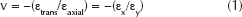Abstract
Materials and Methods
Polycaprolactone (PCL) and polycaprolactone-hydroxyapatite (PCL-HA) scaffolds with 600-μm pore size were fabricated by drop-on-demand printing (DDP) structured porogen method followed with injection molding. Specimens with special dimensions of 4.2×4.2×5.4 mm3 and 6.6×6.6×13.8 mm3 were designed and fabricated for compression and tensile tests, respectively. The mechanical study was performed on both solid and porous PCL and PCL-HA samples. The effect on mechanical properties of the HA content ratio in PCL-HA composites was investigated.
Results
Porous scaffold made of 80/20 PCL-HA composite had an ultimate compressive strength of 3.7±0.2 MPa and compression modulus of 61.4±3.4 MPa, which is in the range of reported trabecular bone's compressive strength. Increasing the concentration of HA in the composites raised compressive properties and stiffness significantly (P<0.05), which demonstrates that PCL-HA composites have the potential for application in bone regeneration. Tensile test of solid PCL and PCL-HA composites showed that the ultimate tensile strength and tensile modulus increased with increases of the concentration of HA in the composites. The tensile test was also conducted on PCL porous scaffold; the result indicated that the scaffold was slightly softer and weaker in tension compared with compression.
Conclusions
Combining compression and tensile test results, our study may guide the possible application of these biomaterials in bone tissue engineering and support further development of microstructure-based models of scaffold mechanical properties.
Introduction
Bone reconstruction is a major health concern worldwide because of a large aging population and increased occurrence of sport-related damage. In the United States alone, there are more than 1.3 million bone-related surgeries annually (1-3). Tissue engineering is a rapidly growing interdisciplinary subject, which offers a promising novel approach to create artificial constructs for organ regeneration, including bone repair (4). In this approach, a porous scaffold is prepared to guide cell attachment, proliferation, differentiation and tissue regeneration in 3 dimensions (3D) (1). The scaffold should also possess a controlled internal architecture, which would favor the growth of blood vessels. There are numerous approaches, such as solvent casting (5), emulsion freeze drying (6), electrospinning (7), salt leaching (8), gas foaming (9), fiber meshes (10), sacrificial compression-molding (11), solid freeform fabrication (SFF) (12) etc., to build a scaffold with a 3D porous structure which serves as substrate for tissue regeneration and provides mechanical support for the ingrowth of new tissue (1, 13, 14). However, most of these methods cannot produce scaffolds with precise structure and controllable pore size. The recent application of SFF in tissue engineering scaffold manufacturing has the advantages that it can fabricate 3D scaffolds with predetermined structure and porosity (12, 13, 15-17), but it is limited by the fact that most of the SFF machines must be adapted to the fluid mechanical properties of each biomaterial under consideration (14, 16, 18). For example, using a drop-on-demand-printing (DDP) or fused deposition modeling method to produce scaffolds, the machine parameters must match the physical properties of the building material – i.e., the viscosity and surface tension. These properties vary greatly among different biomaterials, making the use of a single machine for direct fabrication of scaffolds from multiple biomaterials difficult. With regulation of the machine setup and parameters, SFF can be modified to adapt different materials. For example, De Santis et al have developed an extrusion-based rapid prototyping technique (3D fiber deposition), which can facilitate the 3D porous scaffold printing from pure biopolymer to biocomposites (19-21). To develop an SFF fabrication process in which a single, universal porogen material is used to build porogens that may then be injected with a wide range of biomaterials is another approach. In our group, we combine the SFF technique and reverse engineering to fabricate scaffolds, the manufacturing process (Fig. 1) is based on the following principles and steps: (i) A computer-aided design (CAD) model of bone is created through reverse engineering based on high-resolution images obtained from CT or MRI of the scaffolds (Fig. 1A); (ii) after that, a specially selected material is used to build a structured porogen using SFF technology (Fig. 1B, panel a); (iii) then biopolymer or biocomposite is caste into the porogen and air-solidified (Fig. 1B, panel b). (iv) The porogen is then removed using suitable solvent (Fig. 1B, panel c); and the final scaffold is sintered to improve its structural integrity and mechanical strength for further use in tissue engineering.
