Abstract
Purpose
Extracorporeal circulation circuits used in cardiopulmonary bypass surgeries are increasingly being coated with polymer materials to reduce the thrombogenicity of extracorporeal devices. However, a haemoconcentrator, which corrects haematocrit and electrolyte imbalances, is not coated with polymers. In this study, we sought to assess the filtration performance of polymer-coated haemoconcentrators in order to obtain insight into their prospects for use in clinical applications.
Methods
In vitro experiments were performed to evaluate the water pressure and flow properties of polymer-coated haemoconcentrators by comparing 3 polymer-coated haemoconcentrators with 3 non-coated haemoconcentrators. The cross-sectional surfaces of both types of haemoconcentrators were observed using a scanning electron microscope (SEM).
Results
The slopes of the regression lines for estimating the filtrated fluid flow as a function of the transmembrane pressure were 6.286 ± 0.320 for polymer-coated haemoconcentrators and 3.712 ± 0.170 for non-coated haemoconcentrators. These slopes were found to be significantly different and indicate that the filtration velocity is enhanced in polymer-coated haemoconcentrators over that in non-coated haemoconcentrators. However, the hollow fibre damage observed by SEM was not shown to contribute to higher filtration flow in the polymer-coated haemoconcentrator. Taking these results into consideration, we hypothesise that a polymer coating makes a foreign surface on a hollow fibre slippery, owing to the hydrophobicity of the polymer, thereby enhancing the velocity of the filtration.
Conclusions
The results of this preliminary investigation suggest that a polymer coating can enhance the filtration performance of a haemoconcentrator and that polymer-coated haemoconcentrators might be useful in clinical applications.
Keywords
Introduction
In an extracorporeal circulation circuit used in a cardiopulmonary bypass surgery, a haemoconcentrator, which is composed of a hollow fibre consisting of plastic materials such as polysulfone or polyethersulfone, filtrates blood components smaller than albumin, thus concentrating blood (1). Haemoconcentrators have also been used in perfusion techniques such as dilutional ultrafiltration, modified ultrafiltration, and zero-balance ultrafiltration for haemocatharsis in cardiopulmonary bypass surgeries (2–7). To perform these roles, a haemoconcentrator is required to be in contact blood for long periods of time and thus must be biocompatible.
Currently, extracorporeal circulation circuits used in cardiopulmonary bypass surgeries are coated with biocompatible polymers to prevent the adverse effects induced by contact with blood components and proteins, and many reports have been published on the thrombogenicity reduction of polymer-coated devices (8–10).
Several polymer coating agents, such as poly-2-methoxyethylacrylate (9–11), 2-methacryloyloxyethyl phosphoryl choline (12–16), and glycocalyx-mimetic dextran-modified polyvinyl amine (17), have been observed to be compatible with the human body in various clinical procedures. There have been many reports on the use of polymer coating agents for reduction of platelet activities (9, 10), cell adhesion (12), haemolysis (13), protein adhesion (15), and inflammatory reaction (10, 12, 18), suggesting that polymer coating agents have favourable biological compatibility. In the field of cardiopulmonary bypass surgery, extracorporeal circuit devices have been coated with biocompatible polymers to reduce platelet activation and fibrinogen deposition (9, 10). However, haemoconcentrators are typically not coated with a polymer, and reports on polymer-coated haemoconcentrators have not been published.
In this study, we sought to clarify the filtration performance of a polymer-coated haemoconcentrator for the purpose of assessing its prospects for use in clinical applications.
Methods
Experiments were performed to assess the pressure and flow properties of polymer-coated and non-coated haemoconcentrators. Six haemoconcentrators (Hemocrystal, MERA, Tokyo, Japan) with the same lot number were employed. Three were coated with a polymer coating agent composed of a copolymer consisting of acrylic monomers.
A polymer-coated haemoconcentrator was configured with polyvinyl chloride tubes, 2 polyvinyl chloride chambers, a plastic reservoir, a clamper, an electric occluder, 2 manometers, and a roller pump in an experimental circuit (Fig. 1). Two litres of water were primed in the reservoir and circulated to the experimental circuit at flow rates of 100, 200, and 300 mL/min by clamping the filtration line. The water pressure was measured at each flow rate using manometers installed at the inlet and outlet chambers of the haemoconcentrator. Subsequently, the flow was fixed at 200 mL/min, and after unclamping the filtration line, the flow of the filtrated fluid and the pressure at the inlet and outlet chambers were measured in various situations, such as during occlusion of the tube of the return line by 0%, 50%, and 100%. The same experiments were performed with each of the other 2 polymer-coated haemoconcentrators and the 3 non-coated haemoconcentrators.
