Abstract
Background:
This study aimed to investigate staging changes for Chinese breast cancer patients assessed by the 7th (anatomic) and 8th (prognostic) editions of the AJCC staging manual, and to explore the predictive factors for these changes.
Methods:
Data of patients who received curative surgery for stage I-III breast cancer at Ningbo Medical Center Lihuili Eastern Hospital were retrospectively reviewed. The assessment of staging was according to the criteria of the 7th and 8th editions of the AJCC staging manual. Univariate and multivariate logistic regression analyses were performed to analyze the associations between staging changes and clinicopathological characteristics.
Results:
Staging changes were found in 59.37% of patients and were more likely to be seen in stage IIIA (96.10%) and IIA (85.94%), then IIB (70.33%), IB (68.75%), followed by IA (36.17%) and IIIC (30.08%). In univariate analysis, staging changes were associated with tumor location, clinical tumor size, clinical axillary lymph node status and Ki67 index. However, multivariate analysis found that staging changes were significantly associated with tumor size >2 cm (odds ratio [OR] = 3.263, 95% confidence interval [95% CI], 2.638-4.036), lymph node involvement (OR = 2.261, 95% CI, 1.830-2.794) and high Ki-67 index (OR = 1.661, 95% CI 1.343-2.054).
Conclusions:
Our study demonstrated that there were marked staging changes when 2 different editions of the AJCC staging manual were used. Since prognostic biomarkers are available in routine clinical practice, the more recent staging manual should be followed to select better systemic therapy and give better outcomes for Chinese breast cancer patients.
Keywords
Introduction
Breast cancer has become the leading malignant tumor for women worldwide in the last decade. In 2013, more than 1.8 million women were diagnosed with breast cancer, and approximately 471,000 died of this neoplasm (1). Anatomic factors have always been the cornerstones for staging, in which careful definitions of categories for primary tumor (T), regional lymph node involvement (N) and distant metastases (M) have been refined to reflect updates in technology and clinical evidence. Therefore, TNM staging based solely on anatomic extent of disease has been used to guide decisions regarding whether to apply systemic therapy.
Evolving knowledge of breast cancer biology and the increased validation of various biomarkers for prognosis suggest that several biomarkers should be documented at the first diagnosis. These biomarkers, such as histological grade (HG), estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2) and so on, have become equally or even more important than the anatomic extent of disease to define prognosis, select the optimal systemic therapy, and increasingly, influence the selection of locoregional treatment.
Recently, the 8th edition of the American Joint Committee on Cancer (AJCC) staging manual on breast cancer was published online. In this version, prognostic factors including HG, ER, PR and HER2 are incorporated into the staging manual. Compared with the anatomically based staging manual, the application of the prognostic staging manual assigns 41% of cases to a different group, with either a better or worse outcome. It must be pointed out that these data were based on the US National Cancer Database (NCDB), which reflected the staging changes in well-developed countries. To the best of our knowledge, there has not been any report describing the staging changes in developing countries. Therefore, we attempted to describe the staging changes by using the 7th and 8th editions of the breast staging manuals, and to explore the predictive factors for these changes.
Patients and methods
Patients
This study enrolled breast cancer patients who had undergone breast and axilla surgery at Ningbo Medical Center Lihuili Eastern Hospital between March 2008 and December 2016. All patients were histologically proven to have an invasive ductal carcinoma, and had complete data for clinicopathological features. Exclusion criteria were the following: (i) special type of breast carcinoma, such as medullary carcinoma or tubular carcinoma; (ii) incomplete data for tumor extent; (iii) incomplete biomarkers of ER, PR, HER2 and Ki67 as well as HG; (iv) previous neoadjuvant chemotherapy or neoadjuvant endocrine therapy; (v) bilateral breast cancer; or (vi) inflammatory breast cancer. The principles outlined in the Declaration of Helsinki were followed, and all patients provided written informed consent before their operation.
