Abstract
Background:
There is an unmet need for identification of additional prognostic markers for lung cancer. The aim of this study was to identify novel clinical and immunological predictors of prognosis in lung cancer patients.
Methods:
Lymphocyte subsets CD3+, CD4+, CD8+, CD4+/8+, CD25+, CD69+, CD44+ and CD54+ were quantified in peripheral blood using flow cytometry, for 203 newly diagnosed lung cancer patients and 120 healthy controls.
Results:
The levels of CD3+, CD4+, CD8+, CD4+/CD8+ and CD69+ lymphocytes were significantly lower in patients with lung cancer compared with the healthy control group, while CD54+ and CD44+ lymphocytes were significantly higher. In stage III/IV patients with lymph node metastasis or distant metastasis, the levels of CD44+ and CD54+ lymphocytes were significantly increased compared with patients with stage I/II disease (p<0.05). The levels of CD44+ and CD54+ lymphocytes markedly reduced after chemotherapy, and follow-up analysis indicated that patients found without increase of CD44+ and CD54+ lymphocytes after chemotherapy had survival advantages. Independent predictors of survival in lung cancer patients included clinical stage (hazard ratio [HR] = 2.791; 95% confidence interval [95% CI], 1.42-3.54, p<0.001), CD44+ lymphocytes (HR = 1.282; 95% CI, 1.02-1.49, p = 0.002) and CD54+ lymphocytes (HR = 1.475; 95% CI, 1.22-1.73, p = 0.003). Elevated levels of CD44+ and CD54+ lymphocytes correlated with poor prognosis in lung cancer patients.
Conclusions:
Peripheral blood lymphocyte subsets in patients with lung cancer are different from those in healthy people, and circulating CD44+ and CD54+ lymphocytes seem to be a promising criterion to predict survival in lung cancer patients undergoing chemotherapy.
Introduction
Lung cancer is the leading cause of cancer-related deaths worldwide in both men and women (1-3). About 90% of all lung cancer cases are related to smoking; however, environmental tobacco exposure and some occupational dust exposures are also risk factors for lung cancer (4-6). Despite advances in early detection and standard treatment, lung cancer is often diagnosed at an advanced stage, and the overall 5-year survival rate is only 10%-15%. The prognosis is strongly associated with the disease stage at diagnosis, performance status (PS), weight loss, sex, plasma lactate dehydrogenase (LDH) level and treatment (7-9). In patients undergoing surgical resection, 30%-60% of patients (depending on their stage) develop a recurrence and die from this disease. Consequently, the prognosis remains unsatisfactory, primarily due to frequent tumor recurrence and metastasis (10, 11). Clearly, there is an unmet need for identification of additional prognostic markers for a more informed process of treatment.
There is increasing evidence of a relationship between immune status and prognosis of people with cancer. Research has proven that new forms of T cell-based immunotherapies have synergistic effects and, possibly, result in improved clinical outcomes for cancer patients (12-15). Previous studies have shown that patients with cancers of the breast, liver and oral cavity have significant cellular and humoral immune changes in their affected tissues (16), which provide useful prognostic markers. These immunological parameters are a reflection of the interactions between the tumor and the immune system.
Patients with cancer have impaired immune function, which may be related to tumor progression in these patients. The alteration of human lymphocyte phenotypes in lung cancer patients may be used to evaluate the systemic immune status of patients. This provides an opportunity for serial tracing of the immunological changes before and after treatment. CD44+ lymphocyte is a receptor for hyaluronic acid and can also interact with other ligands, such as collagens, osteopontin. The function of CD44+ lymphocyte is controlled by its posttranslational modifications (17). CD44 protein participates in a wide variety of cellular functions including tumor metastasis. The establishment of metastasis requires that tumor cells acquire new adhesion and migration properties to emigrate from primary sites and colonize distant organs. As a cell–cell and cell–extracellular matrix adhesion protein, CD44 is well positioned to facilitate this process. Furthermore, the interaction of CD44 with other cellular proteins involved in motogenesis and proteolysis is a determinant factor in cell migration and invasion (18, 19). Intercellular adhesion molecule-1 (ICAM-1, also called CD54), a member of the immunoglobulin supergene family, is an inducible surface glycoprotein that mediates adhesion-dependent cell-to-cell interactions (20, 21). CD54 may facilitate movement of cells through the extracellular matrix (22) and plays an important role in lung cancer cell invasion (23). It is associated with an unfavorable prognosis in chronic lymphatic leukemia and Hodgkin’s disease (24, 25).
