Abstract
Background:
The aim of this study was to elucidate the clinicopathological significance of enhancer of zeste homolog 2 (EZH2) expression in gliomas, including its incidence and prognostic role. In addition, we investigated the concordance between immunohistochemistry and polymerase chain reaction for determining the presence or absence of EZH2 in these tumors.
Methods:
The current meta-analysis included 1,049 gliomas with various WHO tumor grades from 12 eligible studies, which were analyzed for positivity of EZH2 and correlation between EZH2 expression and prognosis. Subgroup analyses were performed based on detection methods and WHO tumor grades.
Results:
We found the estimated positive rate of EZH2 in gliomas to be 0.663 (95% confidence interval [95% CI], 0.549-0.761). There was no difference between immunohistochemistry and polymerase chain reaction in determination of EZH2 positivity (0.706, 95% CI, 0.539-0.831 vs. 0.673, 95% CI, 0.472-0.825). The positive rate of EZH2 increased by increasing WHO tumor grade. EZH2 expression was significantly correlated with worse overall and progression-free survival (hazard ratio [HR] = 2.436, 95% CI, 1.350-4.393, and HR = 4.071, 95% CI, 1.325-12.508, respectively). The overall concordance rate between immunohistochemistry and polymerase chain reaction was 0.885 (95% CI, 0.300-0.993).
Conclusions:
EZH2 positivity was significantly correlated with WHO tumor grade and worse prognosis in gliomas.
Introduction
Up-regulation of enhancer of zeste homology 2 (EZH2) has been reported in several malignant tumors, such as tumors of the head and neck, breast, stomach, liver, lung and prostate (1-6). EZH2, which is a component of polycomb repressive complexes 2, is involved in proliferation, invasion and metastasis of cancer cells (7). Additionally, EZH2 leads to methylation and phosphorylation of STAT3 in carcinogenesis of glioblastoma stem-like cells (8). Recent studies have reported that EZH2 is significantly correlated with worse survival in various cancers, including non-small cell lung cancer and female reproductive system cancer (4, 9). The prognostic roles of EZH2 in gliomas are not fully understood (10-16). EZH2 is overexpressed in isocitrate dehydrogenase (IDH)-wildtype glioblastoma (17); however, the incidence of EZH2 has not been fully elucidated in gliomas of various grades. Methods such as immunohistochemistry (IHC) and polymerase chain reaction (PCR) can be used in the detection of EZH2 protein or mRNA expression. Although PCR-based assays are more sensitive, they are more expensive and time-consuming when compared with IHC. PCR-based assays are not available in every pathology laboratory, whereas IHC methods are widely available. In addition, in daily practice, the IHC method is preferentially used as a screening test. However, the diagnostic accuracy of IHC for determining EZH2 positivity in glioma has not yet been fully investigated.
Detailed information such as the positive rate of EZH2 and the concordance between EZH2 IHC and PCR is not fully available for gliomas (11, 12, 14, 16, 18-24). In this study, we performed a meta-analysis to confirm the incidence and prognostic potential of EZH2 in gliomas. In addition, a concordance analysis between EZH2 IHC and PCR was conducted to examine the relative utility of these methods.
Materials and methods
Published study search and selection criteria
Relevant articles were found by searching the PubMed and MEDLINE databases through December 31, 2016, using the following key words: “glioma” and “enhancer of zeste homolog 2 or EZH2.” Titles and abstracts of all articles found were screened for exclusion, and review articles were screened to find additional eligible studies. Search results were then reviewed and included if the study had been performed in human glioma and if there was information about the positive rate of EZH2, the correlations between EZH2 expression and prognosis, and the concordance between EZH2 IHC and PCR. Articles were excluded if they were case reports or not original articles, or non-English-language publications.
