Abstract
Purpose
We have designed a desensitization program that gives good results and is cost effective for kidney-transplant patients who have a potential living donor, who are ABO incompatible (ABOi), and who may or may not have donor-specific alloantibodies (DSAs).
Methods
Desensitization at pretransplant is based on immunosuppressants (such as rituximab, tacrolimus, and mycophenolic acid) and apheresis to retrieve potentially detrimental isoagglutinins and DSAs from blood. In 2011, we implemented immunoadsorption (IA) instead of plasmapheresis in our center as part of the desensitization protocol. Because IA is very tedious and time-consuming, we decided to perform IA and hemodialysis (HD) in tandem instead of performing these methods sequentially. Herein, we report on more than 100 of these tandem procedures and compare the results to those obtained when IA was performed before a HD session.
Results
The tandem process resulted in nursing time being cut by half: from almost 10h30 min/patient to 6 h/patient. When IA was performed alone, body-weight gain was 1 kg (range 0 to 1.75) and the median volume of plasma treated per session was 6500 mL (range 4500 to 10 600). Conversely, during a tandem IA + HD session, the net median weight gain was −1.5 kg (range −4.1 to 0) and the median volume of plasma treated per session was 5000 mL (range 4000 to 8600). In addition, the tandem procedure was as well tolerated as that for IA followed by HD.
Conclusions
Tandem IA plus HD is a safe and cost-effective procedure.
Keywords
Introduction
Due to the burden of end-stage renal disease (ESRD) across the world, increasing numbers of patients are treated with supportive therapies that include hemodialysis (HD) as well as kidney transplantation (KT). Hence, in France, at the moment there are more than 60 000 ESRD patients treated by dialysis therapy, mainly HD (>90% of patients). Of these, about 12 000 are listed on the national kidney-transplant waiting list, but only about 3 000 a year receive a kidney transplant; in most cases (≍88%), this is from a deceased donor. The yearly number of brain-dead patients is plateauing. Therefore, we have to develop living-kidney transplantation. However, in the latter setting, we can be faced with either blood-type (ABO) incompatibility (referred to as ABOi) and/or human-leukocyte antigen (HLA) incompatibility (referred to as HLAi). Hence, in order to use a living donor, when available in the setting of ABOi and/or HLAi, we need to desensitize the recipient at pretransplant to remove potential deleterious antibodies from the blood, i.e., isoagglutinins in the setting of ABOi, and donor-specific anti-HLA allo-antibodies (DSA) in the setting of HLAi (1–5).
Pretransplant desensitization protocols are based on 2 cornerstones: immunosuppressive drugs and apheresis techniques. The latter can remove culprit antibodies from the plasma, whereas the former can prevent re-synthesis of culprit antibodies. In this setting, apheresis can rely on either plasmapheresis (PP; or a derived technique, such as double-filtration plasmapheresis [DFPP]) or immunoadsorption (IA) (6).
PP has been a well-established technique for almost 4 decades: it is easy to setup and is not too expensive. However, it is a non-selective technique (see Tab. I). A few studies have evaluated PP for its use as a serial procedure, i.e., with HD (7–9). Because PP is a non-selective procedure and results in depletion of clotting factors, which are of utmost importance when contemplating kidney transplantation, we decided to desensitize our kidney-transplant candidates using IA instead of PP.
Comparisons between plasmapheresis and immunoadsorption

IA can be either specific or semi-specific (see Tab. I).
With regard to specific (i.e., selective) IA, as yet, there is only 1 type of column available, namely, the Glycorex (Glycosorb ABO; Glycorex Transplantation, Lund, Sweden). These columns are covered with Staphylococcus A protein on which antigens are implanted either from isoagglutinin A (anti-A column) or from isoagglutinin B (anti-B). These columns are registered as non-reusable even though we know that some apheresis centers reuse them after appropriate procedures. These columns are very expensive, around €2 800 euros per column.