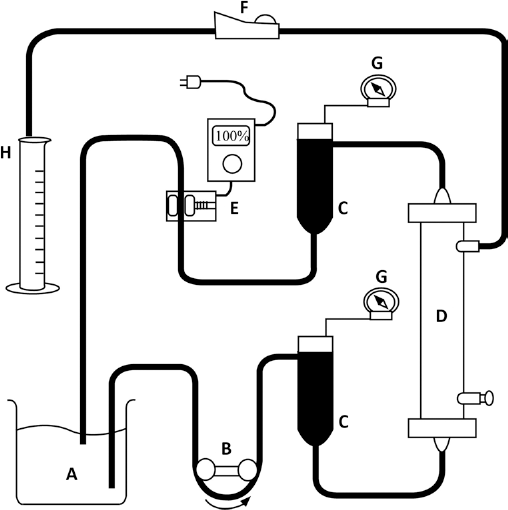
Experimental circuit consisting of (A) a plastic reservoir and polyvinyl chloride tubes, (B) a roller pump, (C) two polyvinyl chloride chambers, and (D) a haemoconcentrator. (E) An electric occluder was attached to the return line, and (F) a clamper was attached to the filtration line. (G) Manometers were connected to both of the chambers. After measuring the volume of the filtrated fluid, pooled water in (H) the measuring cylinder was returned to the plastic reservoir.
After each flow condition was established, at least 5 minutes were allowed to elapse to achieve a stable pressure. Subsequently, the pressure was measured continuously for 10 seconds, the maximum and minimum values were recorded, and the average of these pressures was used to avoid errors caused by the effect of a pressure pulsation wave induced by the roller pump. The volume of the filtrated fluid was measured over a period of 30 seconds using a measuring cylinder. The measurements were performed 3 times in each experiment to minimize the effects of measurement errors. The 3 measured volumes were averaged, and this value was employed in the analysis. The flow of the filtrated fluid was converted to a volume per minute from the measured volume.
All statistical significance results cited in this paper were obtained through Student's t-test. The significance level was set at a = 0.05.
The cross-sectional surfaces of the polymer-coated haemoconcentrators and non-coated haemoconcentrators were observed using a scanning electron microscope (SEM) to determine whether the hollow fibre in the polymer-coated haemoconcentrator had been destroyed.
Results
The loss in pressure in the polymer-coated and non-coated haemoconcentrators was compared at various circuit flow levels (Fig. 2). The loss in pressure was calculated by subtracting the outlet chamber pressure from the inlet chamber pressure. The loss in pressure at a circuit flow of 100 mL/min was 7.83 ± 0.24 (mean ± standard deviation, SD) mmHg for the non-coated haemoconcentrators and 9.16 ± 0.24 mmHg for the polymer-coated haemoconcentrators, which indicates a significant difference (p<0.05). At circuit flows of 200 mL/min and 300 mL/min, the losses in pressure were 16.5 ± 1.08 and 27 ± 2.27 mmHg for the non-coated haemoconcentrators and 17.8 ± 0.62 and 30.2 ± 0.85 mmHg for the polymer-coated haemoconcentrators, which do not indicate significant differences between the non-coated and polymer-coated haemoconcentrators.
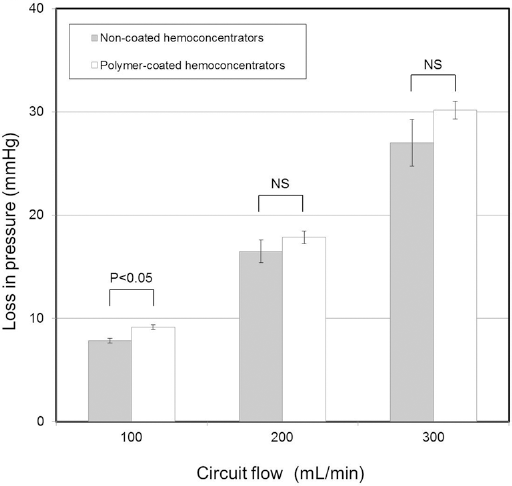
Comparison of the losses in pressure at each circuit flow velocity for non-coated and polymer-coated haemoconcentrators. The loss in pressure was calculated by subtracting the outlet chamber pressure from the inlet chamber pressure of the haemoconcentrator. The grey bars indicate the non-coated haemoconcentrator pressure loss at each circuit flow level. The white bars indicate the polymer-coated haemoconcentrator pressure loss at each circuit flow level. The error bars indicate the upper and lower standard deviations from the means estimated by least-squares regression. NS indicates that there is no significant difference.