Data collection
Clinical data were collected and retrospectively reviewed for age, sex, menstrual status, tumor size, tumor laterality, HG, ER, PR, HER2 and Ki67 index. The Nottingham combined HG was adopted in our study. The grade for a tumor was determined by assessing 3 morphological features: tubule formation, nuclear pleomorphism and number of mitotic figures. A score of 1 (favorable) to 3 (unfavorable) was given to each feature, and the scores were added up. If a combined score was between 3 and 5, it was designated as grade I; if 6 or 7, it was considered grade II; if 8 or 9, grade III (2). According to the revised version of the immunohistochemical testing guidelines in 2010, ER and PR assay should be considered positive if immunostaining is seen in more than 1% of tumor nuclei (3). HER2 positivity was defined as either IHC3+ or gene amplification by fluorescence in situ hybridization (FISH) (4).
After curative surgery for the breast and axilla, adjuvant chemotherapy, radiotherapy and endocrine therapy were administered in appropriate patients according to currently available clinical practice guidelines. Trastuzumab, an anti-HER2 monoclonal agent, was not systematically administered until 2013. Since then, a duration of treatment for 1 year has been recommended for all HER2-positive patients.
The incorporation of tumor grade and ER, PR and HER2 status in the 8th edition of the AJCC staging manual was supported by several studies, one of which was performed by David J. Winchester. However, that analysis was unpublished. Another aim of our study was to investigate the differences in staging changes in developing and developed countries. Due to the unavailable data from the analysis by David J. Winchester, we selected another study, performed by Chen et al (5). In their study, a total of 160,880 patients registered on the NCDB between 2004 and 2011 were enrolled, with 50.34% of patients older than 60 years, 78.68% with ER positivity and 68.58% with PR positivity.
Staging and restaging
All eligible cases were staged using the 2 versions of the AJCC staging manual, the 7th (anatomic) and the 8th (prognostic) editions. The latter took into account not only the information of anatomic parameters but also some prognostic biomarkers, including HER2, ER, PR and HG. The detailed staging manual was found at http://www.cancerstaging.org.