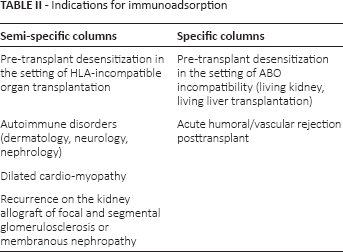
Semi-specific columns (Immunosorba, Globaffin, Fresenius) are columns covered with Staphylococcus A protein. For each session, we need 2 columns that work in parallel on an alternate basis. These columns are marketed as reusable (at least 10 sessions). The cost per patient is around €5 800 and €10 500 when using Immunosorba, and Globaffin, respectively. Semi-specific IA columns adsorb mainly immunoglobulins of isotype IgG, IgM and, to a lesser extent, IgA. They therefore adsorb both the “bad” as well as the “good” antibodies. Semi-specific IA barely removes complement components. However, when the latest generation of semi-specific columns (the Globaffin columns) are used in association with membrane filtration (Monet), it is associated with the removal of immunoglobulins, but also many complement components (10). Apart from being used in the setting of pretransplant desensitization (and sometimes at posttransplant), IA can be used in other settings, for example, in the recurrence of glomerulopathies on kidney allograft, such as focal and segmental glomerulosclerosis; in membranous glomerulopathies, autoimmune disorders (dermatology, neurology, nephrology); or in dilated cardiomyopathy (see Tab. II).
Indications for immunoadsorption
In the setting of kidney transplantation, most patients who need to be desensitized at pretransplant are receiving hemodialysis. When we started our desensitization program in 2011, IA was performed on one day and then hemodialysis afterwards on either the same day or the next day: this was very time-consuming for both the nurses and patients. Thus, because we were familiar with the IA technique, we decided to perform IA plus HD in tandem.
The aim of this study was to compare, in a prospective consecutive manner, the efficacy and effectiveness of IA followed by HD (over 2 days) versus IA and HD performed as a tandem procedure.
Patients and methods
Patients
This was a single-center study that included 19 consecutive hemodialysis patients who were in our department for ABOi and/or HLAi kidney transplantation from a living (un) related donor. Overall, there were 12 ABOi patients and 7 ABOi/HLAi patients. ABOi KT was attempted only when the pre-desensitization titer of one or more specific isoagglutinins was ≤1/128. If this was the case, the patient was given the pretransplant desensitization protocol (see Fig. 1). This included a single injection of rituximab (375 mg/m2) at 30 days pretransplant. Specific IA sessions were started on pretransplant day 10. We always scheduled 4 specific IA sessions (every other day) with titration of the isoagglutinin titer before every IA session. The goal was to achieve a pretransplant titer of ≤1/8.
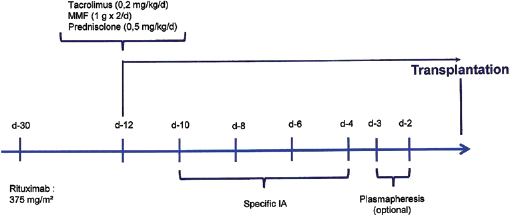
Pretransplant desensitization protocol in the setting of ABO-in-compatible kidney transplantation. MMF = mycophenolate mofetil; d = day; IA = immunoadsorption.
In some cases, when isoagglutinin titers did not decrease rapidly, we implemented a PP session between 2 IA sessions. PP sessions on pretransplant days 3 and 2 were optional. We never performed PP on day −1 pretransplant due to the high risk of bleeding peritransplant. Other immune suppressants – tacrolimus (0.2 mg/kg/d), mycophenolate mofetil (1 g b.i.d.), and prednisolone (0.5 mg/kg/d) – were started at day 12 pretransplant. At the same time, we also started Pneumocystis jirovecii prophylaxis using sulfamethazole/trimethoprim, scheduled for 1 year.
In the setting of ABOi + HLAi kidney transplantation we used a different protocol (see Fig. 2). IV-Ig (1 g/kg during an HD session) was infused at 40 days pretransplant. The patients received 2 doses of rituximab (375 mg/m2) on pretransplant days 30 and 15. Semi-specific IA was started on pretransplant day 17. The number of IA sessions was determined according to the intensity of the pre-sensitization DSA titer: if it had >10 000 units of mean fluorescence intensity (MFI), we planned at least 9 sessions. In cases where DSA(s) MFI was <10 000 we planned 6 sessions. After every 3 sessions we assessed the DSA(s) MFI in order to plan the next block of IA sessions.
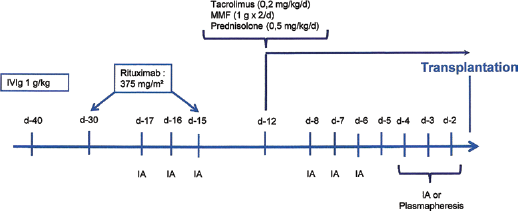
Pretransplant desensitization protocol in the setting of ABO-incompatible/HLA-incompatible kidney transplantation. MMF = mycophenolate mofetil; d = day; IA = semi specific immunoadsorption.