A key challenge for bone tissue engineering is to design a 3D scaffold with proper mechanical strength similar to the replaced bone, good biocompatibility with adjacent tissue and adjustable biodegradability so that it will gradually be replaced by new generated tissue. The scaffolds' material for bone tissue engineering should be osteoinductive and osteoconductive, so that osteoblasts can attach and proliferate on the scaffolds. Poly-ε-caprolactone (PCL) is one of the most common artificial biopolymers used for biomedical and pharmaceutical applications, due to its biodegradability, biocompatibility and high permeability to drugs (22-24). It meets most of the requirements for scaffold material and has been fabricated into scaffolds for cell transplantation. However, for bone tissue engineering purposes, the mechanical strength of PCL is not sufficient. Incorporation of filler particles into biopolymers is one of the promising approaches to yield a composite material with improved mechanical properties (25). Currently, research focused on the polymer-ceramic composites is popular in bone tissue engineering because of their favorable biological and mechanical properties, which include good osteoconductivity, osteoinducivity and favorable strength, as well as their toughness and plasticity via the polymer phase, and the greatly improved mechanical stiffness of the composite matrix (26-32).
In all of the ceramics used for bone tissue engineering, hydroxyapatite (HA) is the most widely used bioceramic for hard tissue repair, owing to its similar structure and chemical composition to the mineral phase of bone (33-36). HA has a relatively high mechanical strength in compared with pure PCL and shows osteoinductive and osteoconductive characteristics. However, HA itself is a hard and brittle material, which makes it unsuitable for handling and processing. The incorporation of HA with PCL combines the advantages of both materials and may make the composite suitable for bone tissue engineering.
In this work, standard specimens of neat PCL, 90/10 and 80/20 PCL-HA for compression and tensile tests were produced by injection molding, and the influence of HA on the mechanical properties of the composites was assessed. To fully characterize the mechanical properties of porous scaffolds and examine their feasibility in bone tissue engineering, porous PCL and PCL-HA scaffolds with precise architectural features (600-μm pores) were fabricated by injection molding of the biomaterials into thermoplastic porogens, which were prepared by the DDP method (37). The compressive and tensile properties of both solid specimens and porous scaffolds made of PCL or PCL-HA were studied in detail.
Materials and Methods
Material preparation
PCL pellets (Mw=65,000; Sigma) and hydroxyapatite (HA) powders (Fisher Scientific; the size of the HA particles was 100 nm or smaller according to scanning electronic microscopy) were dried under vacuum before use. PCL-HA composites were prepared from the mixture of PCL and HA. For that, dry PCL pellets and HA powders were weighed and mixed at the desired ratios in an aluminum specimen tray. After melting the mixture at 75°C, the PCL-HA was homogenized using an ultrasonic probe, and reheated as necessary; total mixing time was approximately 30 minutes. Composites were prepared with weight ratios (w/w) of 90% PCL to 10% HA (90/10 PCL-HA) and 80% PCL to 20% HA (80/20 PCL-HA).
Solid specimen preparation
Cylinder specimen preparation
The cylinder specimens with dimension of L (length) = 15.24 mm and D (diameter) = 4.8 mm for compression test were prepared by injection molding according to the ASTM standard D695-02a. Briefly, the degassed molten PCL or PCL-HA composites were injected into the designed mold at 75°C, and then the mold with the injected materials was kept at 75°C for 0.5 hours under vacuum to remove air bubbles. After that, the whole system was cooled to room temperature, and the cylinder specimen was taken out from the mold for further compression testing.
Dogbone specimen preparation
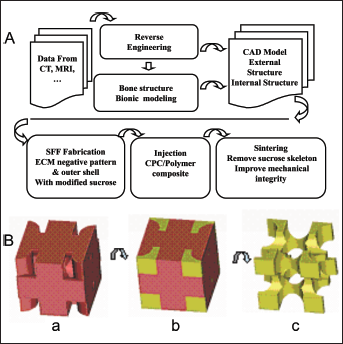
To prepare the dogbone specimen for tensile test, a 3-pieces mold (Fig. 2A, B) was designed by CAD according to the ASTM D638-03 standard and machined at Drexel University machine shop. All of the dogbone specimens were prepared by injection molding, followed by machining operations to ensure all surfaces were free of visible flaws and scratches.