Statistical analysis
Univariate analysis with chi-squared test or Fisher’s exact test was performed to detect predictors for staging change. Then, multivariate analysis including all variables from the univariate analysis that were related to staging change was performed to test the factors’ independence. Statistical significance was defined as a p value <0.05, and hazard ratio (HR) and 95% confidence interval (CI) were also calculated. Statistical tests were 2-sided, and analysis were performed using SPSS v.19.0 Software (SPSS, Chicago, IL, USA).
Results
In our study, a total of 1,617 eligible patients with invasive ductal breast carcinoma were enrolled. The overwhelming majority of patients were women, and only 8 patients were men. The incidence of patients aged 35 or younger and those aged >35 was 10.88% and 90.12%, respectively (Tab. I).
Demographics and clinicopathological characteristics of 1,617 breast cancer patients
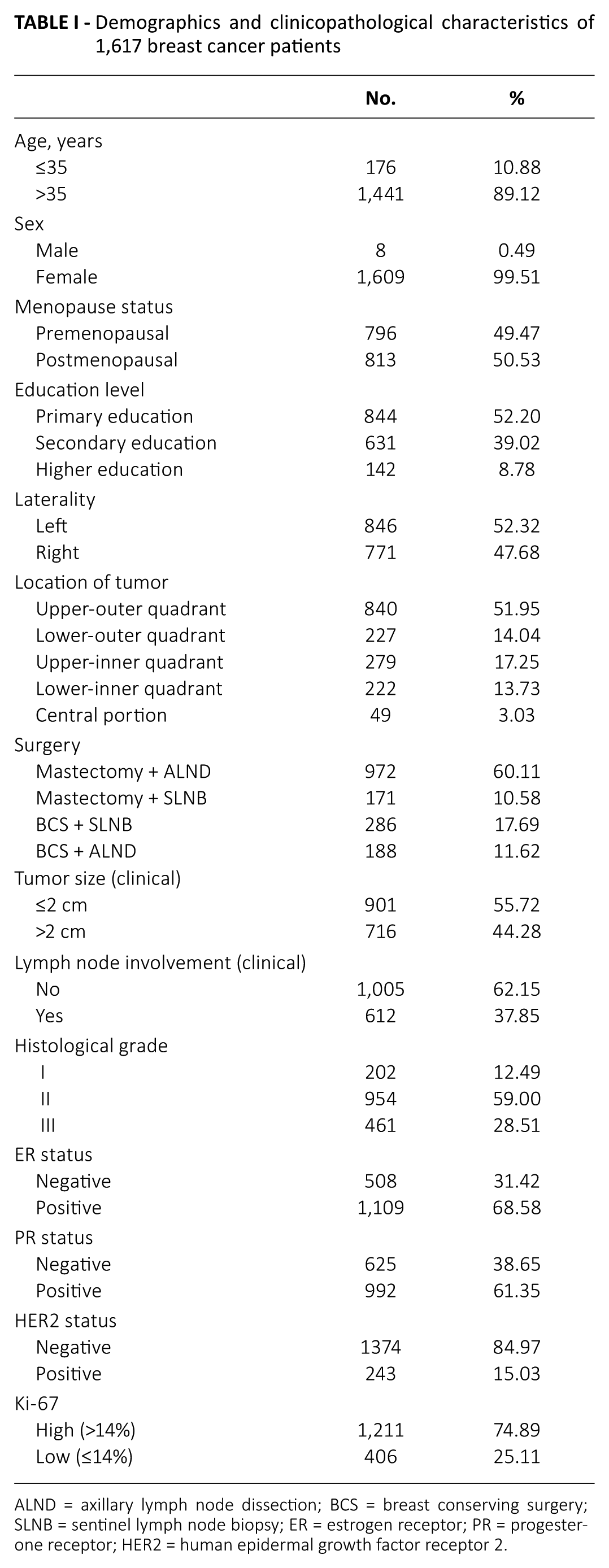
ALND = axillary lymph node dissection; BCS = breast conserving surgery; SLNB = sentinel lymph node biopsy; ER = estrogen receptor; PR = progesterone receptor; HER2 = human epidermal growth factor receptor 2.
The rate of consistent staging for the overall population in our study was 40.63% (n = 657), and those of higher or lower staging assessments by prognostic staging (8th edition AJCC) were 39.76% (n = 643) and 19.61% (n = 317), respectively. The highest consistency based on subgroup analysis were for primary stages IIIC ( 69.92%) and IA (63.83%), then stages IB (31.25%), IIB (29. 67%) and IIA (14.06%), and the lowest was stage IIIA (3.90%). Table II shows the detailed changes in staging according to the primary staging assessment by the 7th edition AJCC staging manual. For overall population, when assessed by the 7th edition AJCC staging manual, the incidences of stages I, II and III were 43.91%, 38.96% and 17.13%, respectively; however, when assessed by the 8th edition staging manual, the rates of stages I, II and III were 49.27%, 24.24% and 28.29%, respectively (p<0.001) (Tab. III).
