Abstract
Purpose
The objective of this study was to investigate cancellous bone response to strontium-doped hydroxyapatite (SrHA) in ovariectomized (OVX) rats.
Methods
Hydroxyapatite (HA) and 10%SrHA (HA with 10 mol% calcium substituted by strontium) implants were prepared and characterized by scanning electronic microscopy (SEM), energy dispersive microanalysis (EDX) and X-ray diffraction (XRD). Twelve weeks after bilateral ovariectomy, 20 rats randomly received HA or 10%SrHA implants in the right distal femur, with 10 animals in each group. Eight weeks after implantation, specimens were harvested and analyzed by micro-computed tomography (micro-CT) and histology.
Results
Compared with HA, 10%SrHA raised the percentage bone volume by 42.6%, bone-to-implant contact by 47.1%, mean trabecular number by 27.3%, mean trabecular thickness by 31.5% and mean connectivity density by 37.4%, while it decreased mean trabecular separation by 20.1% in micro-CT evaluation. 10%SrHA also increased the bone area density by 47.6% in histological analysis.
Conclusions
With the HA implants as controls, the 10%SrHA implants were shown to increase bone density and bone-to-implant contact, and improve trabecular architecture in the vicinity of implant surfaces.
Introduction
Osteoporosis was defined by the World Health Organization in 1993 as a systemic skeletal disease that is characterized by a reduction of bone mass and deterioration of bone microarchitecture, with a consequent increase of bone fragility and susceptibility to fracture (1). Many patients needing orthopedic surgery or implant dentistry treatment are elderly with a high incidence of osteoporosis (2). Hydroxyapatite (HA) is a widely used coating material due to its biocompatibility and osteoconductivity, and has been demonstrated to increase peri-implant bone mass in osteoporotic trabecular bone (3, 4). But in osteoporotic bodies, where bone resorption exceeds formation during bone turnover, decreased bone density and bone contact may occur in the vicinity of the implant surface, ultimately leading to loss of implant fixation (5, 6).
Strontium, with a dual action as both anabolic and anticatabolic (7, 8), has been used for the treatment of osteoporosis in the form of strontium ranelate (9, 10). Recently, strontium-doped HA (SrHA) has been successfully prepared, characterized and demonstrated to maintain the dual effects of strontium in in vitro experiments (11-12-13-14). Studies in intact animals indicated that SrHA cement was biocompatible, osteoconductive and superior to HA and polymethylmethacrylate (PMMA) bone cement in terms of bone-bonding strength in revision hip replacement (15, 16). However, reports about bone healing around SrHA at sites with low bone density are limited. We hypothesized that SrHA would induce increased bone density in the vicinity of the material when compared with HA.
In this study, an osteoporotic animal model was established using female rats 12 weeks after bilateral ovariectomy. Rod-shaped HA or SrHA implants were prepared, characterized, then inserted in the distal femur of the ovariectomized (OVX) animals. Eight weeks after implantation, specimens were harvested for micro-computed tomography (micro-CT) and histological analysis.
Materials and Methods
Preparation and of HA and 10%SrHA
HA and 10%SrHA (HA with 10 mol% calcium substituted by strontium) implants were prepared by a chemical coprecipitation method as previously described (11). In brief, analytical grade (AR) Ca(NO3)2·4H2O, Sr(NO3)2 and (NH4)2HPO4 (Merck, Germany) were dissolved in distilled water at a concentration of 0.2 M, to form the Ca+Sr solution (molar ratio of Ca:Sr=9:1) and the P solution. Then P solution was added into the Ca+Sr solution with 400 rpm stirring at 50°C, to reach the final molar ratio of (Ca+Sr)/P at 10/6. The 25% ammonia solution was added to keep the pH value of the reaction solutions above 10. After addition of P solution into the Ca+Sr solution, the precipitate was maintained in contact with the reaction solution for 5 hours at 50°C under stirring. Subsequently, the precipitate was filtered and washed repeatedly with distilled water and absolute ethanol. Finally, the product was dried at 60°C in an oven. The HA was produced by the same process, but without the addition of Sr(NO3)2.