Computer-aided design (CAD) design of dogbone mold:
Porous scaffold preparation
The DDP machine, printing procedure principle, structured porogen design and preparation, as well as porogen-based DDP fabricated scaffolds were described in our previous work (37). Briefly, after injecting molten PCL or PCL-HA composite into the porogen, the materials were allowed to solidify to room temperature. The porogen materials were removed by 99% ethanol under vigorous shaking for 5 minutes with solvent replacement at least 5 times. The scaffolds were washed with DI water (deionized water) for 20 minutes, 3 times after porogen removal, and then air-dried at room temperature overnight. Scaffolds with 4 different pore sizes – i.e., 200, 300, 400 and 600 μm, were prepared.
Specifically, scaffolds with 600-μm pore size were chosen for mechanical tests in this work. For compression test, the cubic scaffold with dimension of ~4.2×4.2×5.4 mm3 was prepared (Fig. 3A). To test the tensile properties of the scaffolds, a longer porous scaffold with a dimension of ~6.6×6.6×13.8 mm3 was designed (Fig. 2C), and prepared (Fig. 3B) following the above procedure.

Scanning electron microscopy
To assess the morphology of the scaffolds, samples were air-dried overnight, sputter coated with carbon and visualized in an XL-30 Environmental Scanning Electron Microscope (SEM; Philips) using an acceleration voltage of 10 k V, as previously described (25). Total of 3-4 images from independent preparations were evaluated.
Microcomputed tomography
Scaffolds with 600-μm pore size were scanned using a SkyScan 1072 Microtomograph (μCT) scanner (Micro Photonics) with 5-μm resolution and 2-μm detectability at 100 kV. The yielded transmission images can be used to reconstruct cross-sections or the complete 3D internal microstructure. Briefly, the image pixel size was set at 6.1 μm, and the output format for each specimen was 976 serial 1,024×1,024 bitmap images. These slice images were viewed in SkyScan's TView software and reconstructed by CT Analyzer software. By selecting darker thresholds, the struts of a specimen were reconstructed. Conversely, by selecting the white levels of the bitmap images, the pores in the specimen could be visualized. Thresholds of the grayscale images were inverted to allow the measurement of the volume of all pore spaces. The porosity was determined by calculating the ratio of pore volume to total volume. Similarly, the contribution of void space to the strut volume was determined by calculating the ratio of strut internal void volume to total strut volume.
Mechanical testing
Compression test
Compression tests of both the solid cylinders and porous scaffolds made of PCL and PCL-HA were performed on a Tinius Olsen H25KT single-column materials testing machine at a compression rate of 1 mm/min according to ASTM D695-02a standard (38). In addition to the Tinius Olsen H25KT machine, a compression test of both of the above cylinders and scaffolds was performed on an Instron 5543 uniaxial testing system using a 1-kN load cell (solid cylinders) or 100-N load cell (scaffolds) with a compression rate of 1 mm/min. Effective stress was computed based on the microscopic specimens' cross-sectional area.
To assess the potential effects of sterilization by ethanol on the mechanical properties, the cylinder specimens were soaked in ethanol for 5 days, and the compression test was conducted on the specimens with the Instron 5543 uniaxial testing system using a 1-kN load cell with a compression rate of 1 mm/min.
The ultimate compressive strength (UCS) and the compression modulus (CM) were calculated from the effective stress-strain diagrams, as previously described (32). Each test was performed on at least 6 specimens of each sample, and the average UCS and CM were plotted as mean values with standard deviation.
Tensile test
Tensile tests of both PCL and PCL-HA dogbone specimens were performed on a Tinius Olsen H25KT single-column materials testing machine at a strain rate of 5 mm/min according to the ASTM D638-03 standard. The width and thickness of each specimen were measured and recorded before test; for each independent sample, 6 specimens were tested. The tensile strength and tensile modulus were computed as the slope of the stress-strain curve.
For the tensile test on porous scaffolds, a pair of aluminum fixtures, which have a square socket to fix both ends of the scaffold and a narrow bar to be gripped on the test machine, were designed and prepared (Fig. 4). Epoxy (Epoxy adhesive 300, Rosco Laboratories Inc.) was used to glue the scaffolds into the socket of each fixture. Tensile test on the PCL porous scaffolds was performed on a Tinius Olsen H25KT single-column materials testing machine at a strain rate of 5 mm/min until failure.