Data extraction
Data from all eligible studies were extracted by 2 independent authors. The extracted data from each of the eligible studies included the following (11, 12, 14, 16, 18-24): the first author’s name, year of publication, study location, antibody clone and manufacturer, antibody dilution ratio, evaluation criteria, number of patients analyzed and WHO tumor grade. For concordance analysis, we extracted all data associated with results of IHC and PCR. Some patients in 2 reports (21, 23) were duplicated with those of other studies (14, 18). Two reports were excluded in estimation of overall EZH2 positive rate (21, 23). For quantitative aggregation of survival results, the correlation between EZH2 expression and overall or disease-free survival rate was analyzed according to the hazard ratio (HR) using one of the following 3 methods: In studies not quoting the HR or its confidence interval (CI), these variables were calculated from the presented data using the HR point estimate, log-rank statistic or its p value, and the O-E statistic (difference between the number of observed and expected events) or its variance. If those data were unavailable, HR was estimated using the total number of events, number of patients at risk in each group, and the log-rank statistic or its p value. Finally, if the only useful data were in the form of graphical representations of survival distributions, survival rates were extracted at specified times to reconstruct the HR estimate and its variance, under the assumption that patients were censored at a constant rate during the time intervals (25). The published survival curves were read independently by 2 authors to reduce reader variability. The HRs were then combined into an overall HR using Peto’s method (26).
Statistical analysis
For meta-analysis, all data were analyzed using the Comprehensive Meta-Analysis software package (Biostat, Englewood, NJ, USA). We investigated the positive rate of EZH2 in gliomas, regardless of detection method. In addition, the correlations between EZH2 expression and worse survival were evaluated. The concordance rates were measured by agreement rates between EZH2 IHC and PCR in 2 eligible studies (14, 18). Because eligible studies used various IHC and PCR methods and evaluation criteria for various populations, a random-effects model was more suitable than a fixed-effects model. Heterogeneity between the studies was checked using the Q and I2 statistics, and presented using p values. Additionally, sensitivity analysis was used to assess the heterogeneity of eligible studies and the impact of each study on the combined effect. Publication bias was assessed using Begg’s funnel plot and Egger’s test. If significant publication bias was found, the fail-safe N and trim and fill tests were performed to confirm the degree of publication bias. The results were considered statistically significant at a p value of <0.05.
Results
Selection and characteristics
One hundred twenty-four reports were identified in the database search. Thirty-nine reports were duplicated articles and were excluded. Eighteen reports were excluded due to insufficiency or lack of information on the positive rate of EZH2 or the correlation between EZH2 and survival rate. In addition, 45 reports were excluded because they reported the results using animals or cell lines (n = 38) or of other diseases (n = 7), and 10 were excluded because they were not original articles (n = 9) or were not in English (n = 1). Finally, 12 reports were included in this meta-analysis (Fig. 1; Tab. I). We ultimately included 1,049 patients from 12 eligible studies. The number of patients from each eligible study ranged from 24 to 128.
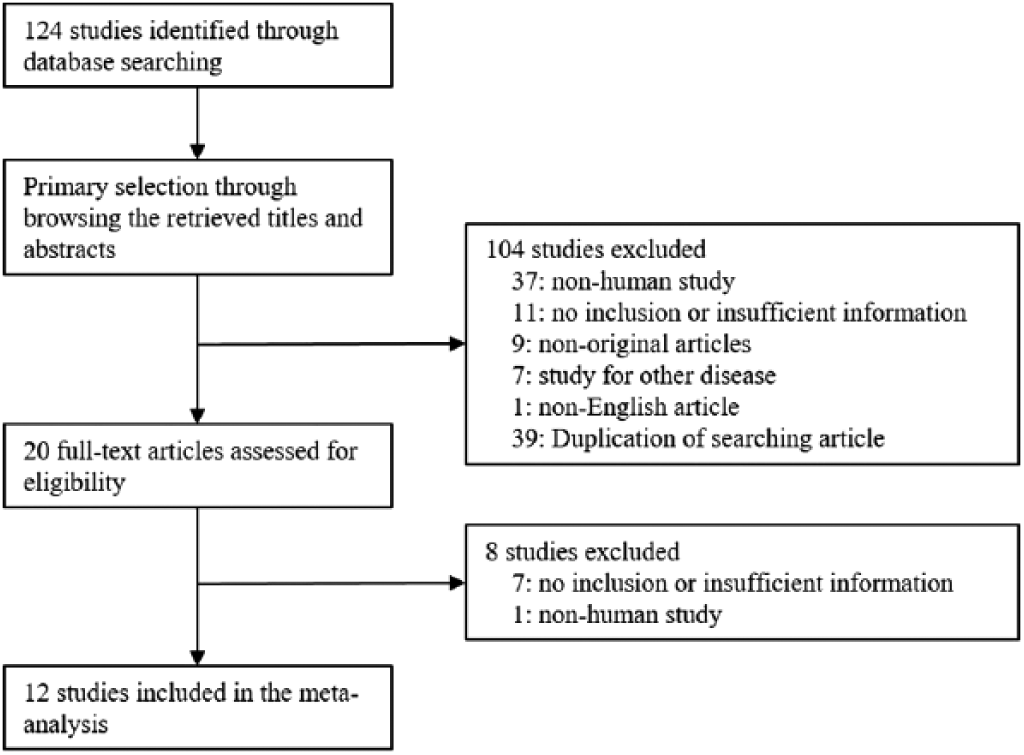
Flow chart of study search and selection methods.