For the characterization and preparation of the HA and 10%SrHA implants, the dried precipitate was ground into powders and sieved through mesh. Powders with a particle size between 2 and 5 μm were collected for this study. These selected powders were then compressed into rod-shaped implants, measuring 1 mm in diameter and 5 mm in length, using a custom-designed mold under a pressure of 20.0 MPa with a tablet machine (769YP-15A; Tianjin High-Tech Co. Ltd, China). The implants (for the in vivo experiment) and some sieved powders (for X-ray diffraction analysis) were then calcined at 1,050°C for 0.5 hours.
Characterization of HA and 10%SrHA
The surface topography of implants was detected using a field emission gun scanning electron microscope (FEG-SEM, Inspect F; FEI, Eindhoven, The Netherlands). To confirm that the SrHA coating had been doped with strontium and to determine the real molar ratio of strontium and calcium in SrHA implants, energy dispersive microanalysis (EDX; Isis 310; Oxford Instruments, Oxford, UK) and inductively coupled plasma mass spectrometry (ICP-MS, VG PQExCell; TJA, USA) measurements were also made. In addition, X-ray diffraction (XRD) patterns of the HA and 10%SrHA powders were determined using the Philips X'Pert Pro MPD diffractometer (PANalytical, Almelo, The Netherlands) at a scanning speed of. 0.75°/min. Cu Kα radiation (λ=1.5418 Å, 40 mA, 40 kV) at a 2θ range of 10°-80° was used. Rietveld refinement of powder XRD was used to perform a quantitative analysis of phase composition. In vitro dissolution tests of SrHA and HA were also performed by exposing 100 mg of powder to 20-mL bicarbonate solution (0.02 M HCO3—, pH 7.4) for 24 hours at 37°C, and the inorganic phosphate release detected by an inductively coupled plasma optical emission spectrometer (ICP-OES; SPECTRO ARCOS, Germany) was used as an index of crystal solubility.
Animals
Twenty 3-month-old female Sprague Dawley rats were included in this study. All of the animals were kept in individual cages under climate-controlled conditions (25°C, 55% humidity, 12-hour/12-hour light/dark cycle). Free access to standard laboratory diet and tap water were provided. All animal experiments were conducted in accordance with the requirements of the Animal Research Committee of Sichuan University.
Surgery Procedure
After bilateral ovariectomy, 12 weeks were allowed to pass for the establishment of the osteoporotic model. Then all rats were randomly divided into 2 groups depending on the different implants they received, with 10 animals in each group. HA or 10%SrHA implants were inserted in the right distal femur as previously described (17, 18). Briefly, a pilot hole was drilled through the intercondylar notch, then a 1.1-mm titanium wire was gradually twisted to make a channel from the distal femur into the medullary canal. Subsequently, the implant was inserted into the channel until beneath the articular surface (Fig. 1). After 8 weeks of healing time, specimens were harvested for the following evaluation.
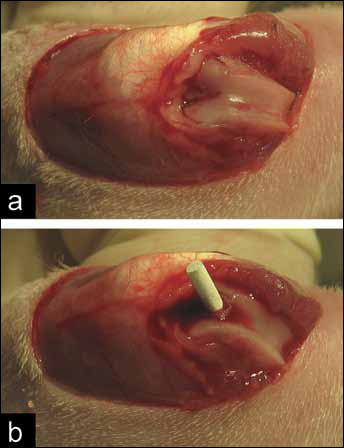
Photographs of the implantation surgery in ovariectomized (OVX) rat: a) with the knee in flexion, the extensor mechanism with the patella was dislocated laterally, and the joint surface of femur was disclosed; b) a pilot hole was drilled through the intercondylar notch, the implant was inserted into the channel until beneath the articular surface.
