Abstract
Purpose
Only mechanical and biological heart valve prostheses are currently commercially available. The former show longer durability but require anticoagulant therapy; the latter display better fluid dynamic behavior but do not have adequate durability. New Polymeric Heart Valves (PHVs) could potentially combine the hemodynamic properties of biological valves with the durability of mechanical valves. This work presents a hydrodynamic evaluation of 2 groups of newly developed supra-annular, trileaflet prosthetic heart valves made from styrenic block copolymers (SBC): Poli-Valves.
Methods
2 types of Poli-Valves made of SBC and differing in polystyrene fraction content were tested under continuous and pulsatile flow conditions as prescribed by ISO 5840 Standard. A pulse duplicator designed ad hoc allowed the valve prototypes to be tested at different flow rates and frequencies. Pressure and flow were recorded; pressure drops, effective orifice area (EOA), and regurgitant volume were computed to assess the behavior of the valve.
Results
Both types of Poli-Valves met the minimum requirements in terms of regurgitation and EOA as specified by the ISO 5840 Standard. Results were compared with 5 mechanical heart valves (MHVs) and 5 tissue heart valves (THVs), currently available on the market.
Conclusions
Based on these results, PHVs based on styrenic block copolymers, as are Poli-Valves, can be considered a promising alternative for heart valve replacement in the near future.
Keywords
Introduction
Nowadays, 2 major types of implantable artificial heart valves are available: mechanical and bioprosthetic heart valves. Although mechanical heart valves (MHVs) have been the most commonly implanted devices in the past years, thanks to their reliable long term durability, bioprosthetic tissue heart valves (THVs) are currently implanted as frequently as mechanical heart valves due to their fluid dynamic behavior and freedom from anticoagulant therapy (1). As suggested by the literature (2, 3) tissue valves have a larger central flow orifice than mechanical valves, which can result in the reduction of flow turbulences. The larger orifice also improves hemodynamic conditions and allows a decrease in transvalvular pressure drops. All these elements can lead to decreased blood trauma.
However, despite the widespread use of artificial heart valves, neither MHVs nor THVs are free from complications. The former show high turbulent stresses, platelet activation, endocarditis, thromboembolic events arising from clot formation and their subsequent detachment. On the other hand, bioprosthetic heart valves suffer from structural valvular deterioration, leaflet calcification and/or tearing, and short term durability (1).
Polymeric heart valves (PHVs) were developed in order to combine the durability of the MHVs with the hemocompatibility of the THVs. The first flexible-leaflet prosthetic heart valves were implanted in the 1960s but, although synthetic heart valves exhibit acceptable short-term mechanical properties, their susceptibility to hydrolytic and oxidative biodegradation and subsequent mechanical failure have limited their successful use (4, 5).
In recent years, advances in polymer science have given rise to an important new class of artificial heart valve predominantly made of polyurethane-based material, which shows good biocompatibility and biostability (6–9). Medical grade segmented polyurethane (PU) has been successfully applied in various cardiovascular devices. PU valves are currently effectively used in commercially available assist devices (10, 11), but no polymeric prostheses have proven long-term durability for clinical implantation so far due to the hydrolytic degradation of the polyurethane chain under
More recently, the development in polymeric material technology, especially in nanocomposite polymerization, has raised interest again in fully polymeric heart valves. Kidane et al (13) proposed nano-modified polyurethanes as innovative biostable materials capable of overcoming the well-known degradation issue for standard PU by the chemical functionalization of its oxidative residuals with poly(carbonate-urea) segment bounded with polyhedral oligomeric silsesquioxanes nanoparticles (14, 15).
An alternative kind of polymer suitable for the design of synthetic heart valve prosthesis is offered by block nanocomposites. The structure of block copolymers can be tailored to achieve mechanical and chemical ad hoc properties starting from the blend of 2 or more polymers. The properties of the final product lie between those of the initial materials (16).