Designed fixture for tensile testing:
To find out the deformation properties of different samples, the Poisson's ratio test was conducted on PCL and PCL-HA dogbone specimens. Before testing, general purpose strain gauges (Vishay micromeasurements & SR-4) were mounted on each specimen to measure the Poisson's ratio. Since the strain gauge is an extremely sensitive device and any small flaw in the bond will affect its performance, extra caution has to be taken while installing the gauge onto specimens. Poisson's ratio was calculated using the ratio of the recorded transverse contraction strain to longitudinal extension strain according to Equation 1:
where ε is the resulting Poisson's ratio, εtrans is the transverse strain (negative for axial tension, positive for axial compression) and εaxial is the axial strain (positive for axial tension, negative for axial compression).
Statistical analysis
All data are presented as means ± standard deviation (SD). The number of samples analyzed is listed for each experiment. Due to unequal variance, comparisons for the compression and tensile tests were performed with a 1-tailed t-test followed by the ANOVA correction for significantly different standard deviations; a P value <0.05 was taken as the threshold for a statistically significant difference.
Results and Discussion
Scaffolds with 4 different pore sizes – i.e., 200, 300, 400 and 600 μm – were prepared by DDP fabrication (38), and their morphology was assessed with SEM. The SEM images of the 90/10 PCL-HA scaffolds (Fig. 5) show that the scaffolds with 400- and 600-μm pore sizes could be fabricated with good shape and integrity (Fig. 5C, D); however, the scaffolds with 200- and 300-μm pore sizes were not that ideal, due to the irregular-shaped pores in the regions which mainly function to maintain the structural integrity (Fig. 5A, B). The PCL porous structure with small pore size (in 2D with 20-μm line widths) was fabricated by Vozzi et al using the microsyringe deposition method, but 3D scaffolds could not be achieved, due to the fabrication limitations (39). The 3D scaffolds with small pore sizes have been fabricated by the direct build SFF method in many groups. For example, Geng et al produced chitosan scaffolds with 200-500-μm pore sizes using the rapid prototyping fabrication method. Their results showed that the produced scaffolds could be customized for overall size and shape for tissue engineering applications (18). Darling et al fabricated PCL scaffolds using precision extrusion deposition of CAD models; scaffolds with 200-300-μm pore sizes and strut widths were confirmed by μCT analysis (40). Zein et al (41) used fused deposition modeling to fabricate PCL scaffolds with 160-700-μm pore sizes, which is similar to the smallest pore size observed in our scaffolds. However, all of the direct build SFF methods mentioned above are limited by the fact that the manufacturing process must be adapted to each printing material. By contrast, the porogen method used in this work requires configuration only once for the ubiquitous porogen which will then be filled with any biomaterials.

SEM images of the 90/10 PCL-HA scaffolds with 200-μm pores (
To make sure of effective mass transfer of oxygen and nutrients to the interior of the scaffold, along with sufficient removal of wastes, it was suggested that a 3D scaffold with a >500-μm pore size might be required to successfully support the bone tissue growth (42). Thus, in this work, scaffolds with 600-μm pore size were chosen for further experimentation. The porosity of the porous PCL, 90/10 and 80/20 PCL-HA was determined by volumetric analysis of 3D reconstructions from μCT data (Fig. 6). For the 600-μm scaffolds, the theoretical porosity based on the porogen design is 59.9%. The measured values for pure PCL, 90/10 and 80/20 PCL-HA scaffolds were 52.6%, 57.2% and 58.2%, respectively. The porosities of 90/10 and 80/20 PCL-HA scaffolds fit fairly well with the theoretically calculated value (<5%). The higher porosities observed in the PCL-HA scaffolds might be caused by the flow resistance of the HA particles, whereas the molten pure PCL flows more freely during injection molding and thus can fill in the porogen more completely. With an increase in the concentration of HA in the PCL-HA composites, the scaffolds become rougher (Fig. 6), which could also be attributed to the large amount of HA particles.
Three-dimensional reconstruction from μCT data.
It is well known that both PCL and HA are biocompatible materials. After porogen extraction, the PCL and PCL-HA scaffolds are still biocompatible and facilitate cell growth and migration (37). Thus, in this work, we mainly focused on mechanical property assessment and the possible influence introduced by the ethanol sterilization process on the mechanical strength of the scaffolds.