Micro-CT Assessment
Immediately after dissection, all specimens (10 per group) were kept in normal saline and scanned on a μCT system (μ-CT 80 scanner; Scanco Medical, Bassersdorf, Switzerland) with an isotropic voxel size of 10 μm. The scanning system was set to 70 kV, 114 μA, 700 ms integration time, for maximal signal-to-noise ratio. The constrained 3-D Gaussian filter (σ=1.2, support=1) was used for image noise suppression (19).
The gray scale of HA was similar to that of bone tissue, so it was difficult to distinguish them by computer alone. To define the interface of the implant and the bone tissue, a circle with a diameter of 1.0 mm covering the implant was drawn, and this circle was further dilated by 2 pixels (10 μm for each pixel) to eliminate the possible influence of HA-induced artifacts and irregularity of the implant transection. Then a new interface of implant and bone tissue was obtained, which was recognized as the bone—implant interface in this study.
The volume of interest (VOI) was defined as the 100 slices from the lowest portion of the epiphyseal plate to the direction of trochanter, within the region extending 250 μm from the bone—implant interface. After segmentation, the percentage bone volume (BV/TV), the bone-to-implant contact (BIC; defined as the ratio between bone and total voxels in direct contact with the implant), the mean trabecular number (Tb.N), the mean trabecular thickness (Tb.Th), the mean connective density (Conn.D) and the mean trabecular separation (Tb.Sp; defined as the mean space between trabeculae) was analyzed within the VOI.
Histological Evaluation
After micro-CT scanning, all specimens (10 per group) were fixed in 4% buffered formaldehyde for 2 days at room temperature, and then decalcified in ethylenediaminetetraacetic acid (EDTA; changed every 3 days) for 5 weeks. After decalcification, specimens were washed, dehydrated in gradient alcohol and embedded in paraffin wax. Subsequently, 4-μm-thick transverse sections were cut contiguously. Sections exactly beneath the epiphyseal plate were chosen and stained with hematoxylin and eosin (H&E). Images were acquired and the bone area density (BAD; defined as the percentage area of bone tissue to the whole area) within the region extending 250 μm from the bone—implant interface was quantitatively analyzed using a Leica DMI 6000B micro-system (Germany).
Statistical Analysis
Data were expressed as means ± standard deviation (SD). Statistical analyses were performed using the statistics package SPSS 17.0 (SPSS, Chicago, IL, USA). A comparison between groups was carried out using Student's t test. Differences were considered significant at P<0.05.
Results
Characterization of HA and 10%SrHA
The surface morphological characteristics of HA and 10%SrHA implants by SEM are shown in Figure 2. The microstructure of both implants consisted of tightly agglomerated powders in micro-size. The elemental concentrations of Ca, Sr, P and O were detected by EDX in the FEG-SEM within an area of 15×15 μm2 (Fig. 2). The molar ratio of Sr/(Ca+Sr) was 10.7%, which was 10.6% in the ICP-MS analysis. Crystallographic identification of the HA and 10%SrHA powder was accomplished by comparing the experimental XRD patterns to standards complied by the Joint Committee on Powder Diffraction and Standards. The XRD patterns of both powders showed mainly the phase spectrum of HA, as well as some tricalcium phosphate (Fig. 3). In a quantitative analysis of phase composition (6 samples per group) by Rietveld refinement of powder XRD, the percentage of tricalcium phosphate was 2.50%±0.34% in HA and 3.00%±0.42% in 10%SrHA powders, but without any statistically significant difference. The inorganic phosphate release detected by ICP-OES did not show any significant difference either (data not shown).
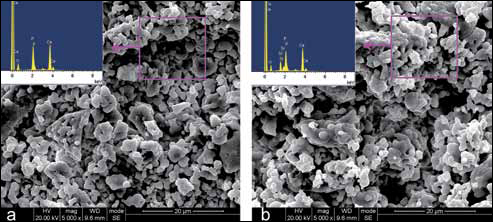
Microphotographs of the surface of hydroxyapatite (HA) (a) and 10%SrHA (HA with 10 mol% calcium substituted by strontium) (b) implants by field emission gun scanning electron microscopy, with the results of energy dispersive microanalysis detection within an area of 15×15 μm2.