Main characteristics of the eligible studies
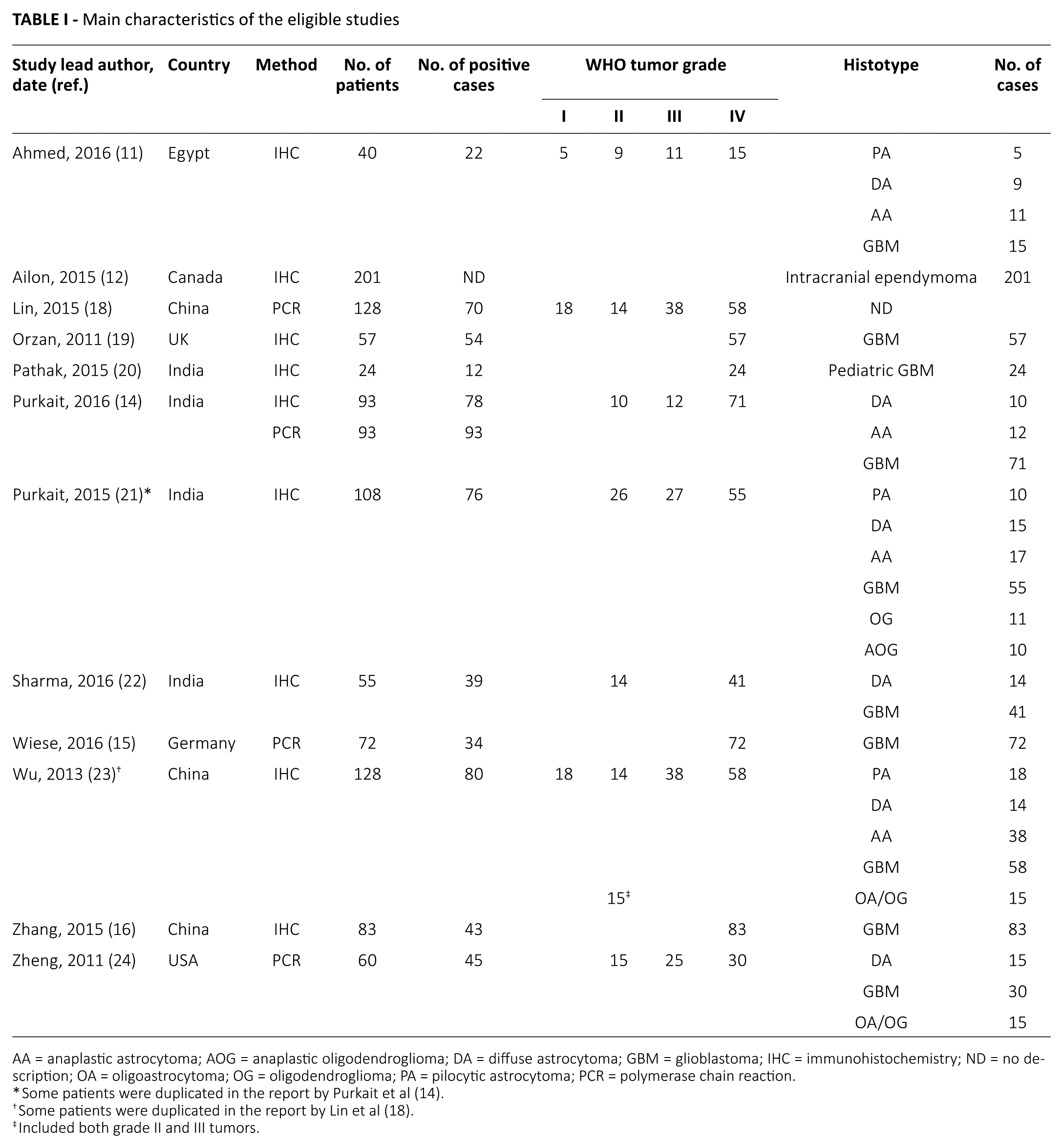
AA = anaplastic astrocytoma; AOG = anaplastic oligodendroglioma; DA = diffuse astrocytoma; GBM = glioblastoma; IHC = immunohistochemistry; ND = no description; OA = oligoastrocytoma; OG = oligodendroglioma; PA = pilocytic astrocytoma; PCR = polymerase chain reaction.