Among the possible blends, novel elastomers based on styrenic block copolymer have being proposed as available polymers for the construction of heart valve prostheses due to their good biocompatibility, promising mechanical properties and oxidation resistance (17–20). Indeed, commercially available block copolymers, such as poly(Styrene-IsoButylene-Styrene) (SIBS; Innovia Polymers) showed superior biochemical stability (21, 22) and good fatigue response (23, 24).
To enhance long-term durability, thrombogenicity and hemodynamic behavior, Wang et al (25) embedded reinforcement polyethylene terephthalate (PET) fabric into poly(styrene-block-isobutylene-block-styrene) (SIBS) and modified the surface of these prosthetic valves with a dimyristoyl phosphatidylcholine (DMPC) coat. These modified SIBS valves failed in animal testing because of material damage and calcification. To avoid these drawbacks, the properties of SIBS had to be improved in order to provide the structural integrity required for long-term use
In particular, we demonstrated that a cylindrical morphology of the microdomains may turn from isotropic to oriented by compression molding in die channels (20). This anisotropic micromorphology, if optimized, could mimic the structure-function of anisotropic collagen in the native valve, improving the performance of the PHV (27).
The aim of this work is to characterize the hydrodynamic behavior of new PHV prototypes (Poli-Valve) fabricated via compression molding under continuous and pulsatile conditions, in order to study the adequacy of the Poli-Valve to be used as an alternative to mechanical and tissue heart valves currently on the market.
Methods
A total of 16 new PHV prototypes (Poli-Valves) were manufactured by compression molding technique from two different commercially available styrene block copolymers (SBC) obtained from Kraton™ Performance Polymers. Two groups of valves were investigated, each one comprising 8 valves:
Group A: valves manufactured from poly(styrene-b-isoprene/butadiene-
Group B: valves manufactured from poly(styrene-
Both materials are linear block copolymers with narrow molecular weight distribution of about 180 kg mol−1 and 130 kg mol−1 for groups A and B, respectively. The different polystyrene fraction that characterizes the 2 materials is responsible for a different stiffness of these polymers. The stiffness is directly related to the polymer mechanical strength, as reported in (19).
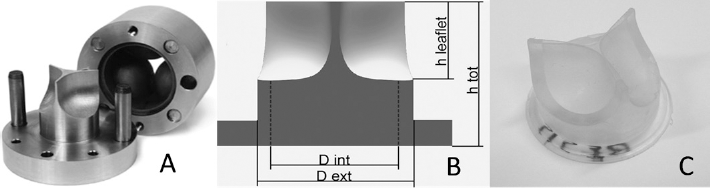
The mold elements (Fig. 1A) were manufactured in aluminum by spark erosion (Cambridge Reactor Design), based on a 3-dimensional geometry replicating the natural configuration of trileaflet heart valves (28). Poli-valve prototypes were obtained by hot pressing at 150°C cuboids (approximately 0.5 cm3) of the block copolymer initially located at the commissural of each of the 3 leaflets of the valve. The Poli-Valve geometric profile and dimensions are reported in Figures 1B and C. Poli-valves belonging to both Group A and Group B were tested
(
Continuous and pulsatile flow tests were performed using distilled water at room temperature (25°C).
Continuous Tests
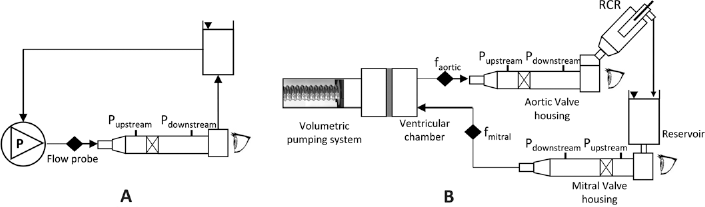
The experimental test bench used to evaluate Poli-Valve pressure drops in continuous flow conditions is shown in Figure 2A. The setup consisted of a reservoir, a centrifugal pump (BIO-MEDICUS - 550 BIOCONSOLE®; Medtronic) and a valve housing unit (HU) designed according to the guidelines of the ISO 5840 Standard. The valves were sealed with O-rings in the HU to avoid any leakage.
