Abstract
In this systematic review, we evaluated the value of serum concentrations of neuron-specific enolase (NSE) in patients with prostate cancer (PCa) in order to clarify the possible role of NSE in the diagnosis, management, treatment and monitoring of PCa. A comprehensive search of the recent literature was conducted to find relevant data on the role of NSE in PCa. Two hundred and eighty-two records were revealed, and 19 articles including 1,772 patients with PCa (either confirmed or suspected) were selected. After reviewing the articles, the major result was that elevated serum NSE appears to correlate with prognosis in advanced PCa, particularly in patients with progressive and metastatic castration-resistant PCa. Based on the existing literature, the role of serum NSE in PCa patients should be further evaluated.
Introduction
Prostate cancer (PCa) is the most frequent tumor in men and a major leading cause of cancer-related deaths in developed countries (1). Prostate tumors range from slowly growing lesions to aggressive tumors, which metastasize rapidly (2). The subtype with neuroendocrine (NE) features represents a highly aggressive variant of PCa that is increasingly recognized in the clinic (3). Although rare as a primary diagnosis, the NE phenotype often emerges during progression of the disease, especially in the castration-resistant stage (3, 4).
NE cells are naturally present in the prostate gland. Their functional roles are unclear, but they are probably involved in the regulation, secretion and differentiation of the prostate (5). In the normal prostate, NE cells are rare and scattered throughout the epithelium, providing trophic signals to surrounding prostate cells (5). The origin of cancerous NE cells in PCa is controversial. It has been proposed that they can arise from stem cells like normal NE cells (6, 7). But, an increasing body of evidence suggests that adenocarcinoma cells can undergo a transdifferentiation process and acquire themselves NE features (3). This form of plasticity is associated with a lower androgen receptor signaling (8). Indeed, NE tumor cells do not express androgen receptors: they grow in an androgen-deprived environment and might stimulate the proliferation of the PCa cells through various signaling modalities, including long-range, endocrine mechanisms and short-mediated signaling, both paracrine or direct cell–cell contact (9, 10). They also increase PCa aggressiveness through the inhibition of apoptosis (11, 12) and promotion of neovascularization (13).
NE cells are recognized by specific immunostaining with antibodies against peptides included in their secretory granules, such as neuron-specific enolase (NSE), chromogranin A (CgA) and synaptophysin (3, 9).
NSE is a cytoplasmic enzyme, the γ homodimer of enolase, and was first found in extracts of brain tissue (14). NSE is a potentially useful marker of neuronal damage in several neurological disorders, as well as a marker for tumors of NE origin, especially those arising in the lung (14). Measurement of NSE levels in patients with these diseases can provide information about the tumor burden, the patient’s prognosis and the response to treatment (14).
In the setting of PCa, an extremely small proportion of patients present at diagnosis with an extensive NE phenotype (15). NE features mostly arise in the setting of concurrent or previously diagnosed conventional adenocarcinoma with progression of PCa to castration-resistant disease (15). Indeed, there is a consensus in the literature confirming that the transdifferentiation from an epithelial-like phenotype to the more aggressive NE-like phenotype, also known as neuroendocrine differentiation (NED), is a mechanism of resistance to standard treatments, that occurs under selective pressure exerted primarily by therapies that lead to androgen level decrease or androgen receptor blockade (3, 15). High levels of serum NE markers, along with rapidly progressive castration-resistant prostate cancer (CRPC), low prostate-specific antigen (PSA) and visceral metastasis, are distinctive features of the NED phenotype, and they may facilitate the recognition of this uncommon and under-recognized subtype of PCa (4). The identification of NED is very important for its implications for treatment and prognosis.
In this systematic review, we assessed the value of serum concentrations of NSE in patients with various stages of PCa to clarify the role of NSE in the diagnosis, management, treatment and monitoring of PCa.
Methods
Search strategy
A comprehensive literature search of the electronic PubMed/MEDLINE, Google Scholar and Embase databases was conducted to find relevant data on the role of NSE in PCa. We used a search algorithm that was based on a combination of the terms: (a) “prostate” or “prostatic” and (b) “NSE” or “neuron-specific enolase.” The literature search was performed from year 2000 to June 10, 2016, to include the most updated data. To expand our search, references of the retrieved articles were also screened for additional data.