Some patients were duplicated in the report by Purkait et al (14).
Some patients were duplicated in the report by Lin et al (18).
Included both grade II and III tumors.
Meta-analysis
The estimated positive rate of EZH2 in cases overall was 0.663 (95% CI, 0.549-0.761; Tab. II). EZH2 positive rates ranged from 47.2% to 94.7%. Subgroup analyses were performed according to the WHO tumor grades and detection methods. The incidence of EZH2 significantly increased by increasing WHO tumor grade. The EZH2 positive rates of WHO grade I, II, III and IV subgroups were 0.100 (95% CI, 0.018-0.399), 0.381 (95% CI, 0.258-0.520), 0.531 (95% CI, 0.358-0.697) and 0.772 (95% CI, 0.613-0.879), respectively (Tab. II). The subgroup of studies utilizing the IHC method showed slightly higher EZH2 positivity than the PCR subgroup (0.706, 95% CI, 0.539-0.831 vs. 0.673, 95% CI, 0.472-0.825). However, there was no statistically significant difference between the IHC and PCR subgroups in the metaregression test (p = 0.848).
Positive rates of EZH2 in gliomas
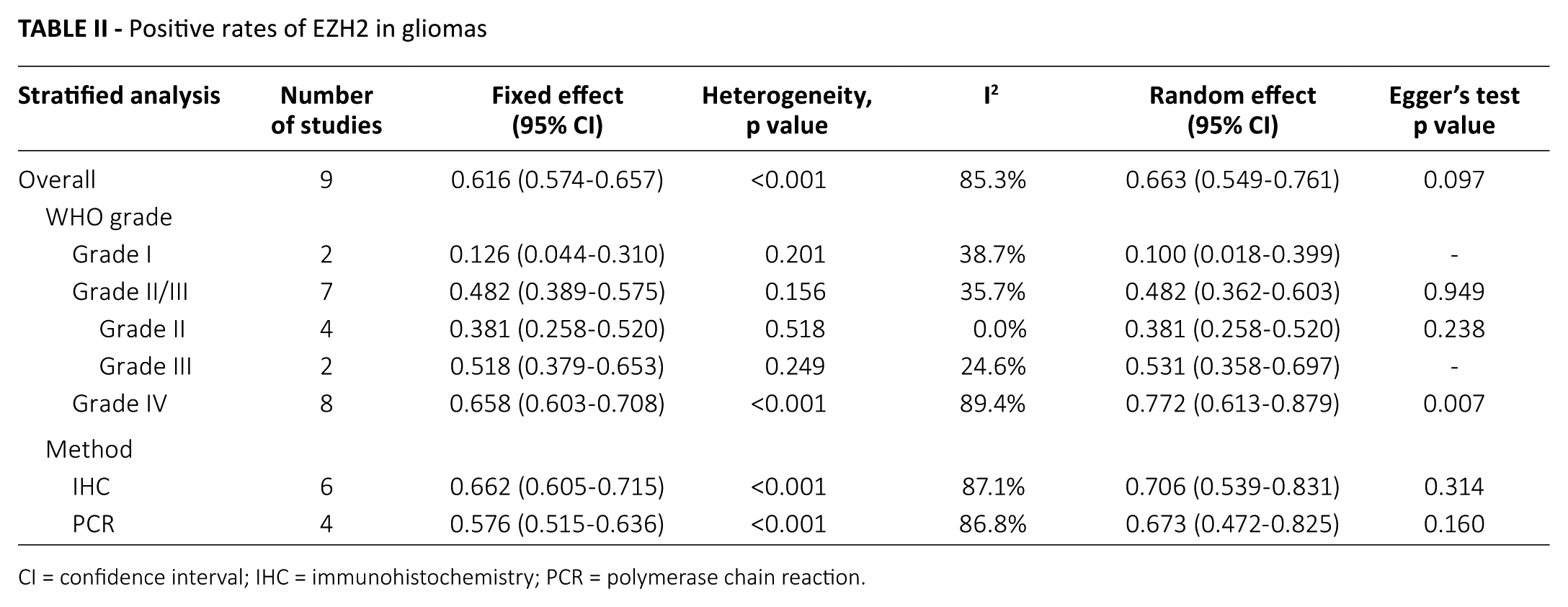
CI = confidence interval; IHC = immunohistochemistry; PCR = polymerase chain reaction.