Outline of the test benches: (
The valves were tested at flow rates ranging from 0 to 10 L/min, with an incremental step of 0.5 L/min. Transvalvular pressure drop was measured 3 times for each valve by pressure transducers (140 PC pressure sensors, Honeywell) to verify the repeatability of the tests.
The experimental setup for the regurgitation test comprised a valve HU (the same used during the continuous flow tests); a reservoir positioned upstream to the valve, at the same geodetic level to provide a constant pressure preload, and a reservoir placed downstream from the valve, at different heights, to vary the backpressure acting on the valve.
During testing, the height of the reservoir was changed to provide a backpressure ranging within 28 to 128 mmHg (5 mmHg steps). Between 2 consecutive measurements, backpressure was set to zero mmHg to allow the valve to recover its initial configuration. The regurgitation rate at each backpressure was measured by using a transit-time ultrasound flowmeter (HT110 series, Transonic Systems).
Pulsatile Tests
Based on a previous work (30), a new pulse duplicator was designed and built up to perform pulsatile flow tests (Fig. 2B). It consists of the following elements: a volumetric pumping system, a ventricular chamber, an aortic valve housing, a systemic impedance simulator and a mitral valve housing.
The volumetric pumping system and the systemic impedance simulator have been previously described in detail (30). The pumping system was driven by an electronic controller allowing the setting of different flow rate waveforms and different frequencies. In this work, the systolic flow rate was replicated by the Swanson and Clark waveform (31), while a modified Talukder and Reul flow rate waveform (32) was used to reproduce the diastolic flow rate. The ventricular chamber allows the simulation of both physiological and pathological stroke volume (SV). The ventricular chamber and the pumping system are divided by a thin silicon membrane (thickness = 0.3 mm). This way the piston can be isolated and filled with distilled water to prevent damage to the mechanical system, while the other elements of the pulse duplicator can be filled with different fluids (e.g., blood or fluids simulating the blood rheological properties) if required. The aortic and the mitral valve housing are the same used for the continuous flow test and are connected with straight tubes to the systemic impedance simulator and to the ventricular element respectively. Furthermore, this setup allows the direct visual observation of both valves at different time steps of the cycle.
According to the UNI EN ISO 5840 Standard, the new pulse duplicator reproduces the physiological pressure and flow waveforms (Fig. 3) (i.e., arterial peak systolic pressure ranging from 100 to 130 mmHg, arterial diastolic pressure from 65 to 85 mmHg, differential pressure across closed aortic valve equal to 95 mmHg).
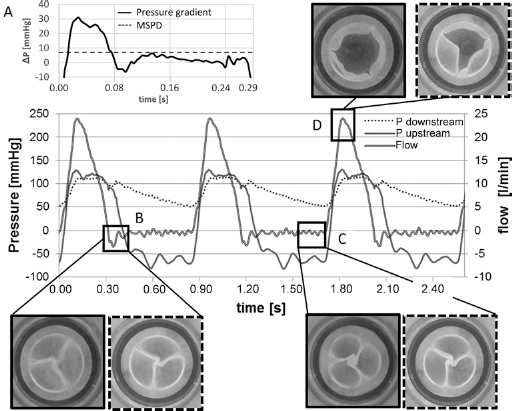
Pressure and flow tracings acquired during a pulsatile test. The frame (
The Poli-Valves were tested at different flow rates and backpressures, as required by the UNI EN ISO 5840 Standard. Transvalvular pressure drop was measured at a constant frequency (70 bpm) and variable flow rate (2, 3.5, 5 and 7 L/ min). Regurgitation volume was determined by testing each valve at a mean flow rate of 5 L/min at 3 different frequencies (45, 70, 120 bpm). At each frequency, 3 backpressures were tested (80, 120, 160 mmHg). Simultaneous measurements were recorded by using transit-time ultrasound flowmeter and pressure transducers located 35 mm upstream and 105 mm downstream from the aortic valve. Transvalvular pressure drop, regurgitation (RG), regurgitant fraction (RF) mean systolic pressure difference (MSPD) and effective orifice area (EOA) were calculated for each valve. Equation [1] was used to calculate MSPD

where Δpi. is the pressure difference across the valve in mmHg and n is the number of samples during the systolic phase.