Current mechanical tests for most of the bone scaffolds are based on compression testing, since this is the simplest way to evaluate the mechanical properties of scaffolds. However, bending loads create local regions of tensile stress, which indicates that tensile strength is equally important for tissue engineering bone scaffolds. Since compressive strength (CS), tensile strength (TS) and elastic modulus are very important factors for bone scaffold fabrication and also a weak point of most of the bone tissue engineering scaffolds, the compressive and tensile properties of both solid cylinders and scaffolds are presented.
The UCS and CM of PCL and PCL-HA solid cylinders are summarized in Table I. The pure PCL cylinders had an average UCS of 14.1±1.8 MPa and a CM of 215.4±6.6 MPa. The CM was in the reported range of 150 and 200 MPa for pure PCL (38). The 90/10 and 80/20 PCL-HA cylinders had an average UCS of 24.6±0.8 and 25.8±1.1 MPa, and a CM of 447.6±24.6 and 546.1±27.8 MPa, respectively. The UCS increased 79% and 86% from pure PCL to 90/10 and 80/20 PCL-HA composites, respectively; while the CM increased 108% and 154% from pure PCL to 90/10 and 80/20 PCL-HA composites. Both the CM and strength of cylinders improved significantly (P<0.05) with increases in the concentration of HA in the composites, which indicates a particular advantage of PCL-HA composites in scaffold preparation for hard tissue engineering.
ULTIMATE COMPRESSIVE STRENGTH (UCS) AND COMPRESSION MODULUS (CM) OF PCL, 90/10 PCL-HA AND 80/20 PCL-HA CYLINDERS AND 600-μM POROUS SCAFFOLDS, TESTED BY TINIUS OLSEN MACHINE (N=6)
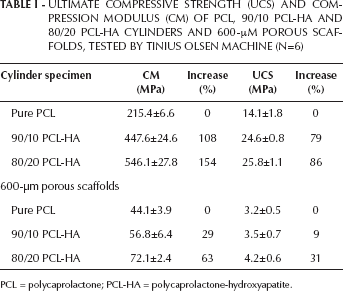
PCL = polycaprolactone; PCL-HA = polycaprolactone-hydroxyapatite.
To assess the feasibility of DDP-produced porous scaffolds for bone regeneration, PCL and PCL-HA scaffolds with 600-μm pore sizes were prepared, and compression tests were performed on the scaffolds. A typical compressive stress-strain curve of PCL porous scaffold is shown in Figure 7, where multiple failure points are presented. The multiple failure points are caused by the collapse of the weakest strut in the scaffold, which happens prior to the collapse of the entire scaffold. The comparison of compression tests on PCL, 80/20 and 90/10 PCL-HA porous scaffolds by the Tinius Olsen machine are presented in Table I. Statistical analysis indicated that the CM was significantly increased (P<0.05) with the addition of HA in PCL; however, UCS does not show any significant difference between the 3 kinds of scaffolds.
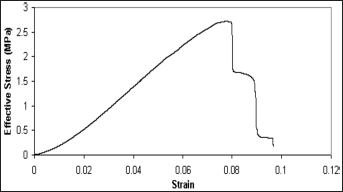
Stress-strain curve for a 600-μm-pore pure polycaprolactone (PCL) scaffold. Multiple failures observed in the curve are due to the failure of the weakest among the struts.
A different testing machine might give a different value for the same specimen, which could further influence the data analysis. To check the consistency of the testing results from the machines with different setup and testing parameters, the compression tests for both solid cylinders and scaffolds were also performed using an Instron 5543 machine. The results summarized in Table II indicate the same trend, in which the mechanical properties of both cylinders and scaffolds are improved with increasing concentrations of HA. However, differences in the data tested by different mechanical testing machines can be observed (compare Tabs. I and II), which suggests that the influence of the testing machine cannot be neglected while doing the data analysis.
ULTIMATE COMPRESSIVE STRENGTH (UCS) AND COMPRESSION MODULUS (CM) OF PCL, 90/10 PCL-HA AND 80/20 PCL-HA CYLINDERS AND 600-μM POROUS SCAFFOLDS, TESTED BY INSTRON 5543 MACHINE (N=6)
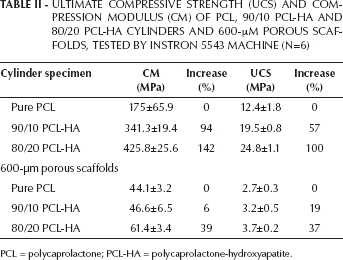
PCL = polycaprolactone; PCL-HA = polycaprolactone-hydroxyapatite.