The WHO grade IV subgroup exhibited a publication bias as determined by the Egger’s test (p = 0.007). To define the degree of publication bias, we conducted the fail-safe N and the trim and fill tests as secondary assessment. The publication bias of the WHO grade IV subgroup was insignificant in both the fail-safe N test and the trim and fill test. In other subgroups, there emerged no significant publication biases.
Next, we investigated the correlation between EZH2 and prognosis and found that EZH2 positivity was significantly correlated with worse overall and progression-free survival rates (HR = 2.436, 95% CI, 1.350-4.393 and HR = 4.071, 95% CI, 1.325-12.508, respectively) (Tab. III). Regarding overall survival, the estimated HRs of IHC and PCR methods were 3.018 (95% CI, 1.616-5.638) and 1.802 (95% CI, 0.592-5.482), respectively. However, a significant correlation was not found between EZH2 and worse prognosis in studies that utilized the PCR method. No publication bias emerged from analysis of the correlation between EZH2 and prognosis.
Correlation between EZH2 and survival in gliomas
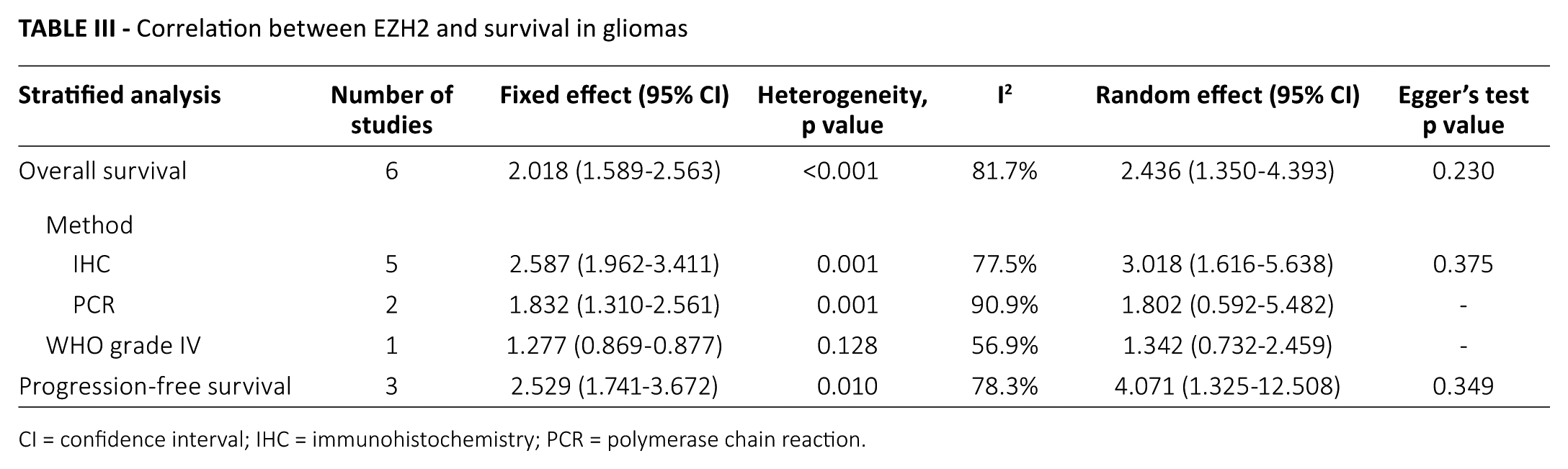
CI = confidence interval; IHC = immunohistochemistry; PCR = polymerase chain reaction.