Study selection
Original articles investigating the usefulness of NSE in PCa patients were eligible for inclusion. The exclusion criteria were (a) articles not within the field of interest of this review; (b) review articles, letters or editorials; (c) case reports or case series (less than 10 patients included); (d) articles not in the English language. Three researchers (B.M., M.P. and E.R.) independently reviewed the titles and abstracts of the retrieved records, applying the inclusion and exclusion criteria mentioned above to determine their eligibility for inclusion. Disagreements were resolved in a consensus meeting.
Methodological quality of included studies
The quality of the studies was assessed using the modified Hayden list (16). This included an accurate description of the study population, the stage of disease, the rationale for the size of the cohort and blinding at the time of interpretation. Studies were assessed according to the Hayden list to highlight sources of bias. The studies were graded from 0 to 6 according to their adherence to this list. Studies that adhered to at least 4 of these 6 requirements to assess study bias were included.
Data extraction
For each study included, information was collected concerning basic data (authors, year of publication, country of origin, type of study), methods (NSE serum assay method, number of patients included and median age) and results (such as level of serum NSE in the diagnosis, prognosis and treatment response assessment; correlation with PSA, CgA, stage or grade; correlation with NSE expression on immunohistochemistry and hormone resistance).
Results
Literature search
The comprehensive literature search of PubMed/MEDLINE, Google Scholar and Embase databases revealed 282 records. Reviewing titles and abstracts, 204 records were excluded because they were not within the field of interest of this manuscript or had been published before the year 2000; 7 records were excluded as review articles, letters or editorials; 22 articles were excluded as case reports and 30 as they were not in the English language. Regarding their quality, all of the included articles adhered to the requirements of the modified Hayden list (16). The full texts of the remaining articles were retrieved. No additional studies were found in screening the references of those selected articles. Finally, 19 articles including 1,772 patients with confirmed or suspicious PCa were selected for the systematic review (17-35) (Fig. 1).
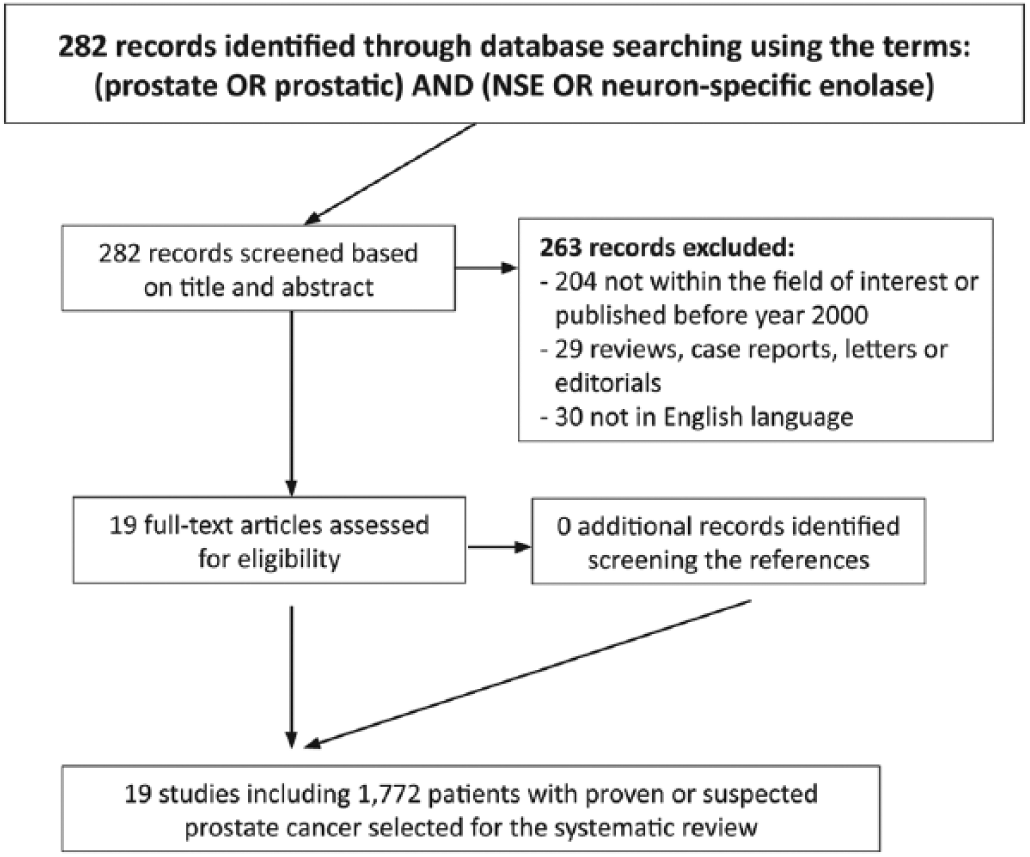
Study selection process for systematic review of the literature on the role of neuron-specific enolase (NSE) in prostate cancer.