The compressive strength of the produced porous scaffolds (3.2-4.2 MPa by Tinius Olsen machine and 2.7-3.7 MPa by Instron) is in line with the reported values for trabecular bone from human mandibles (0.2 to 10.4 MPa) (43). It should be noted that the compressive strength of trabecular bone varies greatly with anatomical location and individual factors (such as bone density and volume fraction etc.) for the sample being measured, as well as strain rate (44). Furthermore, the compressive properties of the porous scaffolds fabricated by the DDP method were compared with the mechanical test results of PCL scaffolds with similar porosity reported by other researchers. The CM of our PCL scaffold (~44 MPa) is slightly higher than the result reported by Hutmacher et al (20-42 MPa) (45) and lower than that reported by Williams et al (52-67 MPa) (12), but it was in the range of the 4-77 MPa reported by Zein et al (41). The UCS was essentially equal to a 0.2% offset yield stress in our experiments, due to the brittle failure mode of the samples. The average UCS of our neat PCL porous scaffold (2.7-3.2 MPa) locates it in the range of reported porous PCL scaffolds' yield stress results (0.4-3.6 MPa) (14, 41, 45). The CM and UCS of both the 90/10 and 80/20 PCL-HA scaffolds were higher than the reported values for PCL scaffolds, which is reasonable due to the reinforcement of HA for PCL (14, 41, 45). The small standard deviation (<10% coefficient of variation) of CM and UCS demonstrates the reproducibility of PCL and PCL-HA scaffolds with 600-μm pore size fabricated by the DDP method for bone tissue engineering.
Before biological studies of the scaffolds, proper sterilization has to be performed. When sterilizing PCL or PCL-HA scaffolds with ethanol (37), the scaffolds may degrade during the process, which may influence the future structural integrity and mechanical properties of the scaffolds. To assess the potential effects of the ethanol sterilization process on the mechanical properties, the compression tests were conducted on cylinder specimens soaked in ethanol for 5 days. The results showed that the integrity of cylinder specimens was not affected by ethanol exposure, but their CM and UCS were reduced significantly (Fig. 8). The decrease of CM is 32% for pure PCL, 34% for 90/10 PCL-HA and 43% for 80/20 PCL-HA, while the decrease of UCS was 60% for pure PCL, and 59% and 56% for 90/10 and 80/20 PCL-HA, respectively. The results indicate that the exposure of scaffolds to ethanol will accelerate the degradation of scaffold which could influence its future mechanical strength. Thus it is suggested that to minimize ethanol exposure time during the sterilization process is extremely necessary.
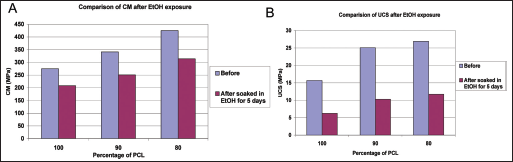
Effect of 5 days of ethanol exposure on compressive mechanical properties of polycaprolactone (PCL) 600-μm pore scaffold.
Tensile tests on solid dogbone specimens made of PCL, 90/10 and 80/20 PCL-HA composites by injection molding were performed using a Tinius Olsen H25KT single-column materials testing machine. The comparison of UTS and TM of PCL, and 90/10 and 80/20 PCL-HA dogbone specimens showed that the increase of HA concentration in the composite raised the TM and UTS as well as the stiffness of the specimens significantly (P<0.05) (Fig. 9). Pure PCL dogbone had an average UTS of 1.9±0.2 MPa and TM of 105±15 MPa. Addition of 10% weight of HA into the dogbone increased the average UTS to 2.1±0.4 MPa (8% increase) and TM to 117±18 MPa (11% increase). The 80/20 PCL-HA dogbone had an average UTS of 2.9±0.3 MPa and a TM of 129±18 MPa, which was a 53% increase in UTS and a 23% increase in TM compared with the neat PCL sample. The results clearly showed that increasing the concentration of HA in the composites increased not only the compressive strength but also the tensile strength, which could be explained by the strong interaction between HA particles and PCL matrix, which transfers the load effectively between HA and PCL in both compression and tensile tests.