Equation [2] from ISO 5840 (29) was used to calculate EOA

where Δp is the mean pressure difference in mmHg, ρ is the density of the test fluid in grams per cubic centimetre, and QRMS is the root mean square of forward flow in millilitres per second given by Equation [3]

where Q(t) is the instantaneous flow at time t while t1 and t2 are time at start and end of the forward flow, respectively.
Regurgitation is given by Equation [4]

where t3 and t4 are time at start and end of the backward flow, respectively. RF% is calculated by Equation [5]

Statistical Analysis
Each valve has been tested for at least 15 consecutive cycles at each test condition. SPSS Statistics version 21 (IBM®) was used to perform statistical tests. All data are reported as mean ± standard deviation (SD). Independent-samples
Results
Continuous Flow Test
Group A showed a statistically lower (p = 3.2e-4) mean transvalvular pressure drop (8.36 ± 1.33 mmHg) than Group B (15.26 ± 1.87 mmHg) at the maximum flow rate (10 L/min) (Fig. 4A).
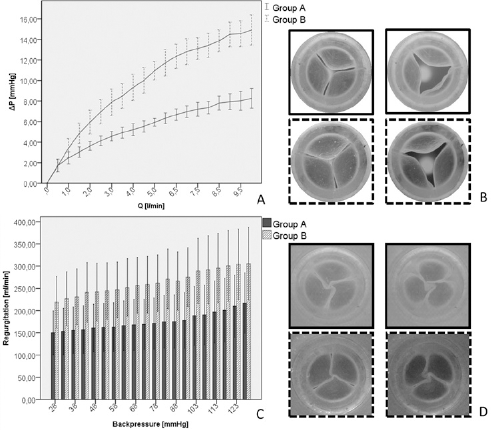
Results of continuous flow test: (
Also the mean regurgitation displayed by Group A was lower (175.3 ± 19 mL/min) than Group B (264 ± 26 mL/ min) (Fig. 4C), but the difference is statistically insignificant (p = 0.077). The small difference in regurgitation between the 2 groups may be ascribed to the different leakage area formed during the valve closing phase (Fig. 4D).
Pulsatile Flow Test
All the tested Poli-Valves exceeded the minimum performance requirements set by the ISO 5840 Standard (Fig. 5): all the 16 PHVs showed EOA> cm2 and a regurgitant volume <10% of the stroke volume (SV = 71.4 mL). The minimum performance requirements correspond to the following pulsatile-flow conditions: heart rate = 70 cycles/min, simulated cardiac output = 5.0 L/min, SV = 71.4 mL, mean aortic pressure = 100 mmHg, and systolic duration = 35%.
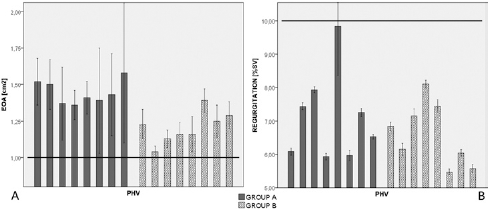
EOA (
Group A showed statistically higher EOA (1.45 ± 0.21 cm2) than Group B (1.21 ± 0.12 cm2) (p = 2.29e-4).
Mean regurgitation volume was comparable between the 2 groups (7.13 ± 1.33% of Group A vs. 6.60 ± 0.94% of Group B) (p>0.05).
The tests showed that the MSPD recorded in Group A was statistically lower (12.20 ± 1.41 mmHg) than in Group B (17.09 ± 3.39 mmHg) (p = 0.0019).