Staging changes from AJCC anatomic staging manual (7th edition) to prognostic staging manual (8th edition)
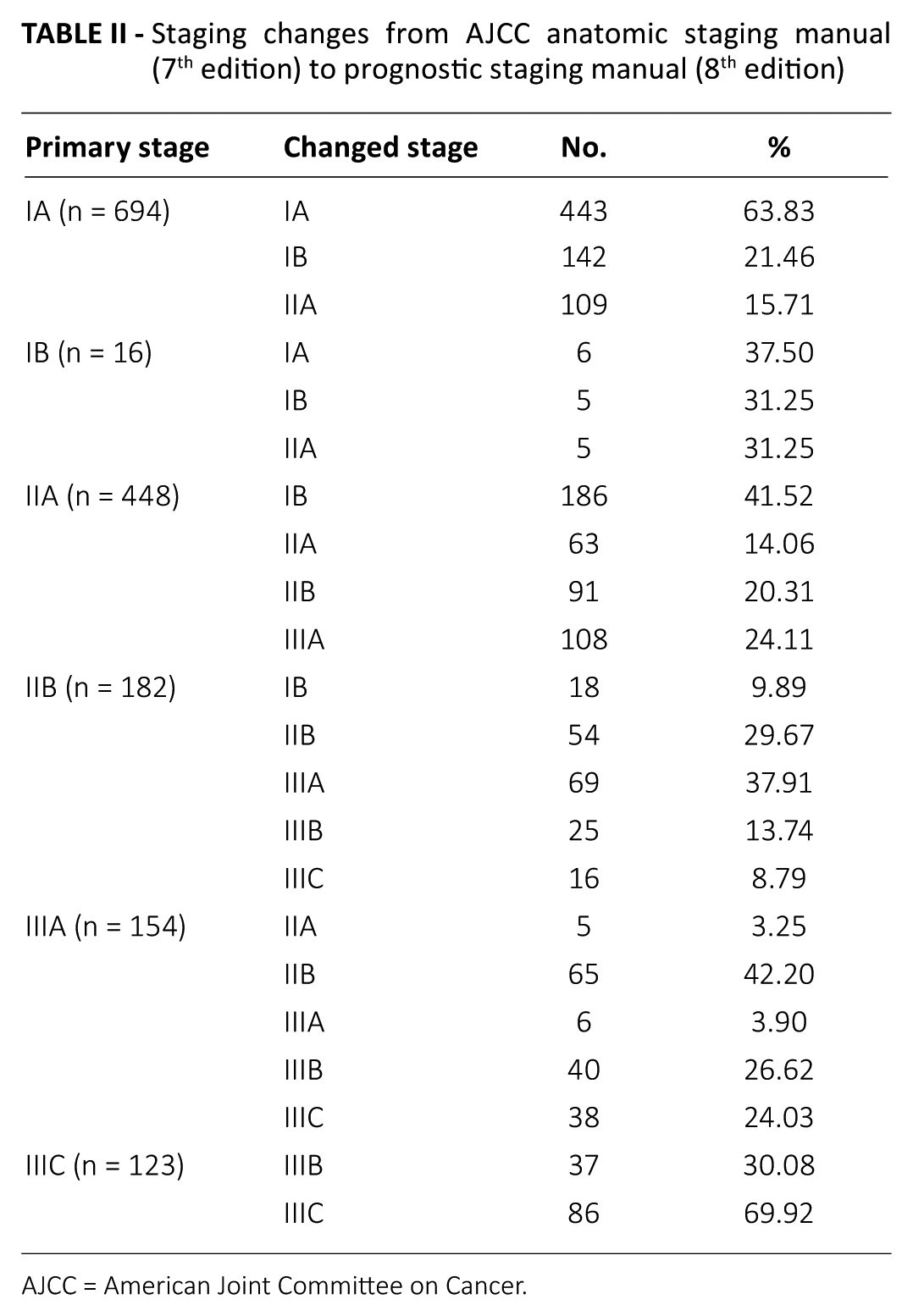
AJCC = American Joint Committee on Cancer.
Distribution of patients according to AJCC anatomic staging manual vs. prognostic staging manual
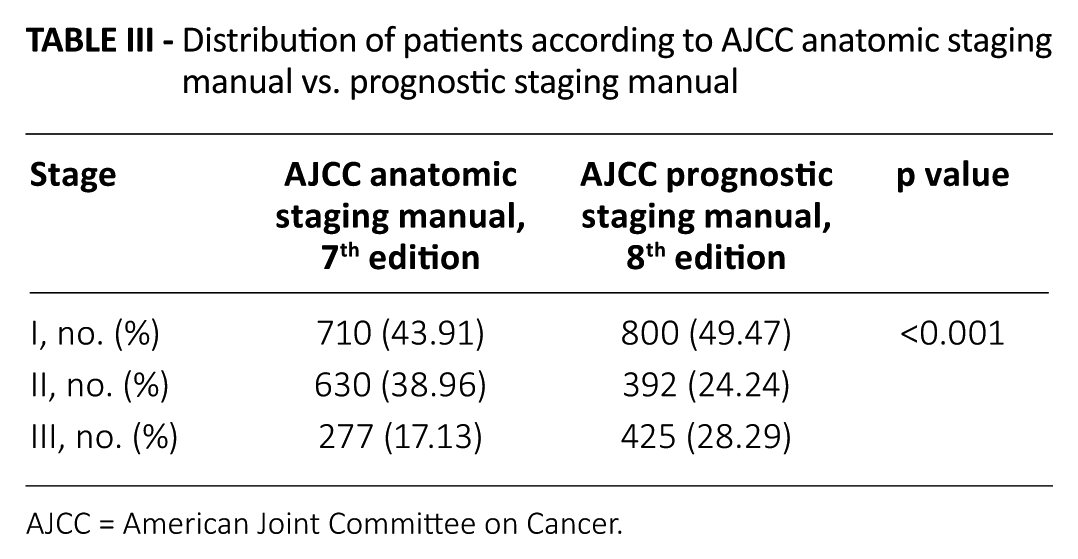
AJCC = American Joint Committee on Cancer.