Qualitative analysis (systematic review)
The characteristics of the selected studies are presented in Table I. The main findings are summarized in Table II and reported below. Most of the articles included were for retrospective studies, and only 5 studies had a prospective design (24, 26, 28, 29, 33).
Characteristics of the included studies on NSE in prostate cancer
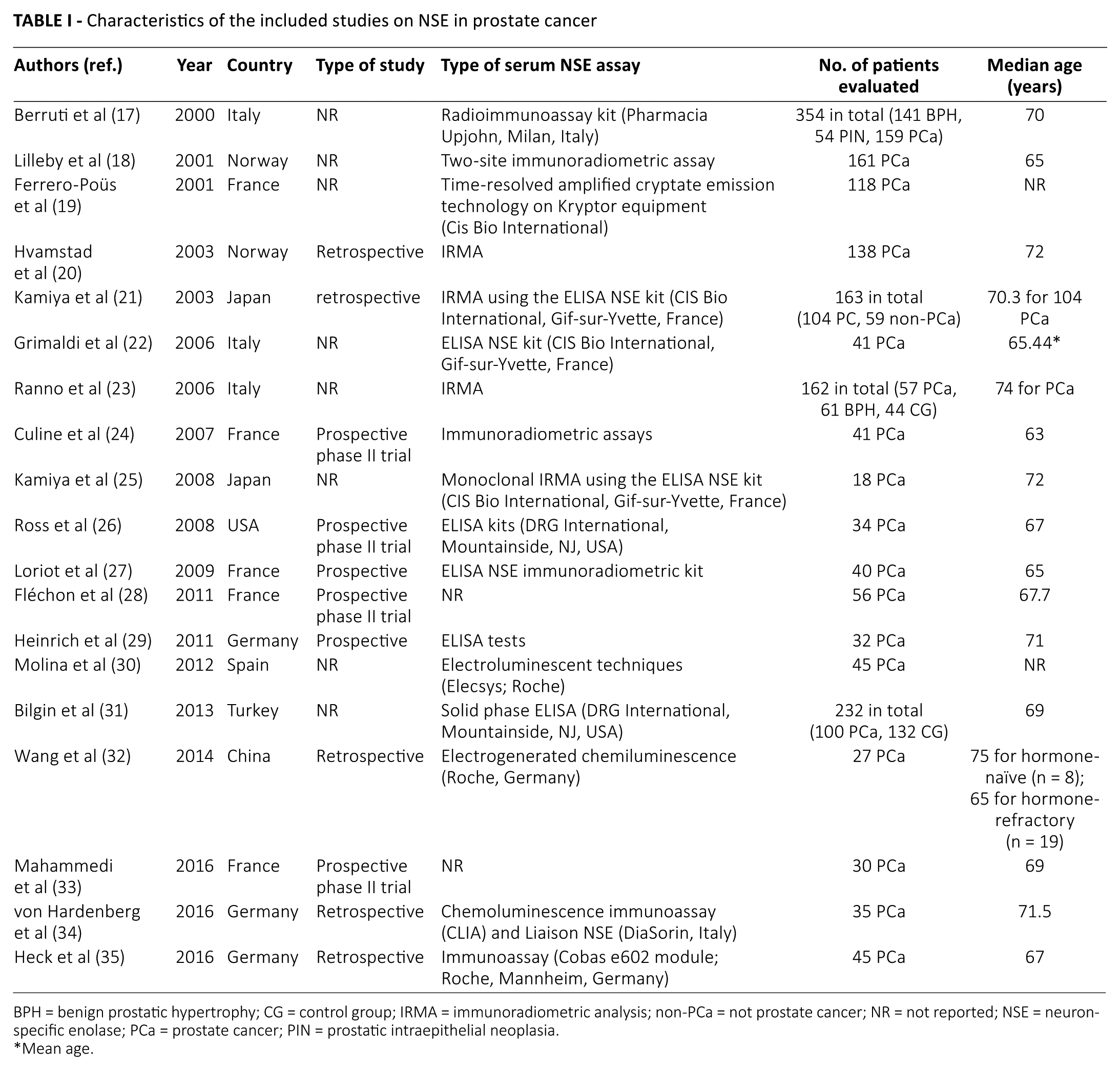
BPH = benign prostatic hypertrophy; CG = control group; IRMA = immunoradiometric analysis; non-PCa = not prostate cancer; NR = not reported; NSE = neuron-specific enolase; PCa = prostate cancer; PIN = prostatic intraepithelial neoplasia.