Procedures
All patients either had an arteriovenous (AV) fistula or a venous central catheter with over 300 ml/min blood flow. Hemodialysis was performed 3 times a week.
Hemodialysis sessions were performed using a 5008 generator (Fresenius, Bad Homburg, Germany) and polysulfone dialysis membranes. The blood volume in the circuit was ∼300 mL. Immunoadsorption was performed as previously described (11).
The plasma was first separated by centrifugation (Com. Tec; Fresenius, Bad Homburg, Germany) and was then passed through 2 Sepharose plus protein A columns (Immunosorba; Fresenius, Bad Homburg, Germany), which were operated in tandem: while one was rinsed and regenerated, the other filtered the plasma. The filtered plasma was then re-infused into the patient. An IA session was usually scheduled to last ∼4 h. The blood volume within the circuit was only 80 mL.
The tandem procedure
HD and IA were performed in tandem: this required Y-shaped arterial and venous lines (Fig. 3). With regards to the connections, first, the cell-separator was setup according to the medical prescriptions, then both the arterial and venous Y connectors were inserted, and then a 3-way tap on the return line was set up. The hemodialysis loop was then set up and connected to red branch on the Y-line as well as to the return on the blue branch of the venous Y-line. After this, the centrifugation cell separator was connected to the monitor (ADAsorb; Medicap, Ulrichstein, Germany) on which the IA columns were placed and the lines were connected to the blue branch of the Y-line.
The IA procedure was started with a blood flow within the cell separator of 30 ml/min; the separated plasma was then passed to the apheresis device, and a CaCI2 syringe was connected within the flow line, according to the patient's medical prescription. Thereafter, there was a global check of the patient's vital signs as well as both monitors. Blood flow was then subsequently and gradually increased within the cell separator to 60 ml/min. Figure 4 shows a patient surrounded by all the above-mentioned devices needed to perform the tandem procedure.
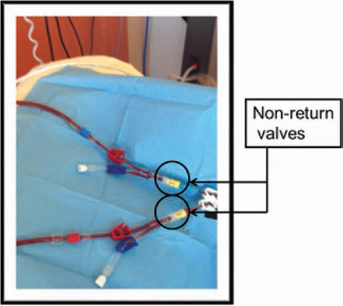
Two Y-shaped lines, connected to a central veno-venous catheter. On each branch there are 2 lines: one for immunoadsorption and the other for hemodialysis.
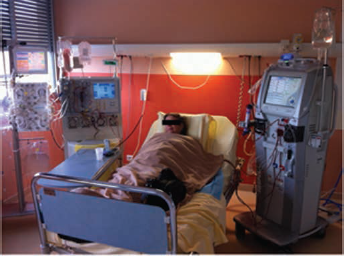
Patient with the 3 devices required to perform immunoadsorption and hemodialysis in tandem. From left to right the apharesis device with the immunoadsorption columns, the cell separator, the patient, and the hemodialysis generator.
Anticoagulation of the apheresis circuit was performed on the apheresis side by ACD citrate: for a blood flow of 24 ml/min we inject 1 ml/min of ACD citrate to assure calcium chelation. The latter is efficient when calcium within the circuit is between 0.3 and 0.5 mmol/l. Calcium restitution is made via the return line with CaCI2 (5 ml/h).
Results
There were 10 patients within group 1 (IA followed by HD) and 9 patients within group 2 (IA+ HD in tandem). All patients were ESRD patients who were being treated by hemodialysis either via an AV fistula or a permanent central venous catheter. All patients were being referred for a living (un)related kidney transplant.
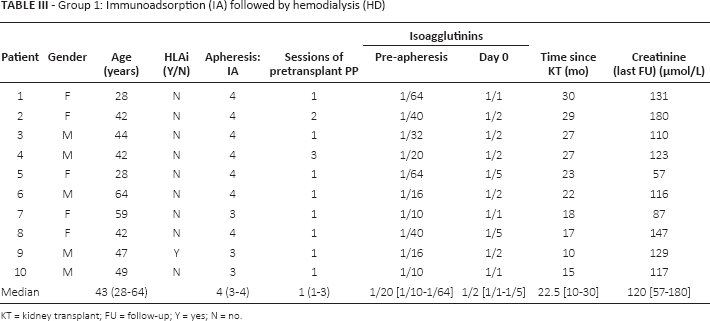
Group 1 consisted of 5 males and 5 females, with a median age of 42 years (range 28–64). All were waiting for their first kidney transplantation. All were ABO-incompatible (ABOi); in addition, 1 had DSA(s) against his donor, i.e. HLAi KT (see Tab. III).