The aim of this cross-sectional study was to explore the distribution of different subtypes of peripheral blood lymphocyte in healthy adults and people with lung cancer using flow cytometry, and to explore whether CD44+ and CD54+ lymphocytes are able to provide prognostic information for lung cancer patients.
Materials and methods
Study population
Cases of newly diagnosed lung cancer patients (n = 203) who had been hospitalized for treatment were collected from September 2012 to March 2014 in the Ningbo First Hospital. The diagnosis of all patients was confirmed by surgical pathology or biopsy, and patients with concurrent severe infectious or autoimmune diseases were excluded. All patients were subject to a first blood-taking before any treatment for their cancer was given. Healthy adults (n = 120) as a control group were chosen from the healthy population who had accepted a physical examination in this hospital. All patients with lung cancer and participants in the control group signed an informed consent form.
Tumor staging and tissue typing
This staging is based on results of inspection using fiberoptic bronchoscopy, chest computed tomography (CT), B ultrasound, magnetic resonance imaging (MRI), positron emission tomography (PET)/CT and so forth, in accordance with the 8th edition of the Union for International Cancer Control (UICC) TNM classification in 2017. There were 169 cases of non-small cell lung cancer (NSCLC), including 134 cases of adenocarcinoma, 31 cases of squamous cell carcinoma, 4 cases of other types and 34 cases of small cell lung cancer (SCLC). Patients were stage I in 12 cases, stage II in 16 cases, stage III in 83 cases and stage IV in 92 cases.
Detection of lymphocyte subsets
Lymphocyte subsets were detected using a FACS Calibur flow cytometer and Smultest IMK-Lymphocyte which is a lymphocyte subpopulation kit from Becton Dickinson (USA). The 2-color fluorescent antibody in the kit was fluorescein isothiocyanate and algae red protein-labeled murine anti-human monoclonal antibody.
For 203 patients and 120 healthy controls prior to being hospitalized for treatment or physically examined, 2 mL of venous blood was taken from each in a fasting state in the morning. This was anticoagulated with heparin and submitted to the laboratory within 2 hours. Corresponding monoclonal antibody (20 μL) and 100 μL of heparin anticoagulated plasma to be tested were added in the flow test tube; mixed evenly for 30 minutes of full reaction at room temperature without light. Hemolysin was added 2 times (2 mL each time), shaken and mixed evenly and placed without light at room temperature for 5 minutes; then subjected to 5 minutes of 1,200 rpm centrifugation with supernatant discarded. Phosphate buffer was added, shaken, mixed evenly; then subjected to 5 minutes of 1,200 rpm centrifugation with supernatant discarded again. Fixing liquid (300 μL) was added, shaken and mixed evenly; then tested using the instrument. Finally, the data were processed and analyzed.
Statistical analysis
The results were expressed as means ± SD. SPSS17.0 was used for analysis of the results. The data were analyzed using an independent sample t-test and paired t-test. Survival curves were generated using Kaplan-Meier survival plots. The correlation between cumulative survival and clinicopathological features was analyzed by Cox analysis. A p value of <0.05 was considered statistically significant.
Results
Patients’ characteristics
Patients were aged 44-81 years with a median age of 64 years, including 171 patients ≥60 years old and 32 patients <60 years old; 142 men and 61 women. One hundred and sixty-six patients received 4-6 cycles of chemotherapy concurrently using 2 drugs containing platinum, and 98 patients were subjected to a second blood-taking for testing 3-4 weeks after chemotherapy. In the control group, 120 healthy individuals (85 men, 35 women, median age 62 years) who matched the cancer group for age and sex were included. All patients were followed up by telephone. The follow-up ended on December 31, 2016, from which 12 patients were lost, and the survivor analysis was conducted with the last follow-up date as the truncated value.
Comparison of lymphocyte subsets between lung cancer group and healthy control group before chemotherapy
The comparison of lymphocyte subsets between the lung cancer group and healthy control group before chemotherapy showed a statistically significant difference in 7 indexes except CD25. In patients with lung cancer, CD3+, CD4+, CD8+, CD4+/CD8+ and CD69+ lymphocytes were significantly lower than those in the healthy control group, with the difference being statistically significant (p<0.01) (Tab. I), while CD54+ and CD44+ lymphocytes were significantly higher than those in the healthy control group, with the difference being statistically significant (p<0.01).
