Abstract
Background
Veno-venous extracorporeal membrane oxygenation (ECMO) is probably the preferable configuration to assist children with respiratory failure who do not respond to maximized conventional therapies. The single-vessel, double-lumen approach through the internal jugular vein is extremely advantageous, especially in infants, where femoral access presents limitations related to the small dimensions of the veins. In case of emergencies, ECMO might need to be started at the bedside, without the availability of fluoroscopic guidance. To our knowledge, a completely percutaneous approach has not been reported before in children younger than 1 year and weighing less than 5 kg.
Methods
We describe 3 cases of emergency bedside, percutaneous, bicaval double-lumen cannulation under real-time transthoracic ultrasound control in 2 neonates and 1 infant.
Results
In our experience, this approach proved to be safe, effective and time saving, while minimizing bleeding from the cannula insertion site. Cannulation times, from decision making to the beginning of ECMO flow, were 30, 28, 25 minutes respectively, from patient 1 to 3. We do not report any cannula-related injury to vessels and heart structures.
Conclusions
Our preliminary data suggest that, with the described precautions, percutaneous, echo-guided, bicaval double-lumen cannulation in neonates and infants could be effective and free from major complications. Further evaluation should be warranted in the neonatal population.
Introduction
Veno-venous (VV) double-lumen cannulation for respiratory failure is gaining greater popularity, accounting for 18.9% of all neonatal cases and 17.0% of all pediatric ones (1). Single vessel approach through the internal jugular vein (IJV) has rendered V-V extracorporeal membrane oxygenation (VV-ECMO) easier, overcoming the limitations imposed by the small dimensions of the femoral veins in newborns. VV-ECMO should be considered as the preferred modality for respiratory support in pediatrics because of the significant reduction in the morbidity compared to veno-arterial access (2, 3). Newer cannulas, like the Avalon Elite (Maquet, Rastatt, Germany), are more suitable for percutaneous placement. Recirculation, a typical problem encountered with standard double-lumen cannulas and with femoral-jugular cannulation, is minimized due to their unique design and anatomic placement. The aspiration ports are located in the superior and inferior vena cava (SVC, IVC), while the infusion port is directed toward the tricuspid valve (TV). Catheter bending and flow obstruction are prevented by wire reinforcement (4, 5).
We describe our experience in 3 cases of emergency percutaneous double-lumen bicaval cannulation in 2 neonates and 1 infant, using the 13 Fr Avalon Elite catheter under real-time transthoracic echo control, focusing on the cannulation technique. We also provide several strategies that, according to our preliminary data, proved effective in preventing major complications recently reported in the literature (6).
Materials and Methods
We reviewed all patients requiring VV-ECMO and cannulated percutaneously with the Avalon Elite cannula from January 1, 2014 to December 31, 2014. Among 8 patients (median age 10.5 months, range: term newborn at 38 weeks gestational age-6 years and 10 months; median weight 8.9 kg, range: 2.43–22 kg) the ones younger than 1 year and weighing less than 5 kg were selected. Patients demographics and characteristics are reported in Table I. Approval from our Institutional Review Board/Ethics Committee was not required for this study; informed consent was obtained from all patients’ parents.
Patients Demographics and Characteristics

ECMO = extracorporeal membrane oxygenation; GA = gestational age; CMV = conventional mechanical ventilation; HFPV = high frequency percussive ventilation; NO = inhaled nitric oxide; OI = oxygenation index.
The same cannulation technique was utilized in all cases at the bedside. To minimize the time of ECMO initiation, cannulation was performed by an intensive care unit (ICU) attending at the bedside, without the need of immediate activation of a surgical team. The cannulating physician was assisted by a scrub nurse, under real-time transthoracic ultrasound control by another ICU attending or cardiologist. The surgical team was alerted and readily available in case of major complications. After sedation and paralysis, the right IJV was cannulated under off-plane, echo direct visualization (SonoSite M-Turbo, Bothell, WA, USA; hockey stick probe, 6–13 MHz) with a 22 gauge (G) cannula. A 0.53 mm guidewire was passed through the 22 G cannula, allowing the introduction of a 4 Fr (5 cm length) Desilet introducer catheter (Vygon, Ecouen, France), after gentle incision of the skin with a No. 11 blade scalpel. The introducer can accept the 0.97 mm guidewire of the 100 cm (12-16-20-24 Fr dilators) guidewire kit for the Avalon Elite 13 Fr cannula. The 0.97 mm guidewire was advanced under transthoracic echocardiographic guidance to the proximal IVC. The progression of the guide was followed on a subcostal bicaval view. Presence of guidewire loops in the right atrium and ventricle (RA, RV) should be excluded on the subcostal 4-chambers view. Looping of the guidewire can be detected by premature complexes on the EKG, rhythm changes or cardiovascular instability (7). Since the SVC and IVC lie in different planes, the operator can find some difficulties in advancing the guide into the IVC. Head flexion, elevation of the right flank, straightening of the J tip or hokey stick shaping of the guidewire extremity can facilitate its progression in the IVC. Moreover, because the tip of the guidewire can sometimes wedge into an hepatic vein (HV), slight manual pressure on the liver can close the HV opening in the IVC, allowing the correct progression of the guidewire after retraction. The guide is then advanced deeply into the IVC. However, care must be taken to avoid its inadvertent looping in the RA-RV, which may occur while pushing forward the guidewire with the J tip wedged into the iliac vein (IV).
