucidated without the compounding variable of mechanical loading in a full-thickness hernia model and with undisclosed proprietary processes.
During the 1-month time course, the only material to display any loss of form was Surgisis™ in the form of delamination. The biodegradation of Surgisis™ has also been demonstrated in a porcine model of ventral hernia which concluded that Surgisis™ was completely resorbed after 17 days (36). The rapid resorption of Surgisis™ has been hypothesised to be compounded by a humeral inflammatory response toward material raised against exposed α-galactosyl residues within the xenograft (37). The rapid delamination seen in this in vivo model reflects clinical outcomes. A recent randomised controlled trial was halted early because of the higher recurrence rates in the Surgisis™ arm (38), and 2 systematic reviews have demonstrated higher recurrence rates than for cross-linked porcine dermis when used in hernia repair (39, 40).
In this study, no differences were observed in tissue in-growth between non-cross-linked implants and a direct HMDI cross-linked equivalent. This is concordant with previous studies using both rodent (41, 42) and porcine (32) ventral hernia models, which demonstrated over time the host response to cross-linked and non-cross-linked equivalent materials normalises despite an initial increased cellular infiltration through the non-cross-linked materials. Early pathologic differences such as cellular infiltration and adhesion formation have been attributed to cross-linking dermal matrices in one study (33). However, that study compared dermis materials from different manufacturers, with distinct decellularisation and delipidation processes, in addition to cross-linking and can as such be considered a material-based study rather than one that isolated findings to one particular fabrication variable. In the present study, as in the study by de Castro Brás et al (41), no pathologic differences were observed between cross-linked and non-cross-linked porcine dermal materials processed in an identical manner with or without an additional cross-linking step using HMDI and the Strattice™ materials also produced from porcine dermal collagen.
Throughout the 1-month time course, none of the dermal prostheses exhibited any observable degradation or reduction in quality of collagenous matrix. Furthermore, it was also not possible to discern differences in any of the materials in their neovascularisation capacity or in their recruitment of lymphocytes, or to observe plasma cells at all, suggesting that the major threat to the integrity of these materials in vivo was during the acute phase of the host inflammatory cascade, further supporting the use of relatively short implant durations in this study. The retention of structural integrity by dermal tissue—derived prosthetics is generally considered to contribute greatly to the facilitation of healthy tissue in-growth (43) and integration with surrounding stromal tissues, as demonstrated herein histopathologically. Interestingly, some animal studies have noted that non-cross-linked biologic prostheses become thin and lose tensile strength after 6 months in vivo (44, 45). The lack of any degradation for Permacol™-based materials has been documented previously in a number of clinical studies, which have concluded that recurrences after Permacol™ are typically a consequence of inappropriate material fixation, rather than an innate mechanical weakness of the material itself (37). This contrasts with the results of other studies where hernia recurrence may be attributed to premature material degradation (4, 44, 45).
Pathologic indexing allowed designation of modifications in host tissue response to a particular material, particularly in the polymorphonuclear cell response and material degeneration and colonisation rates, whereas scores for fibroplasia were considerably more homogenous across the materials (Tab. II). Chronic fibrosis and encapsulation were not observed with any of the implants investigated.
Conclusions
Fabrication of tissue-based prostheses involves numerous manufacturing variables, and the effect of these on down-stream inflammation is not fully understood. Previous studies have compared materials based on single disclosed factors without being certain of the influence of other proprietary manufacturing processes in modulating the observed responses. This study, using controlled variations to the Permacol™ process, in addition to a group of commercially available materials, was able to consider the influence of SDS decellularisation, and HMDI cross-linking in isolation, whilst also comparing dermis against SIS from the perspective of tissue origin. Biologic hernia prostheses implanted subcutaneously in a rat model elicited different foreign body and inflammatory responses according to tissue of origin and processing differences in this in vivo model. SDS induced reagent-specific proinflammatory modifications, and cross-linking dermal collagen using HMDI did not introduce any additional inflammatory/host response characteristics when compared with an exact non-cross-linked equivalent or Strattice™. Strattice™ Firm and Pliable displayed similar pathologies in conjunction with host tissue after an initial greater cellular infiltrate into the pliable variant. Across the entire palette of materials, the only notable cells that varied in number in response to the varying processing parameters were neutrophils.
Future Perspectives
What is Already Known?
Biologic implants are licensed for use in hernia repair and tend to be used in situations where synthetic meshes are contraindicated, such as in contamination. A wide range of implants are available on the market, but it is not known how they compare in terms of host response.
What is New?
This paper characterises the host responses of a wide range of commercially available biologic implants and experimental equivalents, which allows precise conclusions regarding the influence of a number of fabrication variables in down-stream in vivo pathologic response.
Potential Impact on Future Practice
This paper will facilitate appropriate selection of biologic mesh for hernia repair and fundamentally drive future mesh design by outlining acceptable manufacturing criteria for optimally biocompatible implants.