Contrast of lymphocyte subsets in peripheral blood between lung cancer patients and healthy control group
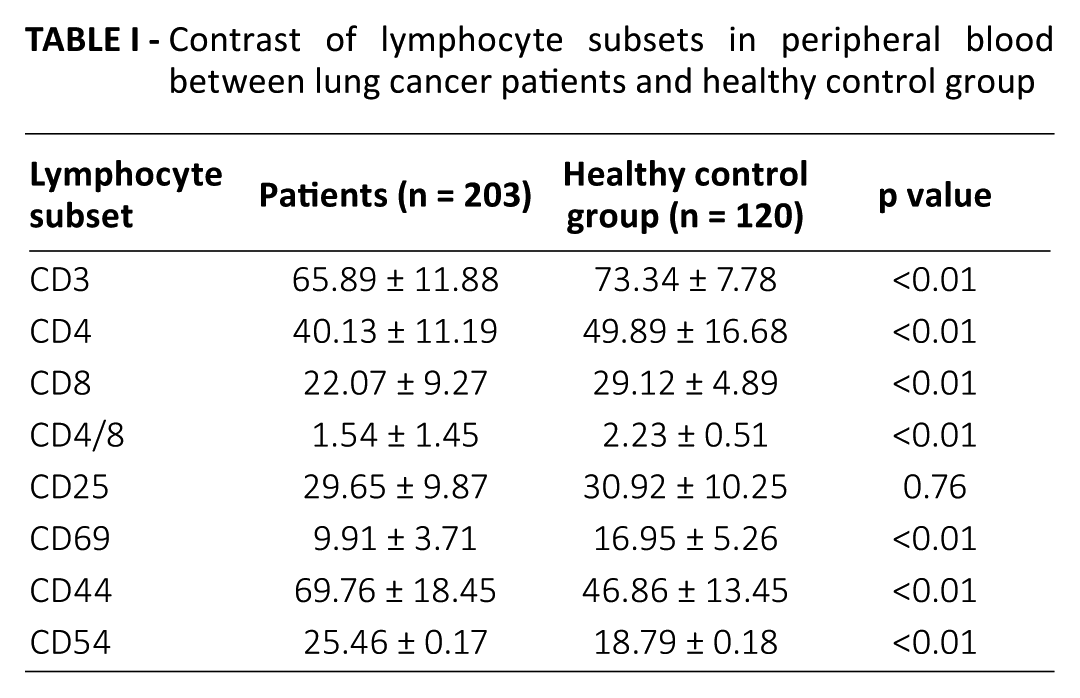
Comparison of peripheral blood lymphocyte subsets in lung cancer group
Classification and statistics were carried out according to sex, age, T/N/M staging system, clinical stage and tissue type. Results show that there were no significant difference in levels of CD3+, CD4+, CD8+, CD25+, CD69+, CD54+ and CD44+ lymphocytes in peripheral blood in male and female patients with lung cancer (Tab. II). The levels of CD3+, CD4+, CD8+ and CD69+ lymphocytes in patients with lung cancer older than 60 years were significantly lower than in those younger than 60 years (p<0.05). CD25+ and CD69+ levels in the T1/T2 group were significantly higher than those in T3/T4 group (p<0.05). CD25+ and CD69+ were significantly lower in patients with lymph node metastasis than in those without, while CD44+ and CD54+ lymphocytes were significantly higher than in those without (p<0.05). CD44+ and CD54+ lymphocytes were significantly higher in patients with distant metastasis than in those in the M0 group (p<0.05). The levels of CD44+ and CD54+ lymphocytes in patients with stage III/IV were higher than in those in stage I/II, while CD3+, CD4+, CD8+, CD4+/CD8+, CD25+ and CD69+ lymphocytes were lower than in those in stage I/II, with the difference being statistically significant (p<0.05). The results of comparisons of patients with different pathological types showed that the expression levels of CD3 in patients with SCLC were significantly lower than in those with adenocarcinoma or squamous cell carcinoma (p<0.05). The other indexes were not statistically significantly different (p>0.05) (Tab. II).