Before serial dilations of the skin, cardiac echo should confirm the straight and uncoiled passage of the guidewire that is marked at the skin exit site as a reference. Once a 12 Fr dilator is easily passed through the skin, the 13 Fr cannula can be advanced under transthoracic echo guidance (bicaval view) until the tip terminates in the IVC, distal to the hepatic veins. Excessive incision or dilation of the skin were avoided to prevent insertion site bleeding.
After ECMO support is started, the correct position of the cannula is confirmed by echo with demonstration of flow from the infusion lumen directed toward the tricuspid valve. The cannula is then definitively secured to the skin. Cannula position was checked by echo every 8 hours, and in the case of ECMO flow decrease with constant revolutions per minute or increase in the negative aspiration pressures.
Results
Emergency transthoracic echo-guided percutaneous cannulation using the 13 Fr Avalon Elite was effectively carried out at the bedside in all patients by the ICU attending. Surgical team activation was never required. Cannulation time, from decision making to beginning of ECMO flow, was 30, 28, and 25 minutes respectively, from patient 1 to 3. ECMO flows through the cannulas were always adequate to meet the patients’ needs, with aspiration negative pressures within safety ranges, and optimal performance of the circuit (Tab. II). Bleeding from the cannula insertion site was not observed in our patients, even after 27 (patient 2) and 23 (patient 3) days of support. We do not report any cannula-related injury to vessels and heart structures. This was clearly demonstrated at autopsy in Patient 2 (Fig. 1). In Patient 2, because of the increase in lung compliance and downward movement of the diaphragm, we experienced the displacement of the tip of the cannula in an HV. Attempts to advance it deeper in the IVC led to recirculation. We decided to tolerate its ending at the confluence of the HV with the IVC, after a minimal retraction.
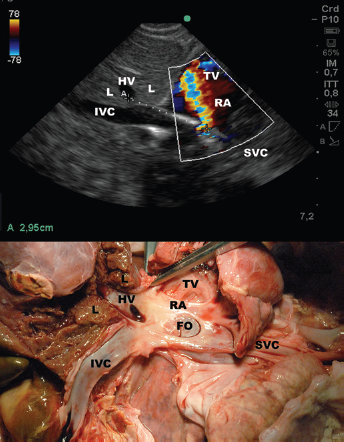
Ultrasound and anatomic correlates in Patient 2. The figure shows that when the arterial flow is correctly directed towards the TV, the tip of the cannula faces the HV opening into the IVC. SVC and HV lie in the same sagittal plane, thus the natural progression of the cannula tip, if advanced, is into the HV. The distance between tip and infusion port of the cannula has been measured with minimal error (2.95 instead of 2.8 cm) with ultrasound. Note the absence of vascular and cardiac cannula-related lesions after 27 days of support (post-mortem study). L = liver; HV = hepatic vein; IVC = inferior vena cava; FO = foramen ovale; RA = right atrium; TV = tricuspid valve; SVC = superior vena cava.
ECMO variables
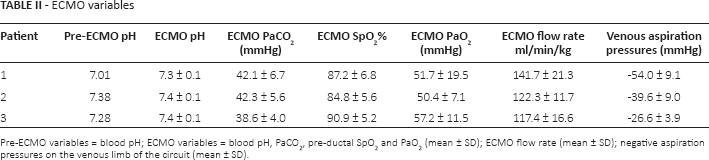
Pre-ECMO variables = blood pH; ECMO variables = blood pH, PaCO2, pre-ductal SpO2 and PaO2 (mean ± SD); ECMO flow rate (mean ± SD); negative aspiration pressures on the venous limb of the circuit (mean ± SD).
Discussion
Our preliminary data show that transthoracic echo-guided percutaneous cannulation of newborns and infants with the 13 Fr Avalon Elite can be effective and straightforward, even at the bedside and in emergency conditions. A true percutaneous approach, using a 4-Fr introducer, allowed an extremely gentle approach to the vessel, which is punctured with a 22 G cannula instead of the 18 G needle included in the Avalon introduction kit.