Mean age.
Main findings of selected studies on the role of serum NSE in proven or suspected PCa patients
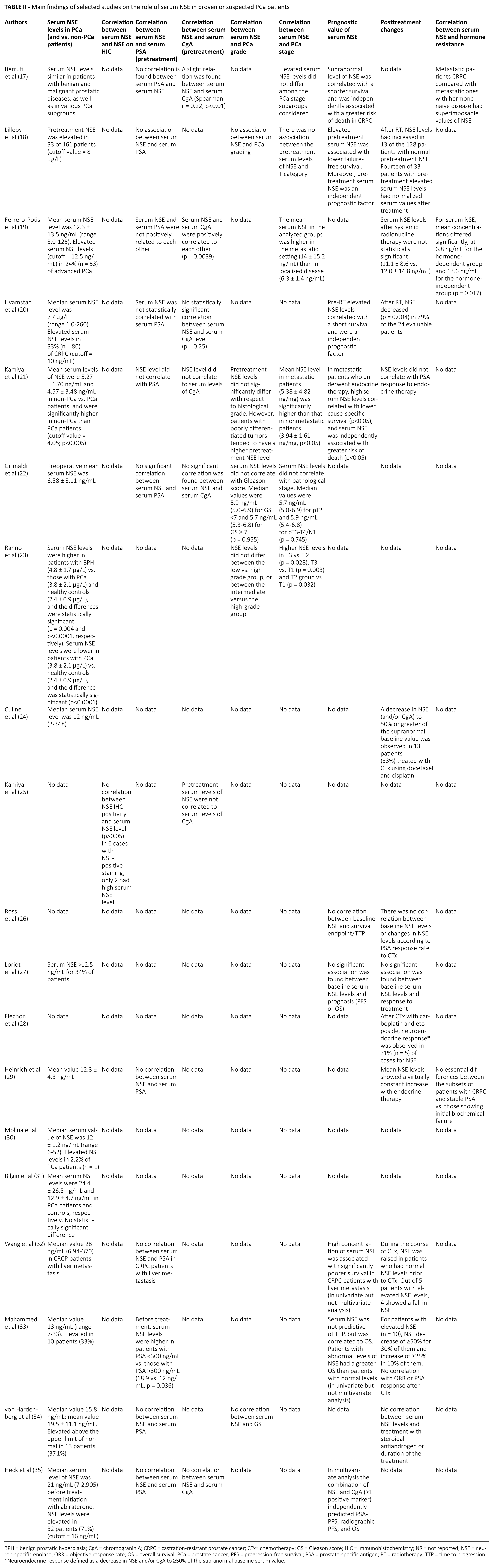
BPH = benign prostatic hyperplasia; CgA = chromogranin A; CRPC = castration-resistant prostate cancer; CTx= chemotherapy; GS = Gleason score; HIC = immunohistochemistry; NR = not reported; NSE = neuron-specific enolase; ORR = objective response rate; OS = overall survival; PCa = prostate cancer; PFS = progression-free survival; PSA = prostate-specific antigen; RT = radiotherapy; TTP = time to progression.
Neuroendocrine response defined as a decrease in NSE and/or CgA to ≥50% of the supranormal baseline serum value.
Serum NSE levels in PCa (and comparison with non-PCa patients)
Twelve articles assessed the possible role of NSE in the diagnosis of various stages of PCa (17-21, 23, 27, 30, 31, 33-35).