Correlation between lymphocyte subpopulations and clinicopathological parameters
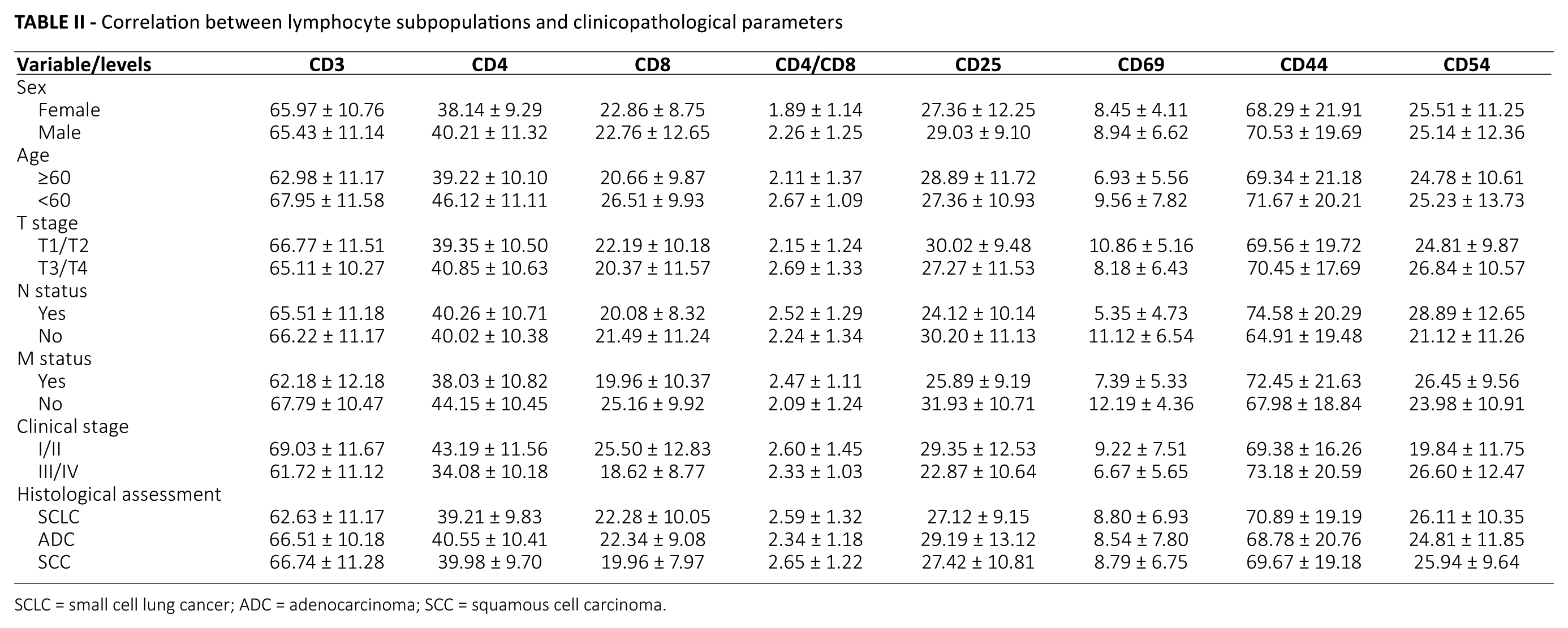
SCLC = small cell lung cancer; ADC = adenocarcinoma; SCC = squamous cell carcinoma.