Also the maximum transvalvular pressure drops were statistically different (p = 3.3e-05) between Group A and Group B. Group A showed a maximum pressure drop equal to 30.09 ± 4.7 mmHg, while in Group B it was 43.98 ± 4.52 mmHg.
Discussion
Steady state and pulsatile hydrodynamic tests were performed to acquire information and indicators on the fluid mechanical behavior of Poli-Valves manufactured from block copolymers having different polystyrene content. The results from continuous and pulsatile flow tests demonstrated that the transvalvular pressure drop is related to the stiffness of the polymer, which increases with polystyrene mass percentage in the material (Figs. 4A and 5A).
Differences between Group A and Group B for both EOA and mean transvalvular pressure drops are statistically significant as demonstrated by independent-sample
This result can be ascribed to the reduced leaflet deformation obtained during the systolic phase in the valves belonging to Group B; in fact, the higher polystyrene weight fraction characterizing the polymer in these valves induces stiffer leaflet behavior thus resulting in a lower valve opening.
Poli-valves showed good opening and closing performances (Fig. 3) mimicking the behavior of natural heart valves.
The regurgitation in the continuous tests is lower in Group A than in Group B; inversely for the pulsatile tests, the regurgitation is lower in Group B than in Group A. However, in both tests the difference is statistically insignificant between the 2 groups.
All the 16 Poli-Valve prototypes meet the minimum requirements specified in the ISO 5840 Standard, both in terms of regurgitation and EOA (Fig. 5), demonstrating their effectiveness. However, the experimental tests performed in this study prove that the valves belonging to Group A show better behavior in terms of EOA and mean pressure drops, probably due to the lower stiffness of the leaflets, which allows a wider opening.
The better performances displayed by the valves belonging to Group A suggest that block copolymers having lower than a 30% styrene fraction should be chosen for Poli-Valve manufacturing.
The fluid dynamic behavior displayed by the 2 groups of Poli-Valves tested in this study were compared (Fig. 6) with literature data for commercial heart valve prostheses having the same tissue annulus diameter (TAD) as Poli-Valves: 5 mechanical (33) and 5 biological (34). Both EOA and regurgitation of mechanical and tissue heart valve prostheses are comparable with the data obtained for Poli-Valves. Mechanical heart valves show the highest mean total regurgitant volume (ranging from 6.08 ± 1.23% to 10.78 ± 1.53%) and EOA (from 1.43 ± 0.11 cm2 to 1.90 ± 0.10 cm2) while tissue valves show the best performances in terms of mean regurgitation (from 3.86 ± 0.85% to 17.96 ± 1.26%). The results of this comparison show that the hydrodynamic behavior of Poli-Valves may make them a valid alternative to the valve prostheses currently available on the market (Fig. 6).
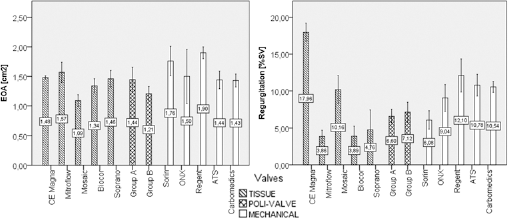
Comparison of the Poli-Valve hydrodynamic performances (EOA left and Regurgitation right) with literature data (33, 34) for tissue and mechanical heart valves at the following pulsatile flow condition: heart rate = 70 cycle/min, simulated cardiac output = 5.0 l/min, mean aortic pressure 100 mmHg and systolic duration = 35%.
The durability of the Poli-Valve not only depends on the mechanical properties of the valve materials but also depends on the manufacturing technique and on the stress distribution in the material. However, the hydrodynamic assessment presented in this paper, together with the positive results of the durability tests, would make a fully polymeric prosthetic heart valve based on polystyrene block copolymer, as is Poli-Valve, an attractive alternative to the mechanical and biomorphic valves currently used.
Footnotes
Conflict of interest: None of the authors has financial interest related to this study to disclose.