Concordance analysis
To evaluate the diagnostic role of EZH2 IHC, concordance analysis between EZH2 IHC and PCR was performed. Overall concordance rate between IHC and PCR was 0.885 (95% CI, 0.300-0.993) (Tab. IV). Concordance rates between IHC and PCR of EZH2 positive and negative cases were 0.961 (95% CI, 0.552-0.998, p = 0.036 in the metaregression test) and 0.639 (95% CI, 0.001-1.000, p = 0.886 in the metaregression test), respectively; however, concordance rates between EZH2 positive and negative cases in the metaregression test were not significantly different (p = 0.456).
Concordance analysis of EZH2 immunohistochemistry and polymerase chain reaction in gliomas
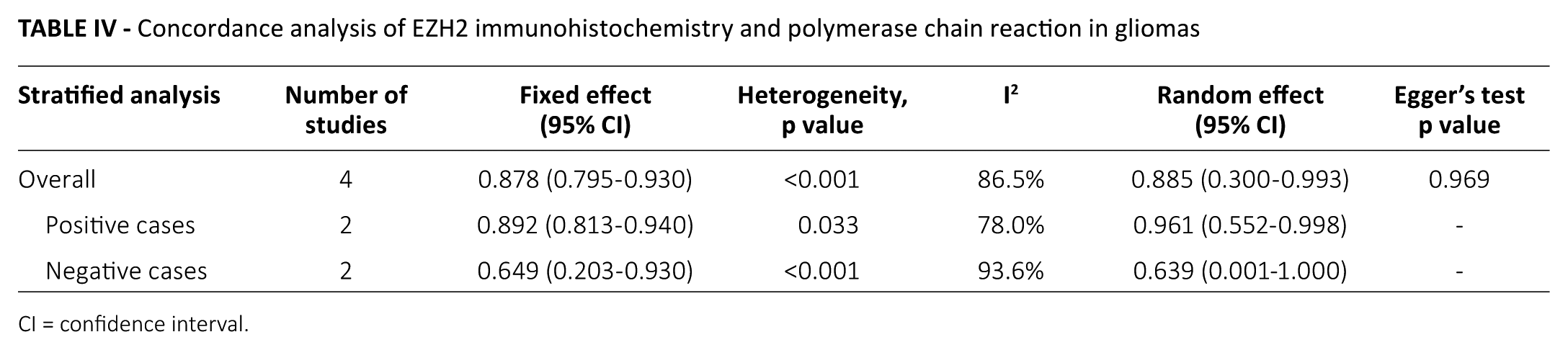
CI = confidence interval.
Discussion
This study investigated EZH2 positivity in human gliomas of WHO grades I-IV from eligible studies. The incidence of EZH2 ranged from 47.2% to 94.7% among eligible studies. The inclusion of tumors with variable grades or detection methods could be responsible for the variability in EZH2 incidence. In a subgroup analysis based on the WHO tumor grade, the positive rates of EZH2 significantly increased by increasing WHO tumor grade (p<0.001 in the metaregression test). The EZH2 positive rates of WHO grade I, II, III and IV gliomas were 0.100, 0.381, 0.531 and 0.772, respectively. Given that many of the cases we included were glioblastoma (WHO grade IV) cases, the overall positive rate of EZH2 is understandably high (0.663, 95% CI, 0.549-0.761). In previous studies, loss-of-function mutations of EZH2 appeared rare in IDH-wildtype pediatric glioblastoma (27, 28). However, in our meta-analysis, the positive rate of EZH2 in pediatric glioblastoma was 0.500 (95% CI, 0.310-0.690), albeit this was lower than the overall positive rate of EZH2 in glioblastoma. More investigations are needed to further delineate EZH2 expression in specific types of gliomas.
Each eligible study used particular detection methods and evaluation criteria for EZH2 positivity. We performed a subgroup analysis based on EZH2 antibody manufacturers and found no significant difference between manufacturers (p = 0.714 in the metaregression test; data not shown). However, subgroup analysis of evaluation criteria of EZH2 IHC could not be performed because of insufficient information. Therefore, the impact of different cutoffs on the EZH2 IHC positivity was unclear.