Comparison before and after chemotherapy
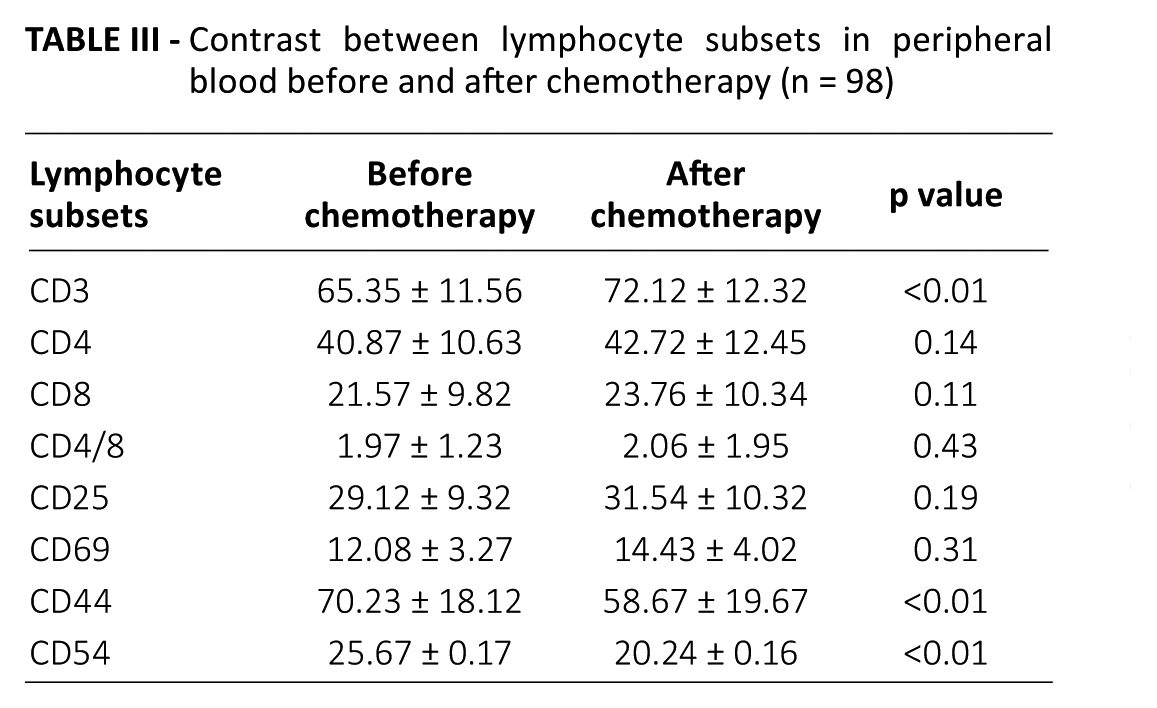
The comparison before and after chemotherapy showed that after chemotherapy, the levels of CD3+, CD4+, CD8+, CD4+/CD8+, CD25+ and CD69+ lymphocytes were all higher than before, but only the difference of CD3+ lymphocytes was statistically significant (p<0.01). CD44+ and CD54+ lymphocytes decreased significantly after chemotherapy (Tab. III).
Contrast between lymphocyte subsets in peripheral blood before and after chemotherapy (n = 98)
After chemotherapy, the number of patients found with increased CD44+ lymphocytes was 47, and those not increased 51; Kaplan-Meier analysis showed that those without increased CD44+ lymphocytes after chemotherapy had survival advantages (p = 0.033) (Tab. IV, Fig. 1A). In addition, after chemotherapy, the number of patients found with increased CD54+ lymphocytes was 54, and those not increased 44; Kaplan-Meier analysis showed that those without increased CD54+ lymphocytes after chemotherapy had survival advantages (p = 0.009) (Tab. IV, Fig. 1B). The number of patients found with both CD44+ and CD54+ lymphocytes increased was 21 after chemotherapy, and those not increased for either CD44+ or CD54+ lymphocytes were 27. Kaplan-Meier analysis showed that those without increased CD44+ or CD54+ lymphocytes after chemotherapy had survival advantages (p = 0.001) (Tab. IV, Fig. 1C). So, the combined expression of CD44 and CD54 can also predict survival of lung cancer patients.