The transmembrane pressure (TMP), which was calculated by averaging the inlet chamber pressure and outlet chamber pressure of the haemoconcentrator, was determined for return line tube occlusion levels of 0%, 50%, and 100%. The filtrated fluid flow results corresponding to the TMPs were plotted for the individual haemoconcentrators, and regression lines were fitted to the results using the least-squares method, as shown in Figure 3. The slopes of the lines for the non-coated haemoconcentrators and polymer-coated haemoconcentrators were 3.712 ± 0.170 and 6.286 ± 0.320, respectively, indicating a significant difference (p<0.05). These results suggest that the flow of the filtrated fluid, which is dependent on the TMP, is greater in polymer-coated haemoconcentrators than in non-coated haemoconcentrators.
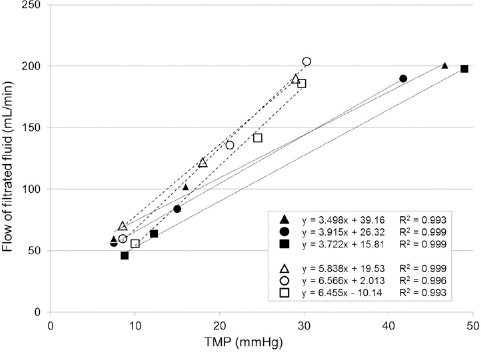
Relationship between transmembrane pressure (TMP) and flow of filtrated fluid. The TMP is calculated by averaging the inlet chamber pressure and outlet chamber pressure of the haemoconcentrator. The black triangles indicate the flow of the filtrated fluid corresponding to the TMP, which is measured at 0%, 50%, and 100% of returned line occlusion, in a non-coated haemoconcentrator. The black circles and the black squares are similarly plotted for other non-coated heamoconcentrators. The white triangles indicate the flow of the filtrated fluid corresponding to the TMP, which is measured at 0%, 50%, and 100% of returned line occlusion, in a polymer-coated haemoconcentrator. The white circles and the white squares are similarly plotted for other polymer-coated heamoconcentrators. Regression equations obtained using the least-squares method are shown. The solid lines are the regression lines for the non-coated haemoconcentrators. The dotted lines are the regression lines for the polymer-coated haemoconcentrators. R 2 is the coefficient of determination.
A polymer-coated haemoconcentrator was sliced into rounds and was observed using an SEM. The SEM results showed that the fibres were not destroyed in the cross-sectional surface of the polymer-coated haemoconcentrator. Furthermore, no shape modification of the cross-sectional surface of a hollow fibre in the polymer-coated haemoconcentrator in comparison to that in the non-coated haemoconcentrator was confirmed, as shown in Figure 4.
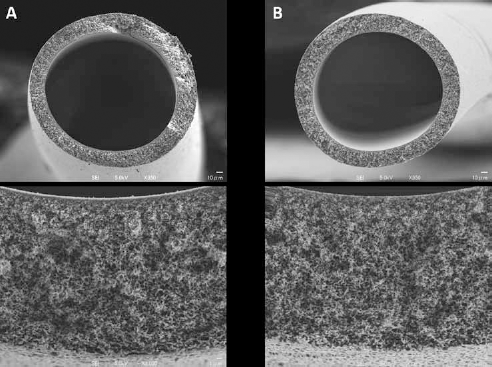
Cross-sectional surfaces of the polymer-coated and non-coated haemoconcentrators observed using a scanning electron microscope (SEM). The left side of the figure shows the cross-sectional surface of (A) a polymer-coated haemoconcentrator observed under a power of 350x (upper view) and 3000x (lower view) by SEM. The right side of the figure shows the cross-sectional surface of (B) a non-coated haemoconcentrator observed at powers of 350x (upper view) and 3000x (lower view) by SEM.