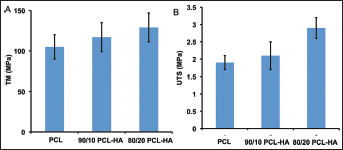
Comparison of the tensile modulus (TM) (
Poisson's ratio for the dogbone specimens was calculated using the recorded transverse contraction strain to longitudinal extension strain (Eq. 1). The mean values ranged from 0.442 for pure PCL to 0.408 for 80/20 PCL-HA (Fig. 10), which is within the range for silicone and some other polymers such as acrylic and polycarbonate (0-0.5). Poisson's ratio under tension showed a general trend for the value to decrease with increasing HA concentration.
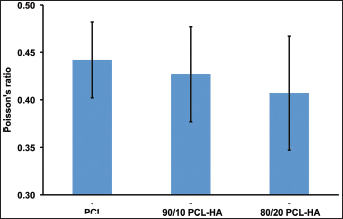
Poisson's ratio for pure polycaprolactone (PCL), and 90/10 and 80/20 polycaprolactone-hydroxyapatite (PCL- HA) composites (n=6).
The performance of tensile tests on porous structural scaffolds is quite difficult compared with on dogbone specimens, due to the difficulty of preparing the porous tensile specimens and the scaffold fixation on the testing machine. Moreover, there is no available standard procedure for tensile testing of porous scaffolds, which makes the tensile test on porous scaffold more difficult. To evaluate the tensile strength of porous scaffolds produced by DDP, and taking into consideration the difficulty of performing standard tensile test procedures on porous specimens, a tensile test on pure PCL scaffolds with 600-μm pore sizes was conducted. Specifically designed PCL scaffold (Fig. 3B) was pulled until failure, and a typical stress-strain curve is presented in Figure 11. The tensile stress–strain curve shows a biphasic behavior: the scaffold is stiffer below about 2% strain, and then it gradually becomes softer up to failure at over 10% strain. The pure PCL scaffolds have a UTS value of 1.4±0.4 MPa and a TM of 25.7±0.5 MPa.
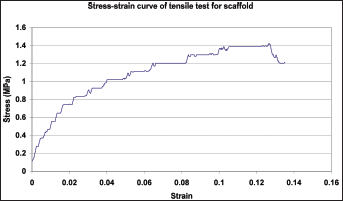
Tensile stress-strain curve of polycaprolactone (PCL) scaffold with 600-μm pores.
Comparing the compressive strength, the values for the tensile strength of both the dogbones and scaffolds are slightly lower. For PCL porous scaffold in this work, the UTS and TM decreased 56% and 41%, compared with UCS and CM, respectively. Accordingly, the UTS and TM of the solid PCL sample decreased by 87% and 51%, compared with UCS and CM. The desired mechanical strength can be regulated through varying the pore size/shape of 3D scaffolds by CAD design and the SFF method. The trend and relationship between the compression and tensile properties will need further analysis. The current results may provide supporting information for developing mechanical performance of biomaterials/composites, and thus guide the application of composite materials in tissue engineering.
Conclusion
PCL, 90/10 and 80/20 PCL-HA composite solid specimens were prepared by injection molding. To fully assess the mechanical properties of PCL and PCL-HA composites, compression and tensile tests were performed on solid cylinder and dogbone specimens, respectively. Compression and tensile tests were conducted on DDP-fabricated porous PCL and PCL-HA scaffolds to measure the mechanical integrity of the scaffolds. The effect of HA content on mechanical properties of PCL-HA solid specimens and porous scaffolds was studied. The results showed the obvious trend that with increasing HA concentration, both the compressive and tensile strength of the composites and scaffolds was improved. Soaking the PCL and PCL-HA cylinder samples in ethanol for 5 days caused a dramatic decrease in compression properties, so that it is recommended that special attention should be paid to the sterilization process, because the procedure uses large amounts of ethanol. This study of the mechanical properties of DDP-fabricated porous scaffolds may have the potential to guide the application of such kinds of scaffolds for tissue engineering and bone repair, and to support development of finite element models which relate scaffold architecture and material properties to scaffold mechanical performance.
Footnotes
Acknowledgements
The authors are grateful to the Centralized Research Facility (CRF) of the College of Engineering, Drexel University, for providing access to the μCT scanner and electron microscopes used in this work.