Contrast of survival times between increased CD44 and CD54 group and not-increased group after chemotherapy
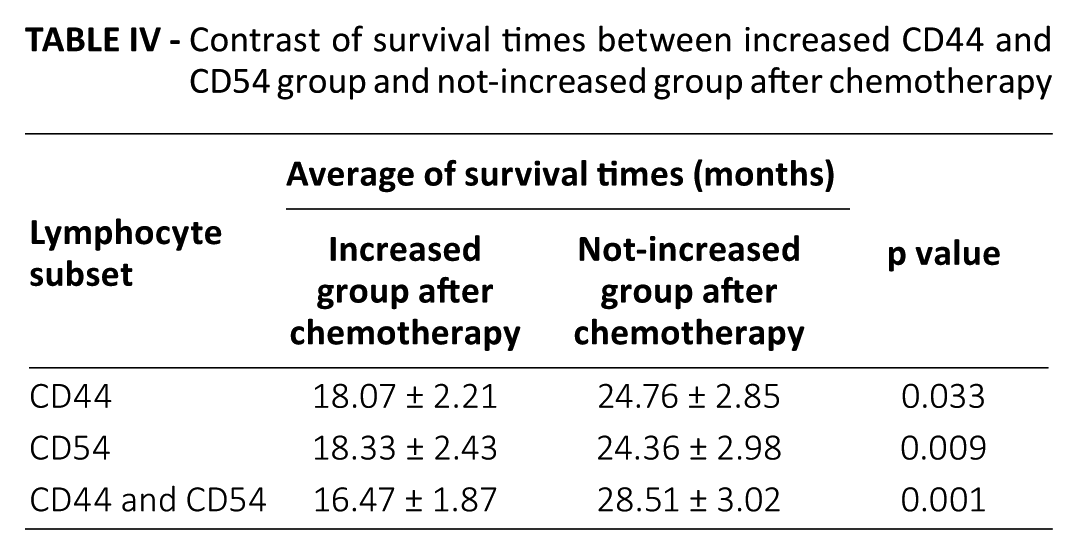
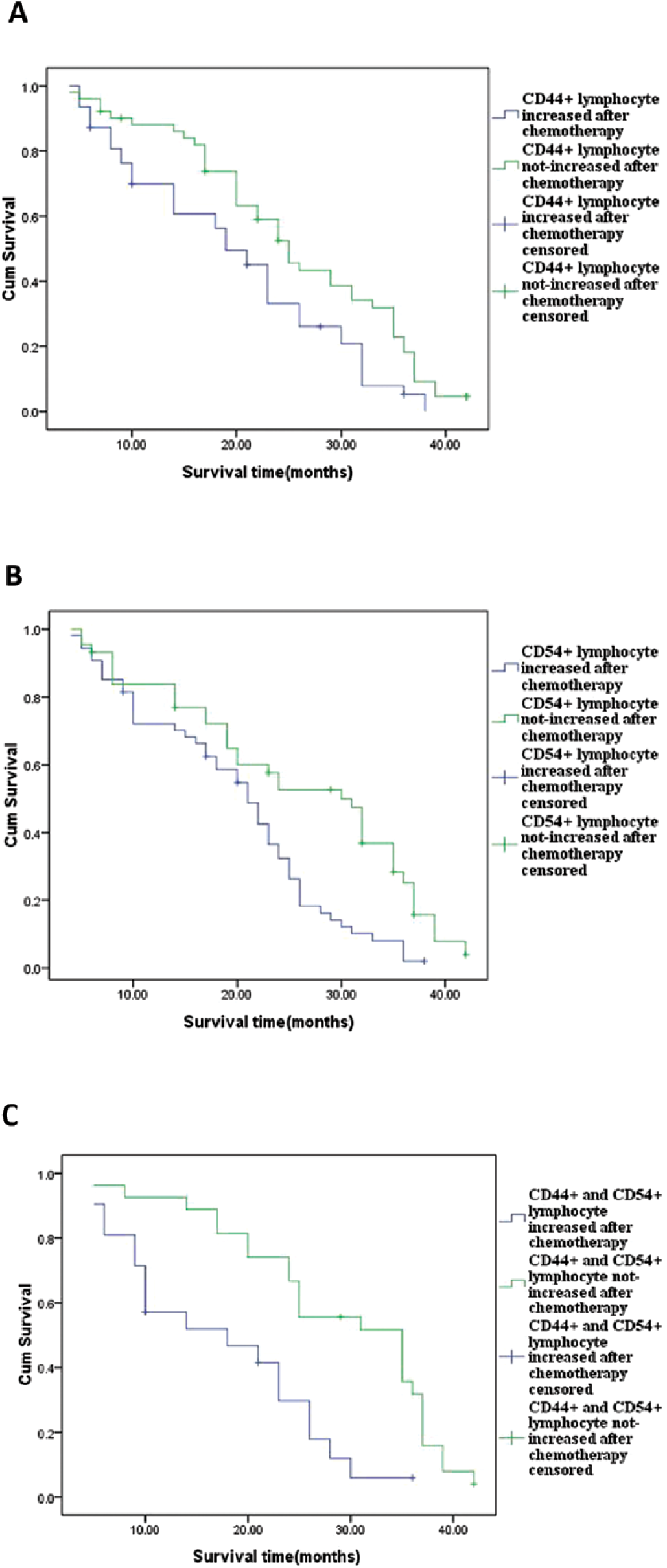
Survival curves for patients with not-increased CD44+ lymphocytes after chemotherapy showed they had greater survival rates (p = 0.033) (
Multivariate analysis comparing outcomes based on altered circulating peripheral blood cells
Multivariate analysis with a Cox regression model based on univariate analysis showed that clinical stage and CD44+ and CD54+ lymphocyte levels were independent predictors of survival in lung cancer patients (p<0.05). The risk ratios of clinical stage and CD44+ and CD54+ lymphocytes were 2.791 (95% confidence interval [95% CI], 1.42-3.54), 1.282 (95% CI, 1.02-1.49) and 1.475 (95% CI, 1.22-1.73), respectively, after adjusting for competing risk factors such as histology type, sex, age, T stage, N stage and lymphocyte subsets (Tab. V).