There is a discrepancy in the literature regarding the difference in serum NSE levels among PCa and non-PCa patients. Berruti et al, in evaluating serum NSE in benign prostatic hypertrophy (BPH), prostatic intraepithelial neoplasia (PIN) and PCa, found that NSE levels were similar in patients with benign and malignant disease (17). Similar findings were recently reported by Bilgin et al, who described low sensitivity (48%) and specificity (36%) for serum NSE in PCa diagnosis (31).
Kamiya et al reported that the mean levels of serum NSE in non-PCa patients were significantly higher than those in PCa patients (p<0.005) (21). On the other hand, Ranno et al found that the mean difference in NSE levels was 1.05 µg/L (p = 0.004) comparing PCa vs. BPH, 1.38 µg/L (p<0.0001) comparing PCa vs. controls and 2.4 µg/L (p<0.0001) comparing BPH vs. controls (23).
Lilleby et al found that pretreatment serum NSE was elevated in almost 20.5% of patients with localized PCa (18), whereas Molina et al reported elevated serum NSE levels in only 2% of PCa patients (30). This rate increased to 24% and 33%, when considering just patients with advanced PCa (19) or symptomatic CRPC (20), respectively. A rate of elevated serum NSE above 30% in progressive CRPC was also found by 4 other studies (27, 33-35). In particular, Heck et al (35) found elevated serum NSE in 71% of metastatic CRCP previously treated with chemotherapy (CTx).
Correlation between serum NSE and its immunohistochemical expression
Only 1 study, by Kamiya et al (25), reported on the connection between the serum levels of NSE with its immunohistochemical expression in tissue specimens from PCa patients. A statistically significant correlation between them was not found.
Correlation between serum NSE and pretreatment PSA
Just 1 (33) out of 11 studies (17-22, 29, 32, 33-35) evaluating the correlation between pretreatment serum level of PSA and serum NSE found a relation, with patients with lower PSA (<300 ng/mL) showing higher NSE levels than men with PSA >300 ng/mL (18.9 vs. 12 ng/mL, respectively; p = 0.036).
Correlation between serum NSE and pretreatment CgA
Seven articles explored the possible correlation between serum levels of NSE and pretreatment serum levels of CgA in patients diagnosed with PCa (17, 19-21, 22, 25, 35), but the results are not consistent.
On the one side, Berruti et al found a statistically significant correlation between NSE and CgA (p<0.01) (17). A similar conclusion was reported by the group of Ferrero-Poüs et al, who described a positive relation between serum NSE and CgA (19). On the other side, the remaining studies did not show a significant correlation between the 2 markers (20-22, 25, 35).
Correlation between serum NSE and Gleason score
In our research, 5 articles assessed the correlation between serum NSE levels and Gleason score. None of them found a correlation between these items (18, 21-23, 34), even though Kamiya et al did notice that patients with poorly differentiated tumors tended to have a higher pretreatment NSE level than those with well-differentiated or moderately differentiated tumors (21).
Correlation between serum NSE and stage
A total of 6 studies in our review investigated the correlation between serum NSE levels and the clinical and/or pathological stage of disease (17-19, 21-23). Three of them showed an association between the pretreatment levels of NSE and T category (19, 21, 23). In particular, Ranno et al found higher levels of NSE levels in their T2 group compared with patients in the T1 stage (p = 0.032) and with those in the T3 stage (p = 0.028). In addition, the differences of NSE levels proved to be strongly significant when comparing stages T1 and T3, with the higher NSE levels in the T3 group (p = 0.003) (23). According to Ferrero-Poüs et al, mean NSE serum level was higher in the metastatic setting than in localized disease (19). Similarly, Kamiya et al found that the mean NSE serum level in metastatic PCa patients was significantly higher than that in nonmetastatic patients (p<0.05) (21). Conversely, 3 other studies did not find any association between the pretreatment serum levels of NSE and T category (17, 18, 22).