To our knowledge, a complete percutaneous, bicaval double-lumen cannulation technique has not been reported in children under 1 year of age and weighing less than 5 kg. It has been shown in the recent literature that percutaneous cannulation can be achieved rapidly, with fewer bleeding complications compared to open or semi-Seldinger techniques (1, 6, 8, 9). Imaging guidance is of paramount importance in order to ensure correct positioning of a bicaval double-lumen cannula. Fluoroscopy is often not available in the ICU, and transthoracic echo represents a valid alternative in newborns and infants (10). Advancement of the guidewire in the IVC-IV is the most complex and time-consuming step, and is common to percutaneous, semi-Seldinger, and open techniques. RA, TV, and RV rupture and tamponade have been described in conjunction with guidewire looping and/or difficult or non monitored advancements of dilators and cannulas (7, 11).
To prevent recirculation, the cannula infusion port should face the TV with the distal end sitting in the IVC (12). The distance between the infusion port and the aspiration tip is 2.8 cm for the 13 Fr Avalon Elite. Optimal positioning of the infusion port could coincide with the tip of the cannula being placed in front of the caval merging of an HV. Since the SVC and the HV lie almost on the same sagittal plane, in case of inadvertent displacement, the natural progression of the cannula tip is into the HV. This might cause ECMO flow limitation or obstruction of the venous drainage from the liver with an acute Budd-Chiari syndrome (13). These complications do not occur if the tip of the cannula is advanced beyond the HV under transthoracic echocardiographic guidance (caval long-axis view). Unfortunately, in some anatomical conditions, as in patient 2 (after displacement) and patient 3 (during cannulation), this maneuver can result in the infusion port being moved into the IVC, with recirculation and severe desaturation (Fig. 1). The cannula was therefore positioned with its ending at the confluence of the HV with the IVC in both cases. We do not report any ECMO flow problems (Tab. II) nor obstructed flow in the HV.
Frequent echo monitoring of the cannula position and eventual repositioning of the head and shoulders allowed optimization of blood drainage with minimal recirculation. Retraction of the cannula in the RA has been reported as a strategy to optimize ECMO flows in the largest experience on the use of the 13 Fr Avalon Elite in neonates. These authors suggest this as a possible cause of the high rate of atrial perforation in their series (6) (see Fig. 2 and the video “Cannula displaced in the right atrium”, available online as supplementary material at www.artificial-organs.com). Moreover, pulling back the cannula can expose the proximal aspiration ports to room air with the risk of embolization. For these reasons, they stopped to use the 13 Fr Avalon cannula in neonates, a controversial conclusion in light of some positive previous experiences (4, 5). We never observed nor would we have tolerated the displacement of the cannula in the RA. This was considered as an indication to emergent repositioning or switch to veno-arterial ECMO, despite good flows and oxygenation, to avoid the risk of atrial perforation. In our patients, maintaining the cannula stable within the IVC, even with the distal end close to an HV, probably prevented major complications and injuries to the heart and vessels (Fig. 1). Most complications would hopefully be overcome with the introduction of the new 13 Fr Avalon Elite-i (RA) catheter, which is sited in the RA, with improved recirculation characteristics and wire reinforcement compared to conventional double lumen cannulae.
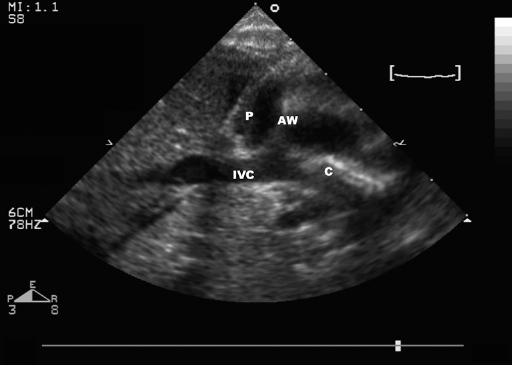
Possible mechanisms of atrial wall perforation. Ultrasound imaging showing the right atrial wall pulled by the aspiration port of an Avalon Elite cannula incorrectly positioned in the right atrium; the atrial wall is detached from the pericardium. Cannulation was performed with an open surgical technique, without using a guidewire. The tip of the cannula was then repositioned with a guidewire in the IVC (patient not included in this series). Aspiration and not only erosion can be implicated in heart perforation. P = pericardium; AW = atrial wall; IVC = inferior vena cava; C = cannula.
To avoid possible lethal complications, unlike other authors, we think that real-time transthoracic echo should guide every step of the procedure (5). In conclusion, we suggest that, with the described caveats, the use of the 13 Fr Avalon Elite deserves further investigation in neonates and infants until data on newer cannulas will be available.
Footnotes
Acknowledgement
We would like to express our gratitude to Prof. John Arnold from the Division of Critical Care Medicine of Boston Children's Hospital, and to Prof. Peter Laussen from the Department of Critical Care Medicine at The Hospital for Sick Children in Toronto for their friendly advice.
Financial support: None.
Conflict of interest: The authors have no conflicts of interest to disclose.