Multivariate analyses of the associations between clinicopathological variables and mean survival time based on univariate analysis in 98 lung cancer patients
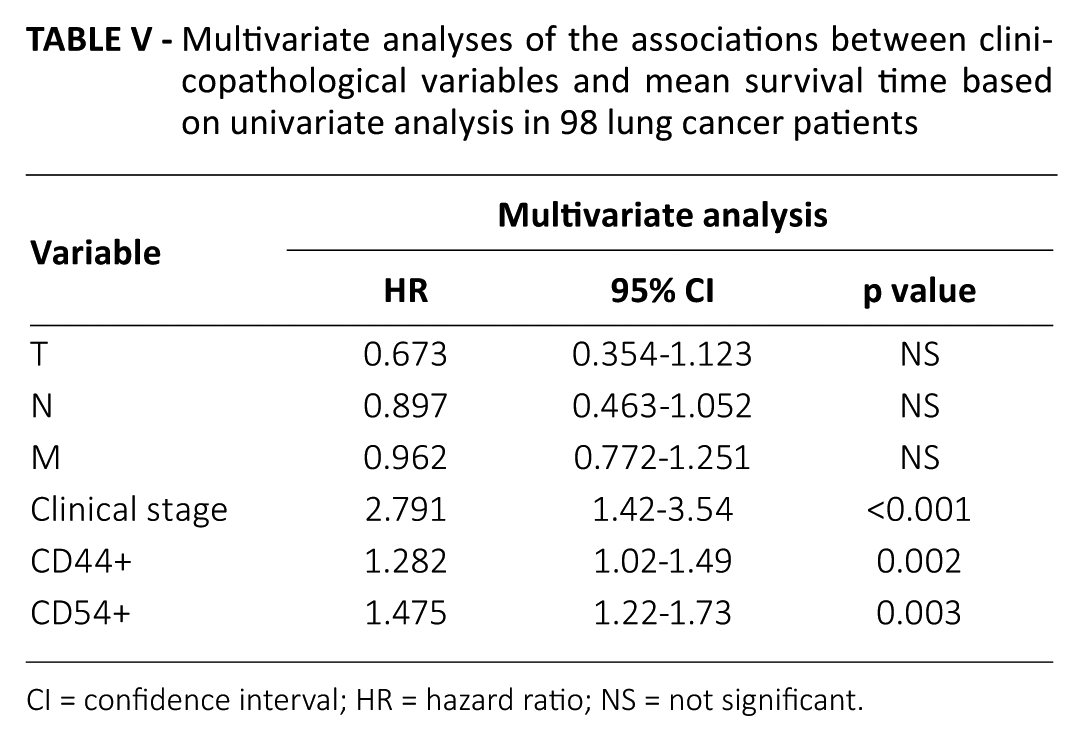
CI = confidence interval; HR = hazard ratio; NS = not significant.
