Abstract

Dear Editor,
Recently, novel hormonal therapies (NHTs) such as abiraterone acetate and enzalutamide have changed the therapeutic scenario for castration-resistant prostate cancer (CRPC) (1). Unfortunately, except for androgen receptor splice variant-7 (AR-V7) (2), no other biomarker can predict the outcome of patients exposed to NHTs; consequently, the optimal sequence of these therapies remains unknown (3).
The identification of a more easily available surrogate marker for survival or efficacy in patients with CRPC treated with either abiraterone acetate or enzalutamide represents an important advance toward finding an active therapy early, particularly with the increase in alternative available therapeutic options. In this context, prostate-specific antigen (PSA) response rate (RR; defined as an improvement of ≥50% in the PSA concentration from the pretreatment baseline value (4)) may represent an early and rapid biomarker.
Recently, from a literature-based analysis involving the largest number of trials of docetaxel-based first-line chemotherapy for patients with CRPC, we found a correlation between PSA RR and overall survival (OS) (5, 6). On the basis of these findings, it would seem reasonable to extend our previous analysis to NHTs in patients with CRPC.
In this letter, we present preliminary results of an ongoing review performed by our group. We reviewed the literature describing controlled or randomized studies of NHTs for CRPC; the complete methodology of search strategies and the inclusion and exclusion criteria were previously reported (1).
Nine studies were chosen. The characteristics of the studies included and the definitions of PSA response are reported in Table I. A total of 8,713 participants were evaluated (4,934 cases in the NHT group and 3,779 cases in the control group). Among these studies, there were 4 postchemotherapy studies and 5 prechemotherapy studies, 4 studies investigated enzalutamide, 3 studies abiraterone acetate and 2 studies orteronel as experimental drug; while a placebo was the control arm of most of the studies.
Trials included in the analysis
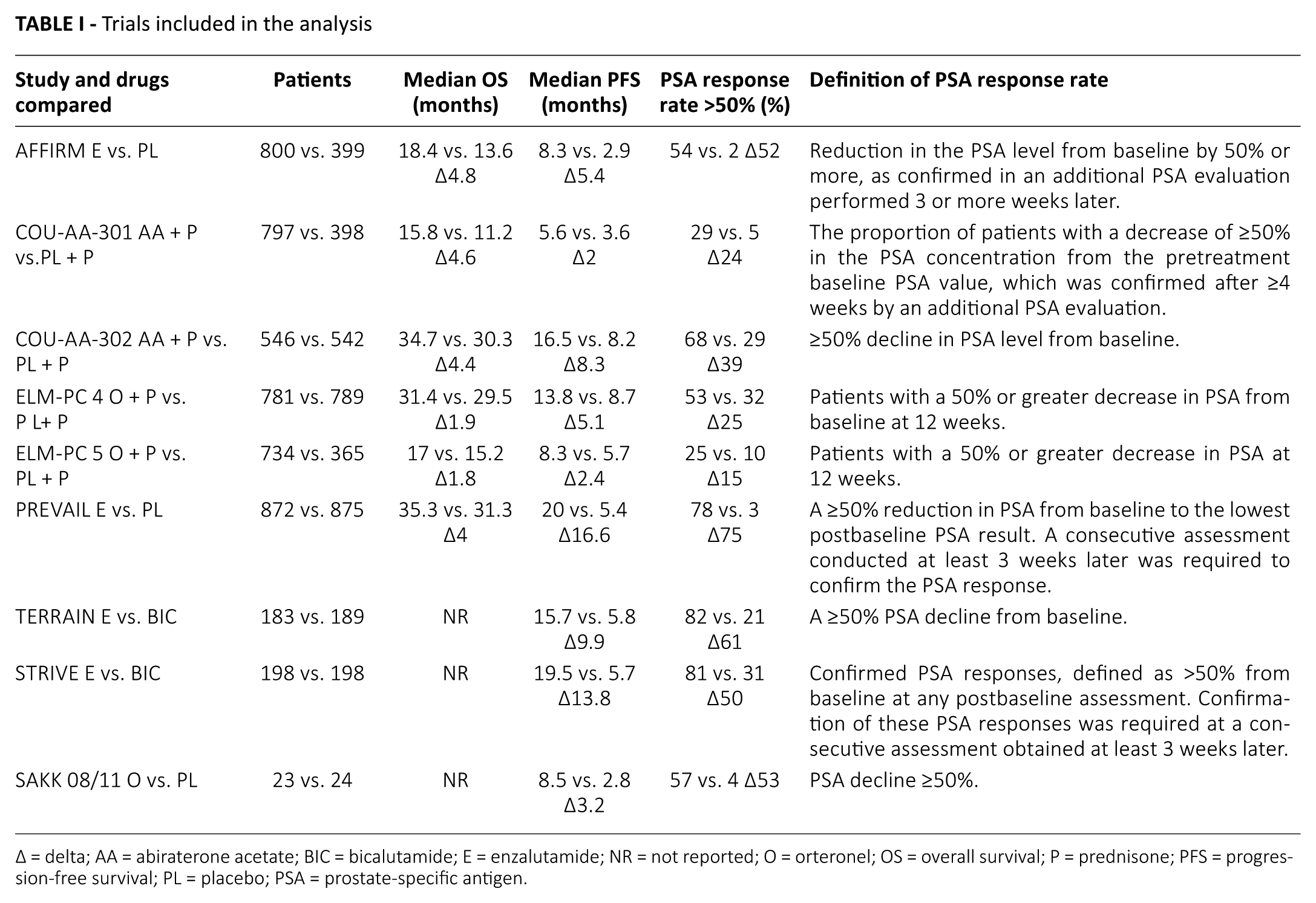
Δ = delta; AA = abiraterone acetate; BIC = bicalutamide; E = enzalutamide; NR = not reported; O = orteronel; OS = overall survival; P = prednisone; PFS = progression-free survival; PL = placebo; PSA = prostate-specific antigen.
The nonparametric Spearman’s rank correlation test (ρ) was used to assess relationships between PSA RR and OS or progression-free survival (PFS). We also derived ΔPSA RR, ΔOS and ΔPFS by subtracting the value in the control arm from the value in the experimental arm. StatIC software, version 2012, was used for the analysis.
Although using the literature rather than patient data analysis does not allow definitive conclusions, our preliminary results showed a good relationship between PSA RR and OS (ρ = 0.71; p = 0.01) and a strong correlation between PSA RR and PFS (ρ = 0.91; p = 0.00). However, with regard to any correlation between ΔPSA RR and ΔOS, ρ decreased to 0.42 and showed a nonsignificant result (p = 0.39), while regarding any correlation between ΔPSA RR and ΔPFS, ρ showed a similar decrease (from 0.91 to 0.71) but the result was significant (p = 0.03).
In conclusion, although the issue of any correlation between PSA RR and survival or efficacy for patients with CRPC treated with NHTs remains unresolved, the present analysis suggests that the role of PSA RR as a surrogate marker for median OS and PFS should be reassessed and should not be ignored for early identification of patients who will not respond to NHTs.
Footnotes
Disclosures
Financial support: No grants or funding have been received for this study.
Conflict of interest: The author has indicated he has no potential conflicts of interest.
