Abstract
Purpose
The purpose of the present study was to investigate the cleaning efficacy of 2 different nickel-titanium (NiTi) rotary systems: a new single system Reciproc compared with the rotary full sequence Mtwo. The amount of debris and the morphology of smear layer were the parameters for the evaluation of the cleanliness of root canals. The null hypothesis of the study was that there would be no significant difference in debris scores and smear layer scores between the 2 systems.
Methods
Forty single-rooted freshly extracted teeth were selected and divided into 2 groups. For each group, all root canals were shaped with 2 different NiTi instruments (Mtwo and Reciproc) and irrigated with 5.25% NaOCl and 17% ethylenediaminetetraacetic acid (EDTA) solutions by 3 trained operators. Specimens were fractured longitudinally and prepared for scanning electron microscopy (SEM) analysis at a standard magnification of ×5,000. The presence/absence of smear layer and the presence/absence of debris at the coronal, middle and apical third of each canal were evaluated using two 5-step scales for scores. Numeric data were analyzed using the Kruskal-Wallis and Mann-Whitney U statistical tests, and significance was set at a P value <0.05.
Results
This study revealed significant differences among the various groups. The Mtwo group presented significantly lower smear layer scores than the Reciproc group (P<0.01) in the middle and apical third of the canal.
Conclusions
Conventional continuous rotary NiTi instruments associated with 5.25% NaOCl and 17% EDTA solutions seem to be better for obtaining clean dentinal canal walls. Reciprocating instruments leave a higher quantity of smear layer which covers dentinal walls and dentinal tubules.
Introduction
Nickel-titanium (NiTi) rotary instruments represent a relatively new approach to the rapid preparation of canals with standardized taper (1-3). In the last decade, many NiTi rotary instruments have been introduced. Several studies (4-6) have demonstrated that they could efficiently create a smooth funnel-form shape with minimal risk of transporting the canals. NiTi rotary instruments were introduced to improve root canal preparation (7). These instruments are commonly associated with a risk of fracture. Fracture may be caused by normal cyclic stresses caused by bending (fatigue failure) and due to shear stresses exceeding the elastic limit of the alloy caused by torque (ductile failure) (8-10). Both the clinician and the instrumentation technique used play a significant role in preventing large shear stresses caused by torque. These stresses dramatically increase when an excessive pressure is applied on the handpiece (11), when the contact area between the canal walls and the cutting edge of the instrument widens (12, 13) or when the canal cross-section is smaller than the inactive tip of the instrument (12, 13). The last case can cause what has been described as taper lock, a phenomenon usually occurring with regularly tapered instruments (14). This risk may be reduced by performing coronal enlargement (15, 16) and by creating a manual (17, 18) and/or mechanical (19) glide path before using NiTi rotary instrumentation. Alternating rotary movement has been proposed as an alternative to continuous rotation in order to reduce the risk of instrument fracture and root canal deformity (20). NiTi instrument binding decreases with the use of an alternating movement, reducing instrument block, and the torsional stress is thus induced (21). Many authors (20-22) have proposed a shaping technique based on the use of conventional NiTi files designed for continuous rotation with reciprocating movement, but today new instruments specifically projected for reciprocating motion are available. A reciprocating motion alternates between clockwise and counterclockwise rotation and may decrease the impact of cyclic fatigue on NiTi rotary instrument life compared with rotational motion (20, 21). When the instrument rotates, it is subjected alternately to compression and flexion, which can produce microfractures and can finally lead to instrument fracture (23).
The new NiTi Reciproc files (VDW, Munich, Germany) are claimed to be able to completely prepare and clean root canals with only one instrument (single-file system). Reciproc files are made of a special NiTi alloy called M-wire that is created by an innovative thermal treatment. The benefits are increased flexibility and improved resistance to cyclic fatigue. The reciprocating working motion consists of a counterclockwise (cutting direction) and a clockwise (release of the instrument), while the angle of the counterclockwise cutting direction is greater than the angle of the reverse direction.