We found that tumor location, tumor size, axillary lymph node status and Ki-67 index were significantly associated with staging change, while there was no relationship with staging change in terms of age, menstrual status, education level or tumor laterality (Tab. IV). These 4 predictive factors which were confirmed to be associated with staging change by univariate analysis, were then brought into the multivariate logistic regression analysis model, and further analysis found that tumor size >2 cm (OR = 3.263, 95% CI, 2.638-4.036), lymph node involvement (OR = 2.261, 95% CI, 1.830-2.794) and high Ki-67 index (OR = 1.661, 95% CI, 1.343-2.054) were 3 independent predictive factors (Tab. V).
Relationship of clinicopathological factors for staging change
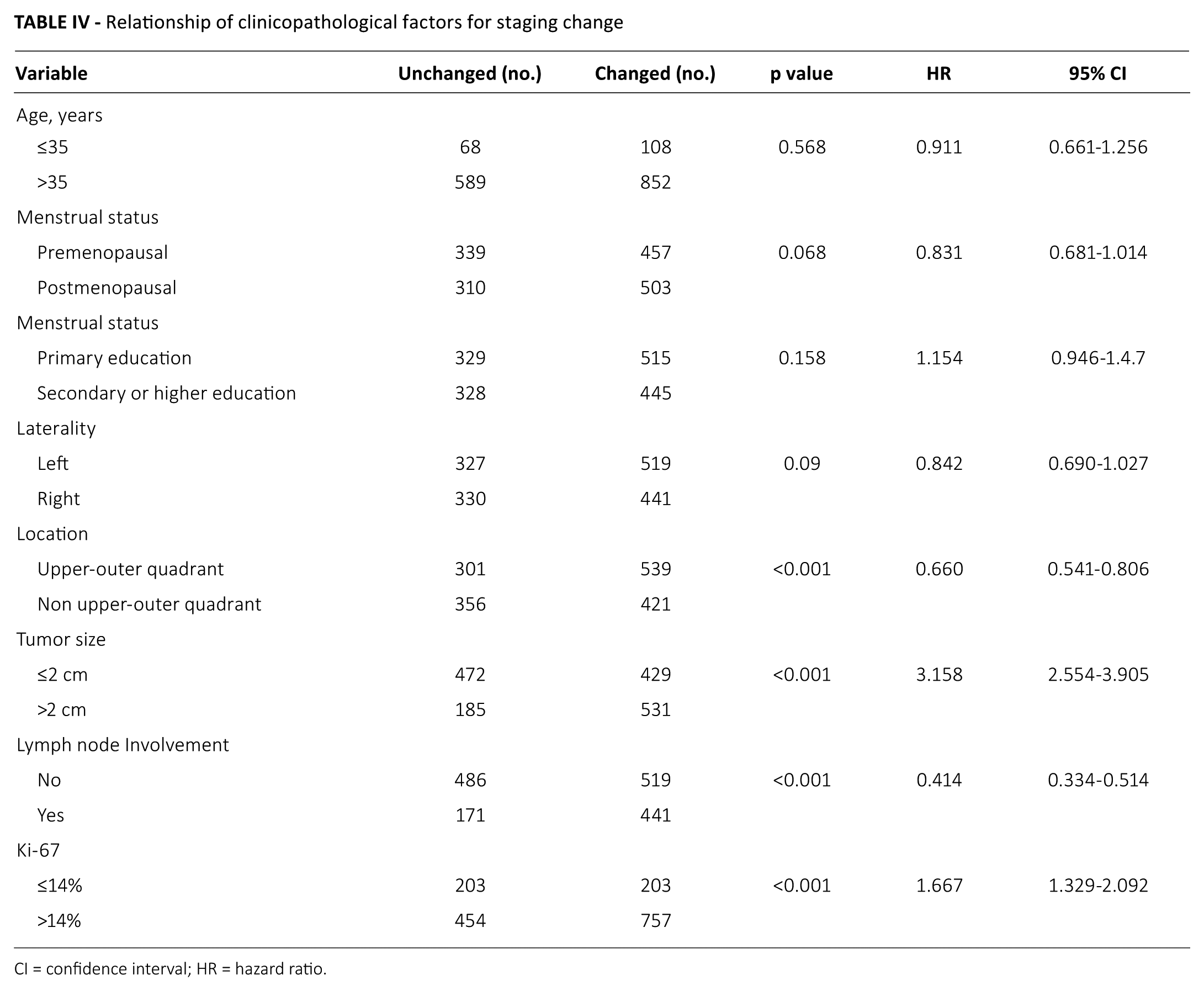
CI = confidence interval; HR = hazard ratio.
Multivariate analysis for staging changes
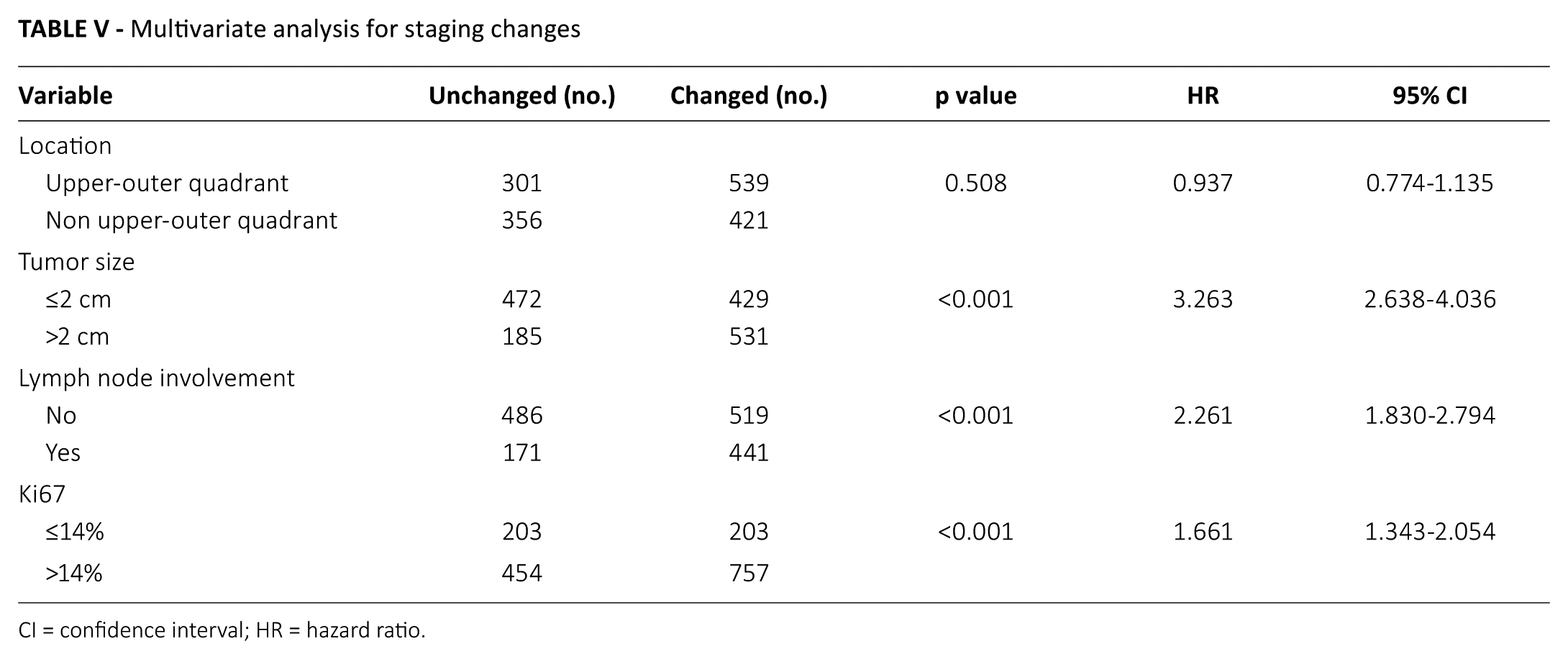
CI = confidence interval; HR = hazard ratio.