Prognostic value of NSE
In our analysis, a total of 9 studies explored the prognostic value of pretreatment serum NSE in PCa (17, 18, 20, 21, 26, 27, 32, 33, 35). Berruti et al (17) found that elevated levels of serum NSE were associated with poor prognosis in PCa patients, especially in CRPC. Likewise, Lilleby et al reported that pretreatment NSE was independently associated with failure-free survival in localized PCa (18). Kamiya et al found that cause-specific survival was significantly poorer in metastatic patients with a high NSE level (p<0.05), and serum NSE was an independent variable associated with a greater risk of death (p<0.05) (21). Also, Wang et al found an association between high concentration of serum NSE and significantly poorer survival in CRPC patients with liver metastasis (32). A similar result was found by the group of Hvamstad et al, who reported a shorter survival for CRPC patients having a high level of pretreatment NSE (20). In contrast, Mahammedi et al (33) reported higher overall survival in progressing CRPC patients with an abnormal level of serum NSE compared with patients with normal values (p = 0.027). Moreover, they found no correlation with time to progression. A correlation of abnormal concentration of serum NSE with overall survival and progression-free survival (PSA or radiographic) was also found by Heck et al (35) in metastatic CRPC patients, previously treated with CTx. However, this last study considered a combination of NSE and CgA, and it is not possible to really evaluate the role of NSE (35).
Finally, both Ross et al (26) and Loriot et al (27) did not find any correlation between serum NSE levels and PCa prognosis.
Posttreatment changes of serum levels of NSE
We found 12 studies that analyzed the changes of serum levels of NSE related to different kinds of therapy such as endocrine therapy, radiotherapy (RT), CTx or radionuclide therapy (18-21, 24, 26-29, 32-34).
Most of them, apart from 4, reported a change during or after the treatment. In fact, in the assessment of Ferrero-Poüs et al, levels of NSE after systemic radionuclide therapy were not increased significantly (19). Also in the paper by Ross et al, there was no correlation between baseline NSE levels or changes in NSE levels according to PSA response rate, albeit the treatment in this case was CTx (26). Furthermore, no significant association was found between baseline serum NSE and response to treatment according to Loriot et al after treating CRPC patients with CTx (27). No correlation between antiandrogen treatment and duration in CRCP patients was found by von Hardenberg and colleagues (34).
Eight studies found a correlation between treatment and NSE changes. Wang et al, during the course of CTx, found that of the 7 patients followed up serially, NSE was raised in the 2 patients who had normal NSE prior to CTx. Out of 5 patients with elevated NSE levels, 4 showed a fall in NSE. In 2 of these, it became normal. It is interesting to note that all 4 patients with a decrease of NE markers during the course of CTx showed a radiologically assessed partial response (n = 3) or stable disease (n = 1) (32), although we cannot know how much this NE response rate was related to NSE or CgA. Also, Culine et al have reported the results of CTx in patients with CRPC and high serum NE markers (elevated serum NSE) and found that, of 41 patients treated, 13 patients (33%) displayed a serum NE marker response (>50% decrease of NSE) (24). In the analysis performed by Flechon et al, an NE response was observed in 31% of cases for NSE after CTx (28). Finally, Mahammedi et al (33), analyzing CRPC patients with elevated NSE at baseline showed an NSE decrease (defined as ≤50%) and an increase (defined as ≥25%) in 30% and 10% of them, respectively, after CTx. However, they did not find any correlation with objective response rate or PSA response.
In an RT setting, data from a study by Lilleby et al showed that NSE levels had increased in 13 out of 128 patients with normal pretreatment NSE, and that 14 out of 33 patients with pretreatment elevated NSE levels had normalized serum values after RT (18). Also, Hvamstad et al analyzed the behavior of NSE after RT treatment and found a statistically significant NSE decrease (p = 0.004) in 79% of the 24 evaluable patients (20).
With regard to endocrine therapy, Heinrich et al noted that mean NSE values had a virtually constant increase with endocrine therapy (29). In contrast, Kamiya et al found that NSE levels did not differ significantly between nonmetastatic PCa and metastatic PCa with regard to PSA response to endocrine therapy (21).
Correlation between NSE serum levels and hormone resistance
Three studies assessed the correlation between NSE and hormonal status in PCa (17, 19, 29). Berruti et al found no significant differences between serum NSE levels of metastatic CRPC versus metastatic hormone-naïve patients (17). Heinrich et al reached the same conclusion in their analysis, where there were no essential differences between the subgroups of patients with CRPC and stable PSA or those showing initial biochemical failure (29). On the other hand, Ferrero-Poüs et al pointed out that, for NSE, the mean concentrations differed significantly, at 6.8 ng/mL for the hormone-dependent group and 13.6 ng/mL for the hormone-independent group (p = 0.017) (19).