Group 1: Immunoadsorption (IA) followed by hemodialysis (HD)
KT = kidney transplant; FU = follow-up; Y = yes; N = no.
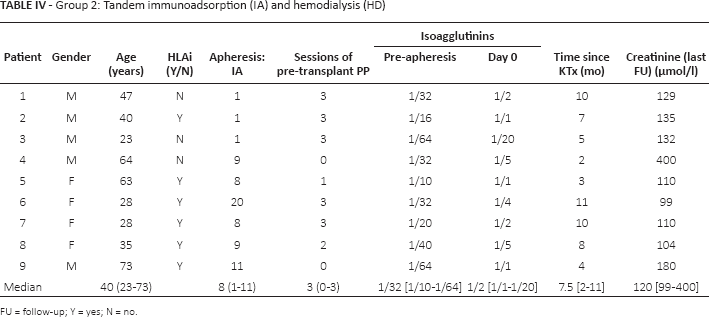
Group 2 consisted of 5 males and 4 females, with a mean age of 40 years (range 23–73). Four were waiting for their first kidney transplant, whereas 5 others were waiting for a re-transplant. All were ABOi and 6 were also HLAi (see Tab. IV). The mean follow-up times at post-KT was 22.5 months (range 10–30) for group 1 and 7.5 months (range 2–11) for group 2.
Group 2: Tandem immunoadsorption (IA) and hemodialysis (HD)
FU = follow-up; Y = yes; N = no.
Pre-transplant desensitization: IA followed by HD versus IA + HD: nursing time
In the sequential procedure, IA took ∼6 h to complete: ∼60 min to prepare the plumbing loop and to connect the patient to the machine/circuit. The IA session took between ∼3 h: 30 min and 4 h: 00 min. It then took ∼60 min to disconnect the loop and to rinse/store the IA columns in cases where it was semi-specific IA. In cases of specific IA, where the columns were not reused, the disconnecting procedure was shorter and took only ∼20 min. HD was performed after IA either on the same or the next day. This was conducted as a regular HD session and took ∼4 h: 30 min: 20 minutes to prepare the loop and connect the patient to the circuit, ∼4 h for the HD session, and then ∼10 min to disconnect the loop (Tab. V). Altogether, this sequential procedure took ∼10 h: 30 min of nursing time and nursing surveillance.
Estimation of nursing time according to the technique used: either immunoadsorption (IA) followed by hemodialysis (HD), or IA and HD as a tandem procedure
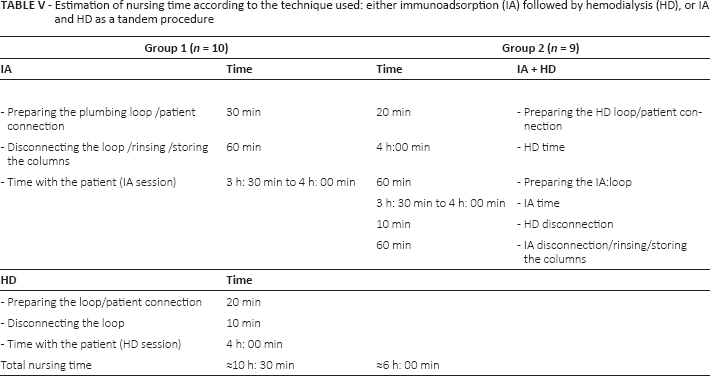
In contrast, the tandem IA + HD procedure could be performed within half a day. It took ∼20 min to prepare the HD loop and to connect the patient and then ∼ 4 h for the HD session. In the meantime, the IA loop was prepared (∼60 min): the IA took between 3h: 30 min and 4 h: 00 min. It took ∼10 min to disconnect the HD circuit and then ∼60 min to disconnect the IA and, if necessary, to rinse and store the columns (see above). In total, the tandem procedure took only ∼6 h: thus, the net nursing time saved was ∼4 h: 30 min per IA session. Hence, the tandem procedure (IA plus HD) was more cost effective compared to one IA session followed by one HD session: this is of considerable importance.
When an IA session was performed sequentially, i.e. before (the day of) an HD session, there was almost always a fluid overload with a net median weight gain at 1 kg (range 0 to 1.75) and the median volume of plasma treated per session was 6 500 mL (range 4500 to 10 600). Conversely, during a tandem IA + HD session, even though IA was associated with fluid overload, this was easily controlled by the hemodialysis machine, and in this setting the net median weight gain was −1.5 kg (range −4.1 to 0) and the median volume of plasma treated per session was 5000 mL (range 4000 to 8600).