Discussion
Although anatomic factors have always been at the foundation of AJCC staging for breast cancer (6), their value for prognosis has encountered a challenge from the emerging importance of prognostic biomarkers such as ER, PR, HER2 and Ki67. Multiple literature reports have certainly addressed the issue of ER, PR and HER2 biomarker expression as prognostic factors for breast cancer outcomes (7-9), and several studies have proposed TNM staging manual revisions to include the additions of ER, PR and HER2 to establish a “B” category TNM staging manual (10-13). Multigene expression assays, including Oncotype DX (14), Mammaprint (15), PAM50 assay (16), Breast-Cancer Index (BCI) assay (17) and Endopredict (18), provide more valuable biologic information for survival prediction. However, they are too complex and expensive to be widely used worldwide, especially in developing countries. In 2017, the AJCC’s Breast Cancer Task Force incorporated the biomarkers in the TNM staging manual, thus estab lishing the newest 8th edition AJCC staging manual, which was also named the prognostic staging manual.
To the best of our knowledge, except for US patients, there has not been any study to investigate disparities in pathological staging for breast cancer between the 7th and 8th edition AJCC staging manuals. Our study is the first to describe the staging changes in low- to middle-income countries (LMCs). Testing important biomarkers, such as ER, PR and HER2, is relatively less costly and readily available to clinicians worldwide, and recent studies have shown that LMCs do assess the status of ER, PR and HER2 in practice (19-23). Especially in China, testing of ER, PR and HER2 has become part of routine clinical practice, which enables physicians to administer personalized systemic therapy, and which enabled our study to compare differences in staging based on the 2 staging manuals.
Our study demonstrated that, when the 2 editions of the AJCC staging manual were used, a change in staging was present in 59.37% (n = 960) of eligible cases, of which 39.76% of cases (n = 643) were upstaged and 19.61% of cases (n = 317) were downstaged. Dr. David J. Winchester, an expert panel member of the AJCC, using data from 238,265 women diagnosed with invasive breast cancer in 2010 who were included in the NCDB, performed an unpublished analysis and found a 41% disparity between the 7th and 8th edition AJCC staging manuals.
The difference between the results of that comparison and our study can be explained by the following: Firstly, our study was a single-institution study with a sample size of 1,617 cases, which was a much smaller sample than that from the NCDB, and this may be the primary cause of the difference. Secondly, the subjects in the 2 studies were obviously different regarding race and age. The patients in our study were Asian, while they were overwhelmingly White or Black in the NCDB sample. The median age of patients on the NCDB was not available because the analysis was unpublished; however, another study of 160,880 patients between 2004 and 2011 from the NCDB registry showed that half of the patients were more than sixty years old (5). We could extrapolate from that that the median age of 238,265 patients would be very much the same as that of the 160,880 patients, which was dramatically older than that in our study (50 years). Thirdly, the rates of hormonal receptor positivity (for ER and PR) were higher in the NCDB analysis, with a rate of 78.68% and 69.44%, respectively, than that in our study, with a rate of 68.58% and 61.35%, respectively.