Discussion
NED is a feature of invasive carcinomas in which a transdifferentiation from an epithelial-like phenotype to the more aggressive NE-like phenotype occurs (15). In the setting of PCa, NED is a highly heterogeneous phenomenon. A consensus group recently proposed a formal classification of prostatic NE tumors ranging from purely NE cancers (such as small cell carcinomas) to conventional adenocarcinomas that exhibits focal or diffuse NED (9, 36). Furthermore, the onset of NED varies widely across PCa patient groups: a relatively small subset of PCa patients show NE features at the time of diagnosis (before any therapy) (37), while others develop an NED after treatment (10).
In clinical practice, a de novo small cell carcinoma is diagnosed in a small proportion of patients, but NED features frequently arise in the context of an adenocarcinoma as a mechanism of resistance to standard treatments, particularly to endocrine therapy (3, 15); indeed, it is associated with progression to castration-resistant disease, generally representing the lethal late stage in prostate tumorigenesis (15).
Given its pathogenesis, NED is often suspected and looked for by clinicians in patients with advanced, multitreated disease no longer responsive to standard therapy. This stage of the disease is difficult to monitor with PSA measurement, and NED markers can provide useful additional information (17). The most important and commonly used markers of NED are CgA and NSE (10). Measurement of serum markers has the advantage of being more representative and an objective indicator of NED in PCa compared with immunohistochemical ones, as it corresponds to the entire primary tumor and its associated metastases (5, 10). On the other hand, while obtained more easily and less invasively, serum markers only provide an indirect measure of NED. In addition, blood levels of some NED markers can also increase in non-NE settings (10).
Several studies have evaluated the role of NSE and CgA serum levels as NED markers in PCa. CgA is reported to be the most reliable serum marker in PCa (17, 37). The aim of our systematic review was to investigate the usefulness of serum NSE in PCa in order to provide more evidence-based data in this setting.
Overall, based on recent literature findings, NSE serum levels appear to be inaccurate in discriminating between PCa and benign prostatic disease. Two studies did not find any differences (17, 31), whereas another 2, showing differences in serum NSE between PCa and benign tissue, used mixed stages of tumors (21, 23). However, there seems to be a greater number of patients, from 30% to 37%, having elevated levels of serum NSE at diagnosis of progressive CRPC (20, 27, 33, 34). This result confirms the elevation of NSE in the bloodstream when the cancer becomes no longer responsive to hormonal treatment.
In our analysis, just 1 study had investigated the correlation between NSE serum levels and its immunohistochemical expression in PCa specimens, and it found no statistical difference (25). Measurement of serum NSE is very easy and less invasive, but it has the disadvantage of being only indirectly correlated to the disease, as prostatic non-endocrine cells can express it as well (10). However, since NED is often a focal process (18), histopathological markers can be less accurate than serum markers. Currently, it is not possible to draw any definitive conclusions, and more studies are necessary to clarify this point.
The lack of correlation between serum NSE and PSA may indirectly support the information provided by NED markers that is complementary to PSA (17). Each marker may have clinical and prognostic significance in contexts that are very different from each other. Thus, evaluating both markers might help cover a wider range of situations that can arise during the clinical course of the disease. The only study establishing a significant correlation confirmed an inverse relation with higher NSE levels in the presence of lower PSA (33). Globally, these data suggest that NSE- and PSA-producing cells develop relatively independently of each other during the progression to hormone-refractory PCa, since the development of NED cells seems not to be androgen-dependent, in contrast to the growth of PSA-producing malignant cells (20).
As CgA and NSE can be considered NE markers, some similarities might be expected in their serum concentrations during the development of PCa (19); however, they may not follow the same pattern. In fact, several studies did not find a correlation between these two NE markers (20-22, 25). Measurement of NSE may be clinically unstable (23), and the differences between serum NSE and CgA may be explained by the focality of NSE-producing cells, different mechanism of secretion or storage and lack of standard measurement kits (18). Globally speaking, CgA appears to reflect the NE phenotype of PCa more accurately than NSE (17, 38). In contrast to CgA, NSE was expected to be discharged in peripheral blood by cytolysis. Thus, a high concentration of serum NSE may reflect rapid turnover of mitosis and cytolysis of cancer cells, indicating higher malignant potential (21). Moreover, it is unclear which marker is more useful during each stage of the development of the disease. It seems that circulating levels of CgA may be more useful in patients with metastatic CRPC (17, 18, 23, 38, 39), whereas NSE is useful for localized disease (18, 38). By contrast, Grimaldi et al (22) did not find NSE as useful tumor marker for NED in organ-confined PCa, and Hvmstad et al (20) and Kamiya et al (21) reported the utility of serum NSE for metastatic patients. However, serum levels of markers may be higher in patients with advanced disease, especially with metastases (30), and thus they may be more valuable in this setting. Based on the existing literature, the role and the utility of these markers in diagnosis, prognosis and monitoring of PCa with NED features still remains unclear.