The purpose of the present study was to investigate the cleaning efficacy of 2 different NiTi rotary systems: the new single system Reciproc compared with the rotary full sequence Mtwo. The amount of debris and the morphology of smear layer were parameters for the evaluation of the cleanliness of root canals obtained after instrumentation. The null hypothesis of the study was that there would be no significant difference in debris scores and smear layer scores between the 2 systems.
Materials and Methods
A total of 40 single-rooted human teeth freshly extracted for periodontal reasons were selected for this study and placed in saline at room temperature immediately after extraction. The inclusion criteria were morphological similarity, single-canal roots, straight roots, absence of root decay and absence of previous endodontic treatment. The crown of each tooth was removed at the level of the cementum-enamel junction (CEJ) to obtain root segments of approximately 12 mm in length. Two longitudinal grooves were prepared on the palatal/lingual and buccal surfaces of each root with a diamond bur used with a high-speed water-cooled handpiece to facilitate vertical splitting with a chisel after canal instrumentation. In teeth with intact root apices, patency of apical foramina was standardized using size 10 stainless steel K-Files (Dentsply Maillefer). Teeth with apical diameters larger than size 15 or with an altered apex were not included in the study.
Root canal instrumentation
Teeth were randomly numbered and divided into two groups: group A (n=20): Mtwo (Sweden & Martina, Due Carrare, Padova, Italy); and group B (n=20): Reciproc (VDW, Munich, Germany).
Root canals were irrigated with 1 mL of 5.25% NaOCl (Niclor 5; Ogna, Muggiò, Italy) followed by 1 mL of 17% ethylenediaminetetraacetic acid (EDTA; E.D.T.A. 17%; Ogna, Muggiò, Italy) after each change of instrument for group A and after 3 in-and-out movements for group B. After canal preparation, a final 1-mL aliquot of 17% EDTA was left in situ for 2 minutes and replaced by 1 mL of 5.25% NaOCl for 3 minutes. A final flush with ethanol for 30 seconds concluded the preparation. Each canal was dried with calibrated paper points (Absorbent Paper Points; Denstply-Maillefer, Konstanz, Germany).
All irrigation procedures were delivered with a 27-gauge needle (Molteni, Firenze, Italy). All instruments were set into permanent rotation with a 6:1 contra-angle handpiece (Sirona, Bensheim, Germany) powered by a torque-limited electric motor (VDW Silver Reciproc motor; VDW). For each Mtwo file, the individual torque limit and rotational speed programmed in the file library of the motor were used, while Reciproc files were used in a reciprocating working motion generated by the motor.
Samples were prepared with two different instruments:
Group A: All Mtwo instruments (Sweden & Martina, Due Carrare, Padova, Italy) were used with the single-length technique according to the manufacturer's instructions using a gentle in-and-out motion at 300 rpm and maximum torque (6 N/cm). Four instruments were used: Mtwo 10/.04, Mtwo 15/.05, Mtwo 20/.06 and Mtwo 25/.06. Once the instrument had negotiated to the end of the canal and had rotated freely, it was removed.
Group B: A R25 Reciproc file (VDW, Munich, Germany) having a size at the tip 0.25 mm and a taper of 0.05 over the first 3 mm was used in a reciprocating, slow in-and-out pecking motion on the dedicated reciprocating motor (Silver Reciproc, VDW) used with the manufacturer configuration setup (at the preset program “Reciproc ALL” specific to the Reciproc instruments). The flutes of the instrument were cleaned after 3 in-and-out movements and working length was reached with slow in-and-out pecking motion.