Discussion
B and T lymphocytes belong to a group of white blood cells and play a crucial role in cell-mediated immunity, especially antitumor immunity (26, 27). The immunosurveillance theory suggests that the immune system routinely patrols the cells of the body, and upon recognition of a cell or group of cells that has become cancerous, it will attempt to destroy it, thus preventing the growth of some tumors (26-29). In cancer patients with low immune function, tumor cells easily escape the immune monitoring of the body, thereby creating the conditions for the development of the tumor. In this study, levels of CD3+, CD4+, CD8+ and CD69+ lymphocytes in peripheral blood of patients with lung cancer were significantly lower than in those of normal controls, which further confirmed the suggestion that T and B lymphocytes were damaged during tumor progression. Furthermore, in the lung cancer group, the levels of CD3+, CD4+, CD8+, CD25+ and CD69+ lymphocytes were significantly lower in stage III/IV than in I/II (p<0.05); and in patients with lymph node metastasis or distance metastases or who were in the T3/4 group were lower than in those without metastases or who were not in the T1/2 group. All of these differences were significant (p<0.05). The changes of the indexes indicated that the more advanced the patient’s condition was, the worse their immune status became. This is consistent with previous reports of Yang et al (30) in NSCLC patients and Lee et al (31) in patients with head and neck carcinoma. The decline in CD4+/CD8+ ratio was reported to be involved in tumor progression as the ratio directly reflects the status of the organic T-lymphocyte immune function (32-34). This could be the reason for the lower percentage of CD4+/CD8+ lymphocytes in the III/IV group as compared with the I/II group.
The traditional view considered that tumor chemotherapy directly killed tumor cells or induced tumor cell apoptosis, and meanwhile these standard therapies cause immunosuppression. But this traditional view has been challenged in recent years. Studies have shown that platinum-based standard chemotherapy could stimulate antitumor immunity, in particular by the induction of tumor-specific CD8+ T cells and by promoting infiltration of inflammatory antigen-presenting cells (APCs) harboring T-cell costimulatory ligands into the tumor (35, 36). Recently, Talebian Yazdi et al (37) also demonstrated that standard platinum-based chemotherapy used in advanced pulmonary adenocarcinoma patients has no profound adverse effects with respect to immune cell composition and function. In our study, after chemotherapy, the levels of CD4+, CD8+, CD25+ and CD69+ lymphocytes in peripheral blood of 98 lung cancer patients were slightly higher than in those before chemotherapy, which confirmed that the impact of chemotherapy on immune function is not negative.
CD44+ and CD54+ lymphocytes participate in cell–cell and cell–matrix interactions (21, 38, 39) and mediate functions such as extracellular matrix cell adhesion (40, 41). These attributes are essential for the invasion, migration and metastasis of tumor cells and could possibly explain the increase in CD44+ and CD54+ lymphocytes in the M1 group as compared with the M0 group (p<0.05). The results we found were similar to those in the previous studies – namely, CD44 or CD54 expression levels were closely related with tumor occurrence and development, especially tumor metastasis (40-44). The measurement of lymphocyte subsets in peripheral blood lymphocytes is likely to reflect the immune response of patients and therefore may be linked to survival in lung cancer patients. Our results showed that the increases of CD44+ and CD54+ lymphocytes were strongly correlated with the decrease of mean survival time of lung cancer patients, which was demonstrated by the significantly shorter survival times in patients with high CD44+ and CD54+ lymphocytes as compared with those with low CD44+ and CD54+ lymphocytes after chemotherapy. Cox regression model analysis indicated that CD44+ and CD54+ cell count were independent predictors of survival (p<0.05). The risk ratio of CD44+ and CD54+ lymphocytes after adjustment for competing risk factors of sex, age and stage of disease was found to be 1.282 (95% CI, 1.02-1.49) and 1.475 (95% CI, 1.22-1.73), respectively, suggesting that CD44+ and CD54+ lymphocytes are important factors in determining the outcome of lung cancer patients after treatment and that the mortality rate could increase with an increase in CD44+ and CD54+ lymphocytes.
In conclusion, lung cancer patients have low levels of immune function and their immune status deteriorates with the progression of cancer. The clinical monitoring of immune function on a regular basis is helpful in predicting disease changes and assessing prognosis. This study also found that CD44+ and CD54+ lymphocytes are related to tumor metastasis and clinical staging and have prognostic significance for predicting outcomes in chemotherapy patients.
However, this study had some limitations. The number of patients was relatively small, as the data were collected from a single institution. The underlying mechanism, which might involve the role of CD44+ and CD54+ lymphocytes in tumor invasion and metastasis, needs to be further investigated.
Footnotes
Acknowledgements
We are grateful to Dr. Guoqing Qian for helpful comments.
Disclosures
Financial support: This research was supported by a grant from the Ningbo No. 1 Hospital Science and Technology Fund (No. 2013YJ20).
Conflict of interest: The authors declare they have no conflicts of interest regarding the present study.