In our subgroup analysis based on detection methods, the difference of EZH2 positivity between IHC and PCR methods was insignificant (p = 0.848 in the metaregression test). Molecular tests, such as PCR, possess disadvantages such as high costs, the need for multiple steps, skill and time compared with IHC. Therefore, we performed a concordance analysis to confirm the diagnostic accuracy of EZH2 IHC and determined an overall concordance rate between IHC and PCR of 0.885 (95% CI, 0.300-0.993). To evaluate the cause of discordance, an additional subgroup analysis was performed to examine EZH2 IHC positivity versus negativity. The concordance rate of EZH2 positive cases was higher than that of EZH2 negative cases (0.961, 95% CI, 0.552-0.998 vs. 0.639, 95% CI, 0.001-1.000). In other words, in EZH2 IHC, false negative cases were frequently found. Discordance between EZH2 IHC and PCR may result from the discrepancy between EZH2 mRNA expression and EZH2 protein expression after translation. In addition, individual evaluation criteria for EZH2 IHC and PCR may have influenced concordance rates between detection methods. Prospective studies with standardized evaluation criteria for EZH2 IHC and PCR would more precisely elucidate the difference in results obtained with these 2 methods.
Our result that EZH2 was correlated with worse survival in glioma is consistent with a previously published meta-analysis (10). However, EZH2 can also exist in lower grade gliomas (WHO I to III); therefore, the diagnostic role of EZH2 may be limited in gliomas. Interestingly, in glioblastomas, there was no significant correlation between EZH2 positivity and overall survival rate (HR = 1.342, 95% CI, 0.732-2.459). A higher EZH2 positive rate may affect the correlation between EZH2 positivity and survival. Therefore, assessment of EZH2 positivity may be more useful for predicting prognosis in lower grade gliomas. However, in the lower grade gliomas included in this study, subgroup analysis for the correlation between EZH2 positivity and survival could not be performed due to insufficient information.
In the current meta-analysis, cases with EZH2 positivity by IHC endured a worse overall survival rate (HR = 3.018, 95% CI, 1.616-5.638). However, in the 2 eligible studies employing the PCR method, we did not find a significant correlation between EZH2 presence and worse overall survival (HR = 1.802, 95% CI, 0.592-5.482), as in a previous meta-analysis (10). There was a discrepancy between 2 eligible studies regarding the correlation with survival (15, 18). The HRs of each of the eligible studies were 1.020 (95% CI, 0.630-1.651) and 3.174 (95% CI, 1.991-5.061), respectively. The criteria for EZH2 positivity in PCR may influence the correlation between EZH2 expression and survival. Further cumulative studies will be required to elucidate the optimal method for predicting prognosis using EZH2.
There were some limitations to the current meta-analysis. First, due to the nature of the eligible studies, we included more glioblastoma cases (WHO grade IV) than lower grade glial tumors. The impact of this composition of glial tumors on EZH2 positivity and the subsequent correlation between EZH2 and survival rate is unclear. Currently, an insufficient number of studies exist in the literature to address this issue. Second, while the present study was the first meta-analysis of the concordance rate between detection methods in gliomas, a thorough diagnostic test accuracy review could not be conducted due to the small number of studies included. Third, the optimal cutoff value for EZH2 IHC or PCR positivity has not been elucidated in gliomas. The positive rates of EZH2 and concordance rates between IHC and PCR could differ by cutoff value. Unfortunately, a subgroup analysis to clarify this could not be performed due to insufficient information in the eligible studies. Fourth, to evaluate the impact of ependymomas on overall estimated HR, sensitivity analysis was performed. The HRs of overall and progression-free survival were slightly increased in sensitivity analysis (HR = 2.813, 95% CI, 1.366-5.791 and HR = 6.404, 95% CI, 3.073-13.345, respectively). However, to confirm the impact of EZH2 on the prognostic role of ependymomas, further cumulative evaluations will be needed. Fifth, although our results were largely consistent with those of a previous meta-analysis, the detailed results, such as the value of HR, differed slightly (overall survival: HR = 2.18, 95% CI, 1.25-3.80 vs. HR = 2.436, 95% CI, 1.350-4.393; and progression-free survival: HR = 2.23, 95% CI, 1.56-3.19 vs. HR = 4.071, 95% CI, 1.325-12.508) (10). To avoid selection bias introduced by different follow-up periods, extraction after a 60-month follow-up period was conducted in the present study. However, information on follow-up periods was not shown in the previous meta-analysis.
In conclusion, EZH2 expression was significantly correlated with worse overall and disease-free survival in gliomas. In addition, EZH2 positivity was higher in advanced WHO grades of glioma. However, further studies will require detailed evaluation criteria to determine the optimal EZH2 detection method.
Footnotes
Disclosures
Financial support: None.
Conflict of interest: The authors declare that they have no conflict of interest.