Discussion
Although devices used in cardiopulmonary bypass circuits are coated with polymers, reports on polymer-coated haemoconcentrators have not been published. It is reasonable to expect that a polymer coating, which would result due to the accumulation of a polymer coating agent on a foreign surface, would reduce the cross-sectional area of the fluid path and thereby increase drag in a circulation pathway. However, Figure 2 shows that a polymer coating does not increase drag in a circulation pathway, except in the low-flow-rate region. Furthermore, Figure 3 suggests that a polymer coating increases filtration flow. We propose the following 2 hypotheses for these results: (i) the polymer coating induces destruction of the hollow fibre, or (ii) the hydrophobic nature of the polymer coating increases the filtration flow (19). If the process of coating destroys the hollow fibre, such as by ripping, this would increase the filtration flow, but this would be unacceptable in a clinical setting. However, Figure 4 indicates no destruction of the hollow fibre, which contradicts the first hypothesis. With regard to the hydrophobic nature of the polymer coating, reports on the enhancement of slippage of liquids on solid walls by polymer coating have been published (20). Reports on the enhancement of Newtonian liquid behaviour by hydrophobicity have also been published (21, 22). Taking those reports and our results into consideration, we conclude that polymer coating makes the surface of the flow pathway more slippery, and thus enhances the filtration velocity.
Our findings suggest that polymer-coated haemoconcentrators might have both advantages and disadvantages in clinical application to extracorporeal circulation. A higher filtration velocity suggests that the velocity of the haemoconcentrate is also high, permitting a rapid correction of the haematocrit level. Furthermore, a higher filtration velocity permits faster correction of an electrolyte imbalance. Therefore, a higher filtration velocity might be advantageous in cardiopulmonary bypass surgeries.
On the other hand, there is a conceivable disadvantage related to blood shear stress. In vitro and in-animal experiments have shown that the blood shear stress is proportional to the flow or the mean viscosity and is inversely proportional to the cube of the lumen radius (23). Therefore, a higher filtration velocity could increase the shear stress in blood, resulting in damage to blood components. However, reductions in cell adhesion and protein adhesion in polymer-coated devices have been reported (12, 15). This means that cell and protein adhesion are greater in a non-coated haemoconcentrator than in a polymer-coated haemoconcentrator. The lumen radius of the fibre may be narrowed by the adhesions, resulting in a higher shear stress. Experimental testing is required to determine whether the shear stress associated with either a polymer-coated surface or a non-coated surface is disadvantageous.
Another conceivable disadvantage is alteration of the sieving coefficient of blood components. If a higher filtration velocity is achieved through an increase in the pore size of the hollow fibre, this could alter the sieving coefficient of the blood component, which would impair appropriate haemoconcentration and haemofiltration. Albumin leakage due to alteration of the sieving efficient would be a serious consequence for cardiopulmonary bypass surgeries. Moret et al. reported that lower albumin concentration induced hypotension due to lower colloid osmotic pressure (24). Testing is therefore needed to confirm that no albumin leakage occurs.
Based on these considerations, we conclude that polymer-coated haemoconcentrators might perform better than non-coated ones and offer the advantage of biocompatibility, although there are issues associated with polymer-coated haemoconcentrators that need to be examined. We believe that our findings will contribute to future investigations of the suitability of polymer-coated haemoconcentrators for use in clinical applications.
Limitations
The major limitation of this study is that the experiments were conducted using water. The loss of pressure and TMP in a Newtonian fluid differs from that in a non-Newtonian fluid, which suggests that further experiments conducted using blood are required in the next phase of this investigation.
Conclusions
The results of this preliminary study suggest that the use of a polymer-coated haemoconcentrator in an extracorporeal circulation circuit can increase the velocity of filtration because of the hydrophobicity of the polymer coating. In addition to biocompatibility, polymer-coated haemoconcentrators may offer such advantages as faster haematocrit correction and faster electrolyte imbalance correction, although further tests are required, including tests conducted using blood rather than water. We believe that our findings provide support for clinical applications of polymer-coated haemoconcentrators.
Footnotes
Acknowledgement
We are grateful for the valuable advice provided by Hironori Izutani, Professor of Cardiovascular and Thoracic Surgery at Ehime University Graduate School of Medicine, Ehime, Japan, and by Masuhide Yakehiro and Hiroshi Sumida, Professors of Physiology at Hiroshima International University, Hiroshima, Japan. We are also grateful for the analytical assistance provided by Kazumasa Nohara of A-kit Corp., Gifu, Japan.
Financial support: This research was conducted without any specific grant from a funding agency in the public, commercial, or non-profit sectors.
Conflict of interest: There are no conflicts of interest to declare.