No correlation between serum NSE and Gleason score was found in the articles included in this systematic review (18, 21-23). Just 1 study found that patients with poorly differentiated tumors tended to have a higher pretreatment NSE serum level (21). Regarding the correlation between NSE and PCa stage, only 3 studies demonstrated that increased PCa stage was associated with higher NSE serum levels (19, 21, 23), especially in metastatic versus localized PCa (19, 21). This information further confirms that NED is a feature of advanced PCa. In this setting, the role of NED is better established also at the prognostic level. In fact, pretreatment serum level of NSE may predict survival of metastatic PCa patients treated with endocrine therapy (21). Nevertheless, the prognostic accuracy of pretreatment serum NSE is clearer in patients with CRPC, for whom elevated NSE correlates with a shorter survival (17, 20, 32, 35). However, 3 prospective studies did not find any correlation with prognosis in metastatic disease (26) or CRPC (27, 33). In fact, a clear correlation with castration status has not yet been well established (17, 19, 29).
The use of NSE as a marker of treatment response in PCa is still controversial as well (18-21, 27-29). An NSE response after CTx was found in CRPC patients (24, 28, 32, 33). Three of these studies were phase II trials, which means that they were conducted prospectively and in a controlled setting (24, 28, 33). However, any correlation that was found by Ross et al was in a phase II trial including metastatic patients (26), and by Loriot et al, in a prospective trial including CRPC patients (27). For RT (18-20) or endocrine therapy (21, 29, 34), the data are varied, but NSE level response seems more useful in the context of advanced disease, even if more prospective studies are needed. These data further confirm concerns about the accuracy of NE markers as surrogate parameters for monitoring treatment efficiency (24).
Some limitations of this systematic review should be underlined, such as the heterogeneity of the articles included (mostly related to the various characteristics of PCa patients) and the low number of prospective studies available. In fact, we did not perform a meta-analysis, because the recent literature shows discordant findings, lack of homogeneity in patient selection and different laboratory assays in serum NSE determination, thus making a comparison among different studies challenging. Other limitations of this study may have resulted from potential sources of publication bias, considering that our research was limited to the English language and did not include gray literature.
One last thing to consider among the limitations of this work is the variability of methodologies used to measure serum NSE, as reported in Table I. This discordance may have played a role in the difficulty of finding consistent results. To date, there is no specific consensus on which methodology is to be preferred in determining serum NSE, so agreement about an international reference standard is needed.
Conclusions
Until now, results have been conflicting, and the most meaningful data seem to be with regard to the elevation of serum NSE in advanced PCa, especially in cases of progressive metastatic CRPC after hormonal treatment. NSE might have a prognostic value, but any definitive conclusion is currently not possible. Based on the recent available literature, the significance of NSE in PCa patients should be further evaluated to clarify its role in the diagnosis, prognosis and monitoring of NE disease. This systematic review is of interest since it clarifies some of the gaps in the evidence and gives suggestions for new research. To apply this biomarker in clinical management, appropriate validations in a vast majority of authoritative prospective cohort studies are necessary.
As stated, this study had several limitations, but it provides a solid foundation for better exploiting this biomarker in clinical practice to identify diagnostic and therapeutic choices in an increasingly personalized medical setting.
In addition, we chose to conduct a systematic review, as, unlike a narrative review, it was thus possible to extract conclusions from the literature data, collecting and critically analyzing multiple research studies. Systematic reviews are key to the practice of evidence-based medicine, and a review of existing studies is often quicker and cheaper than embarking on a new study.
Footnotes
Disclosures
Financial support: No grants or funding have been received for this study.
Conflict of interest: The authors declare that they have no conflicts of interest.