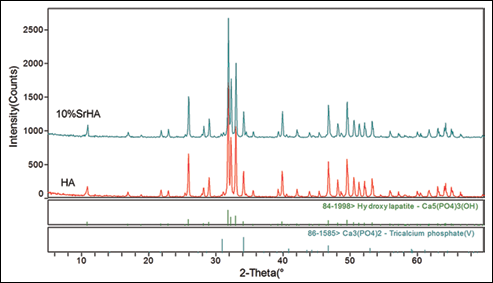
X-ray diffraction patterns of hydroxyapatite (HA) and 10%SrHA (HA with 10 mol% calcium substituted by strontium) powders.
Animals
All of the animals completed the 8-week healing period uneventfully, and the specimens with implants were harvested for the following evaluation.
Micro-CT Assessment
Transverse micro-CT slices beneath the lowest portion of the epiphyseal plate of the femur are presented in Figure 4. Compared with HA, 10%SrHA increased the BV/TV by 42.6%, BIC by 47.1% (P<0.01), the Tb.N by 27.3%, the Tb.Th by 31.5% and the Conn.D by 37.4%, while it decreased the Tb.Sp by 20.1% (P<0.05) in a quantitative analysis (Fig. 5).
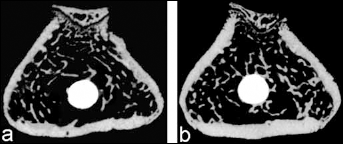
Transverse micro-CT slices from hydroxyapatite (HA) (a) and 10%SrHA (HA with 10 mol% calcium substituted by strontium) (b), beneath the lowest portion of the femur epiphyseal plate.
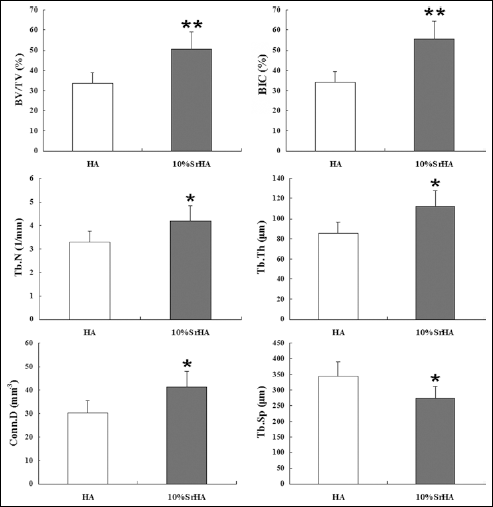
Quantitative results of the micro-CT assessment for hydroxyapatite (HA) and 10%SrHA (HA with 10 mol% calcium substituted by strontium) within the volume of interest. Data were expressed as means ± SD, and the error bars represent SD; n=10 specimens per group. *P<0.05 and **P<0.01, vs. HA group (by Student's t test). BV/TV = percentage bone volume; BIC = bone-to-implant contact; Tb.N = the mean trabecular number; Tb.Th = the mean trabecular thickness; Conn.D = the mean connectivity density; Tb.Sp = the mean trabecular separation.
Histological Evaluation
Transverse histological images beneath the lowest portion of the femoral epiphyseal plate are presented in Figure 6. More tightly connected trabeculae in the vicinity of the 10%SrHA implants were observed in comparison with the HA implants. In a quantitative evaluation, the BAD was 67.3% for the 10%SrHA and 45.6% for the HA implants. Compared with HA, the BAD was increased by 47.6% in the 10%SrHA group (P<0.01).
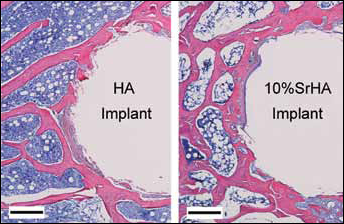
Transverse histological images beneath the lowest portion of the femur epiphyseal plate (stained with hematoxylin and eosin, original magnification ×100). Scale bars = 200 μm. HA = hydroxyapatite; 10%SrHA = HA with 10 mol% calcium substituted by strontium.