Scanning electron microscopy preparation and examination
Each sample were dipped in liquid nitrogen immediately after canal preparation and split longitudinally into 2 halves (24) with a stainless steel chisel. The sections were then prepared for scanning electron microscopy (SEM) analysis. The samples were dehydrated in graded concentration ethanol solutions (FLUKA; Sigma-Aldrich S.r.l., Milan, Italy) and then gold (Au=99.99) sputtered (Agar Sputter Coater B7340; Agar Scientific Ltd., Stansted, UK) and observed with SEM (EVO MA 10; Carl Zeiss SMT AG, Germany). After a general survey of the canal wall from the apex to the most coronal part, 6 SEM photomicrographs were obtained at a standard magnification of ×5,000 at each third (coronal, middle and apical). Each root segment was approximately 12 mm in length: 4 mm for coronal, 4 mm for middle and 4 mm for apical third. The images were saved digitally with specific software (Smart Sem User Interface), and scored by 3 trained operators blinded to the preparation procedures.
Scoring system
The absence or presence of debris and smear layer were rated and scored with the system proposed by Hülsmann et al (25). Separate evaluations were recorded for debris and smear layer, the cleanliness of each root canal was evaluated in 3 areas (coronal, middle and apical third of the root) with the following numerical scales:
Score system for debris (dentine chips, pulp remnants and particles loosely attached to the canal wall):
Score 1: clean root canal wall, only a few small debris particles.
Score 2: a few small agglomerations of debris.
Score 3: many agglomerations of debris, covering less than 50% of the root canal wall.
Score 4: more than 50% of the root canal wall covered by debris.
Score 5: complete or nearly complete root canal wall covered by debris.
Score system for smear layer (dentine particles, remnants of vital or necrotic pulp tissue, bacterial components and retained irrigant):
Score 1: no smear layer, with orifices of dentinal tubules open.
Score 2: small amount of smear layer, and some dentinal tubules open.
Score 3: homogenous smear layer covering the root canal wall, only a few dentinal tubules open.
Score 4: complete root canal wall covered by a homogenous smear layer, with no open dentinal tubules.
Score 5: heavy, homogenous smear layer covering the entire root canal wall.
Statistical analysis
Debris and smear layer scores were separately recorded. Data were analyzed with the Kruskal-Wallis test. The Mann-Whitney U test was performed for post hoc comparisons. Significance was set at a P value <0.05.
Results
The mean amounts of debris and smear layer scores of the 2 groups are reported in Tables I-IV.
AVERAGE SCORES FOR DEBRIS IN CORONAL, MIDDLE AND APICAL THIRD OF CANALS

Values with the same superscript letters were not statistically different at P=0.05.
SUMMARY SCORES FOR DEBRIS
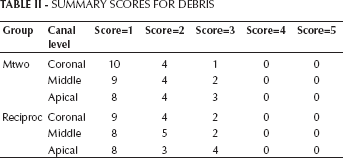
AVERAGE SCORES FOR SMEAR LAYER IN CORONAL, MIDDLE AND APICAL THIRD OF CANALS
Values with the same superscript letters were not statistically different at P=0.05.
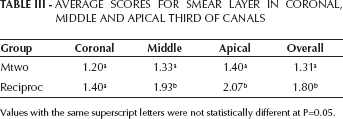
SUMMARY SCORES FOR SMEAR LAYER
The Kruskal-Wallis test showed the presence of significant differences among the different groups (P<0.05).
The Mann-Whitney U test showed no significant difference in debris scores between the Mtwo and Reciproc groups when comparing coronal, middle and apical thirds (P>0.05). Moreover, the Mann-Whitney U test showed no significant difference in smear layer scores between Mtwo and Reciproc groups when comparing coronal thirds (P>0.05). But when comparing both middle and apical thirds, the Mtwo group showed significantly lower scores than the Reciproc (P<0.05).
Figures 1 and 2 show representative samples of scanning electron micrographs (magnification ×5,000) of the root canal dentin surface of groups A and B.