The consistency rates for each subgroup were different. The highest rate was 69.92% in primary stage IIIC disease and 63.83% in primary stage IA disease. Stage IIIC disease always had an unfavorable biology, including with moderate to high HG, low ER or PR positivity and relative high HER2 expression, so IIIC disease was always categorized as prognostic stage IIIC. In contrast, IA disease always had a favorable biology, including low to moderate HG, high ER and PR expression and low HER2 expression. So stage IA disease was always categorized as prognostic stage IA.
The lowest rate of consistent staging was 3.90% in primary stage IIIA. Primary stage IIIA disease with different biomarkers had a very different prognosis. With favorable biomarkers, the prognosis was as good as that for IIA and IIB; with unfavorable biomarkers, the prognosis was as bad as that for IIIB and IIIC. In our study, the consistency rate for stage IIA was 14.06%, stage IIB was 29.67%. Among patients with anatomic stage IIA disease, 41.52% with favorable biomarkers were downstaged to stage I, and 24.11% of those patients with unfavorable biomarkers were upstaged to stage III. When it came to anatomic IIB disease, 60.44% of patients were upstaged to stage III and 9.89% were downstaged to stage I. This would result in about 70% patients who were either upstaged to prognostic stage III or downstaged to prognostic stage I.
Three factors – namely, clinical tumor size, clinical lymph node status and Ki-67 index – were determined to be independent predictors for staging changes, both in univariate and multivariate analysis. For staging change, clinical tumor size >2 cm had an odds ratio of 3.263 versus tumor size ≤2 cm, lymph node involvement had a odds ratio of 2.261 versus no lymph node involvement and high Ki67 index had a odds ratio of 1.661 versus low Ki-67 index. As discussed above, T1 disease (tumor size ≤2 cm) or N0 disease (negative lymph node) was always associated with favorable biology, which would result in a high probability of prognostic stage I disease; however, disease with tumor size >2 cm or lymph node involvement, most of which patients in our study were diagnosed as having stage II disease, had a higher probability of staging change. Ki-67 is a proliferation index, and high Ki-67 index is inversely associated with favorable biology, such as ER or PR positivity, so disease with a high Ki-67 index is more likely to be upstaged using the prognostic staging system. However, Ki-67 was not considered sufficiently reliable to add to the AJCC staging criteria, because of its known lack of reproducibility, especially between different laboratories, as well as the lack of agreement on an optimal cutoff point.
The clinical significance of our study should be acknowledged. Firstly, in the present study, there is a high discordance rate of staging when the 2 different editions of AJCC staging manual applied, which suggests that since prognostic biomarkers, including ER, PR, HER2 and HG are available in routine clinical practice, we should follow and use the more recent staging manual to select better systemic therapy and, therefore, give better outcomes. Secondly, the ability to predict benefits from or resistance to specific treatments is of major clinical relevance. It is now possible to identify a group of patients who have invasive breast cancer with a prognosis so favorable that they may forgo systemic chemotherapy.
The limitations of this study also should be considered. First, it was a retrospective, single-institution study with a small sample size, which may have decreased its reliability. Next, there were no cases of anatomic stage IIIB disease in our study, and the number of IB cases was not adequate to observe any changes when the 8th edition of the staging manual was applied. Finally, survival data were not available in our study, and therefore, the feasibility and accuracy of the 8th edition (prognostic) staging manual could not be further verified in our study.
Conclusions
This is the first study to describe staging changes by the using AJCC anatomic and prognostic staging manuals for Chinese breast cancer patients. There was a high incidence of staging changes, since prognostic biomarkers, including ER, PR, HER2 and HG, are now available in routine clinical practice, we should follow the more recent staging manual to select better individualized therapy and, therefore, give better outcomes. Of course, multi-institution, prospective studies with large samples and long follow-up timed are needed for validation.
Footnotes
Disclosures
Financial support: None.
Conflict of interest: None of the authors have received any research grants from any company, and all authors declare they have no conflicts of interest.