Discussion
In this study, HA and 10%SrHA implants were prepared and characterized by SEM, EDX, ICP-MS and XRD. The 10%SrHA implants had a similar surface morphology and phase composition to HA. In vivo experiments using osteoporotic rats induced by bilateral ovariectomy indicated that 10%SrHA increased peri-implant bone density and bone-to-implant contact, and improved the trabecular microarchitecture around the implant compared with HA.
The dual effects of strontium to increase bone formation and inhibit bone resorption have been widely reported, including from the results of in vitro and in vivo experiments and clinical trials (20-21-22-23-24-25). In addition, strontium doped in calcium phosphate or bone cement still showed a positive effect on osteoblasts and an inhibitory effect on osteoclasts in in vitro experiments (26-27-28-29-30). Animal experiments using rabbit or goat models indicated that SrHA cement was biocompatible, osteoconductive, and superior to HA or PMMA cement in terms of bone-bonding strength in revision hip replacements (15, 16). However, reports about bone response to SrHA in osteoporotic bone are limited. Osteoporotic patients, usually the elderly, are frequently in need of implant therapy due to denture or because of bone defects. Due to excessive bone resorption and deficient bone formation in osteoporotic patients, they may be subject to depressed implant fixation (31, 32). Thus, it is imperative to investigate different therapeutic interventions or new biomaterials to increase bone mass and density around implants. Considering the effects of strontium, SrHA may be a promising bone substitute material, implant coating or scaffold for bone tissue engineering.
In this study, the cancellous bone response to 10%SrHA implants was investigated in an ovariectomized (OVX) rat model, a standard animal model for postmenopausal osteoporosis induced by estrogen deficiency. However, why would 10%SrHA in this study increase bone density in the vicinity of implants when compared with HA? The powder size, surface morphology and phase composition of the 10%SrHA implants were all similar to those of the HA implants, and should not be the reason for differences in bone formation. In this study, the XRD pattern of the SrHA seemed to be slightly shifted to higher angles relative to to that for HA, which indicated an increased crystalline size in SrHA (a=b=9.44295 Å and c=6.90661 Å) compared with HA (a=b=9.40539 Å and c=6.86263 Å). But information about the relationship between crystalline size and bone formation was limited. In a previous report using SrHA with an Sr/(Sr+Ca) molar ratio in the range of 12.7%-60.0%, a significantly increased phosphorus release compared with HA was observed (33), but that was not confirmed in this study. Thus, the plausible explanation was that the stronger effect of SrHA on bone mass and quality compared with HA was due to the effect of the strontium itself and not the different characterization of the materials.
Systemic application of strontium has been widely applied but may lead to potential adverse reactions, such as a drug rash with eosinophilia, toxic epidermal necrolysis and systemic symptoms (DRESS) syndrome (34-35-36). Thus, local use of strontium via SrHA might be an alternative method to take advantage of the benefits of strontium and avoid potential adverse reactions. In addition, direct contact of SrHA with bone tissue and the local release of strontium ions may exert even stronger effects on peri-implant bone formation than systemically administered strontium.
The limitations of this study should be pointed out. First, increased bone density around SrHA implants was noted in this study, but the driving force for the increased bone density was not identified. It might be due to increased bone formation, decreased bone resorption or both. Second, increased bone density can be a good thing, but neither overenhanced bone formation nor overinhibited bone resorption (e.g., osteophytosis) is the result we want. Thus, a longer observation time may be needed for evaluation of the long-term efficiency and safety of SrHA. Lastly, the physiological conditions of OVX rats and postmenopausal osteoporotic patients may be different, so caution should be taken in predicting the effect of SrHA in humans.
In summary, the results in this study indicated that 8 weeks after insertion in the distal femur of OVX rats, the 10%SrHA implants increased peri-implant bone density and bone-to-implant contact, and improved trabecular microarchitecture around implants compared with HA implants. It is suggested that SrHA may be a promising material for use as a bone substitute, implant coating or scaffold for bone tissue engineering.