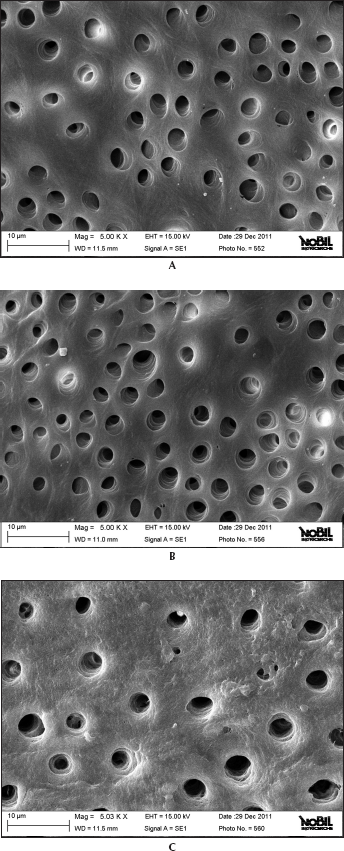
Representative samples of scanning electron micrographs of the root canal dentin surface instrumented with Mtwo (group A) at coronal, middle and apical third of the root (magnification ×5,000).

Representative samples of scanning electron micrographs of the root canal dentin surface instrumented with Reciproc (group B) at coronal, middle and apical third of the root (magnification ×5,000).
Discussion
The null hypothesis for the present study was rejected. Significant differences were found between the 2 groups for the NiTi instruments used.
The goal of endodontic treatment is to remove all necrotic or vital organic tissue and dentin debris created by instrumentation from the root canal system and to create root canals free from bacteria (26). It is well-known that during root canal preparation, the action of endodontic instruments produces a smear layer, which is compacted along dentinal walls (27). Its elimination seems to be of great importance, since it can allow NaOCl to penetrate more easily into the dentinal tubules, thus enhancing its bactericidal action (28-30). Moreover, the smear layer may affect the sealing efficiency of root canal obturation, acting as a physical barrier interfering with adhesion of sealers to canal walls (31, 32).
All NiTi rotary instruments have been shown to produce moderate to heavy smear layers that need to be removed with the use of irrigating solutions. Chelating agents such as EDTA are currently used to remove the smear layer formed during preparation of the root canals (33). The association of EDTA and NaOCl solutions is the gold standard in chemomechanical preparation of the root canals.
A funnel-shaped canal with a circular base is not the common configuration in root canal anatomy, and recently, cross-sectional root canal configurations have been classified as round, oval, long oval, flattened or irregular (34). Preparation of infected root canals requires removal of the inner layer of dentin, and for NiTi instruments it is particularly hard to achieve when preparing long oval root canals (34). So the NiTi file preparation of nonround canals leaves a large portion of the canal surface uninstrumented. If the NiTi instrument does not work on dentine surface, no smear layer is created by the instrument (35).
EDTA acts upon the inorganic components of the smear layer, decalcifies the peritubular and intertubular dentine and leaves the collagen exposed. Subsequently, the use of NaOCl dissolves the collagen, leaving the entrances of the dentinal tubules more open and exposed (33). For this reason, an irrigation regimen similar to the methodology purposed by Foschi et al (3) was used, with the alternation of EDTA and NaOCl at each change of instrument for conventional rotary instruments. In the same way, alternation of EDTA and NaOCl was performed after 3 in-and-out movements of the Reciproc single file.
All instruments were evaluated in accordance with the manufacturers' directions. All protocol and instrument operative sequences were followed. Irrigation procedures were standardized for all experimental groups. The same trained operator shaped all root samples.
This study demonstrated that the 2 NiTi rotary systems produced different dentine surfaces on root canal walls. SEM analysis revealed that conventional rotating files associated with EDTA and NaOCl irrigation leave dentine surfaces substantially free from any smear layer. Despite some minor differences, all continuously rotating NiTi instruments were able to remove the smear layer produced during instrumentation and subsequently dissolved by EDTA. Previous SEM studies investigated the effect of NiTi rotary instruments on dentine and obtained similar results (3, 35-38). The combination of NaOCl and EDTA was probably responsible for the removal of the smear layer and for the removal of a large portion of the circumferential dentine collagen and mineralized dentine wall from the greater part of the tubules, as confirmed by Foschi et al (3). The present study also confirmed that the apical third was the area where more debris was still visible under SEM inspection (3).
It is likely that rotating NiTi instruments produce fine dentine particles and shavings that are spread and compacted along dentine walls and then partially dissolved by EDTA and removed coronally via flute spaces. Mtwo, thanks to its “italic S” cross-section with only 2 cutting edges, favors debris elimination and gives SEM images generally free from smear layer, with a major part of the dentinal tubules completely opened.
From the SEM pictures, the Mtwo instruments seemed to be very aggressive. They produced more debris and removed more dentine than the WaveOne system. Hence, the Mtwo produced large amounts of smear layer that was removed by EDTA, and this suggests a great/adequate capacity to remove debris created during instrumentation. SEM pictures of the WaveOne suggest a soft and incomplete action in removing dentine. Despite this modest activity on the surface, it creates a packed and less soluble smear layer that EDTA is not able to remove. In other words, WaveOne seems to produce a “well-packed” smear layer. This is not a desirable property.
In the literature, few studies have investigated the effectiveness of reciprocating files in removing smear layers from root canal walls. Even if associated with EDTA and NaOCl irrigating solutions, this study demonstrated they are not so effective in removing any smear layer produced during endodontic instrumentation. A lot of smear layer was present along the dentinal walls, in the apical and middle portions.
Only a previous study by Bürklein et al (38) investigated cleaning effectiveness of reciprocating single-file systems (19). The study compared Reciproc and WaveOne (reciprocating instruments) versus Protaper and Mtwo (continuous rotating instruments) using SEM photographs. In the present study, cleanliness was evaluated in 3 areas (apical, middle and coronal third), with separate evaluations for debris (×200 magnification) and smear layer (×1,000 magnification). All instruments were used according to the manufacturers' protocols, with only 2.5% NaOCl irrigation after each instrument. No EDTA was employed. For debris removal, Mtwo and Reciproc instruments achieved significantly better results than the other instruments in the apical third of the canals. In the middle and coronal parts, no significant differences were found among Mtwo, Reciproc and Wave One, while Protaper showed significantly more residual debris. The results for remaining smear layer were similar and not significantly different for the different parts of the canals. This means that, under the conditions of the Bürklein et al (38) study, the use of Mtwo and Reciproc instruments resulted in better canal cleanliness in the apical part compared with the ProTaper and WaveOne. But completely cleaned root canals were never found in this study. It is important to note that EDTA was not used. This certainly affected the overall results.
In this study, many samples instrumented with continuous rotating instruments showed completely cleaned dentine canal walls, while greater presence of smear layer was scored in samples treated with Reciproc, even when irrigating protocols with 5.25% NaOCl and 17% EDTA were respected.
The major objective of the study by Bürklein et al (38) was to compare the shaping and cleaning effectiveness of the different instruments, so they selected a simple irrigation protocol with only NaOCl, avoiding any influence from different irrigation solutions and chelating agents. The cleaning efficiency evaluated in the present investigation was enhanced using a combination of NaOCl and EDTA as a chelating agent, to correspond to clinical conditions, in accordance with previous studies (3, 33-35).
Within the limitations of this study, and not in accordance with the results of Bürklein et al (38), reciprocating instruments seemed to be not as effective in promoting the cleanliness of root canal walls and in removing any smear layer from dentine. Reciprocating instruments leave a higher quantity of smear layer, and due to this fact, dentinal tubules are not completely opened. Further investigations are needed to verify or refute these conflicting preliminary results.
Footnotes
Acknowledgements
We are grateful to Dr. Clara Cassinelli (Nobil Bio Ricerche S.r.l., Portacomaro, Asti, Italy) for providing the SEM images and technical assistance.