Efficiency of IA
In group 1, most patients (9/10) were ABOi candidates, whereas only one was an ABOi/HLAi candidate. On average, each patient received 4 (range 3–4) IA sessions at pretransplant. In addition, in order to lower the isoagglutinin titers, the patients received, on average, one (range 1 to 3) plasmapheresis session, which was performed on the day between the IA sessions. With regards to the isoagglutinin titers, the initial titer was 1/20 (range 1/10 to 1/64), and this value was reduced to ½ (1/1 to 1/5) by the day of kidney transplantation.
In group 2, 6 of the 9 patients were HLA-incompatible as well as being ABOi. Thus, in this situation, the patients needed more IA sessions to decrease DSA titers. Hence, they received 8 (range 1 to 11) IA sessions and also 3 (range 0 to 3) PP sessions on the day between the IA sessions. With regards to isoagglutinin titers: these decreased from 1/32 (range 1/10 to 1/64) before the IA program to ½ (1/1 to 1/20) by the day of kidney transplantation.
Posttransplant outcomes
The follow-up at posttransplant was longer for group 1 (22.5 months; range 10 to 30), compared to group 2 (7.5 months; range 2 to 11). All patients are still alive and all transplants are still functioning. The median serum-creatinine levels at the end of follow-up were 120 μmol/1 (range 57 to 180) in group 1 and 120 μmol/1 (range 99 to 300) in group 2. Three patients presented with a humoral/vascular acute rejection episode.
Patient 4, on day 10 posttransplant, had decreased urine output and increased serum creatinine. The isoagglutinin titer was not increased (1/8). We performed a kidney biopsy, which showed an interstitial hemorrhage, polymorphonuclear interstitial infiltration, and C4d staining was diffusely positive. Because of this vascular/humoral acute rejection, we treated the patient with 6 plasmapheresis sessions, pulses of methylprednisolone (10 mg/kg for 3 consecutive days), plus rituximab (375 mg/m2, 1 dose). He then improved rapidly and, by the last follow-up, his serum creatinine was 120 μmol/1.
Two patients from group 2 presented with an acute rejection: in 1 case this was an acute T-cell-mediated rejection, which occurred on day 20 posttransplant. This patient was treated with 3 pulses of methylprednisolone (10 mg/kg each); he then recovered rapidly and his current serum creatinine is 100 μmol/1. The second patient (no. 4) first presented, on postop day 1, with a renal-vein thrombosis that required surgical repair and anticoagulation therapy. Within the next few days, he experienced delayed graft function with oliguria. By day 10 posttransplant, he had developed schistocytic anemia and thrombopenia. The isoagglutinin titer was 1/4. He underwent a kidney biopsy that showed acute vascular rejection with many glomerular thrombi. Therefore, he was treated with daily plasmapheresis (total: 10), but this was not effective for the schistocytic anemia or thrombopenia. At this point, we decided to place him on eculizumab therapy (1200 mg/week for 2 weeks and, thereafter, 900 mg every 2 weeks for up to 3 months posttransplantation). He progressively recovered from the delayed graft function. By 1 month posttransplant, his serum creatinine was ∼300 μmol/1 and became stabilized at this level thereafter.
Discussion
Our study is, to the best of our knowledge, the first to report on tandem immunoadsorption and hemodialysis. We have demonstrated that it was safe and efficient. In the past there have been some reports on coupling plasmapheresis to hemodialysis in serial procedures, but not as a tandem procedure. Serial procedures are much more tedious and impractical because when there is a problem on one circuit the whole procedure has to be stopped. Conversely, in a tandem procedure, because the 2 procedures are totally independent, one can be stopped while the other is continued.
Bhowmik et al were the first to report on tandem plasmapheresis (PE)/hemodialysis in 2 patients. The plasma-filter was inserted into the extracorporeal circuit after the hemodialyzer. A total of 8 such sessions using tandem PE and hemodialysis were performed. The total procedure was completed in the same time as is required for routine hemodialysis. The total amount of priming fluid was also less when PE and hemodialysis were performed separately. They concluded that it was economically beneficial to the hospital and also convenient to the patient. Apart from transient episodes of hypotension, which were corrected by saline infusion, no other complications were noted (7). Later, Dechmann-Sültemeyer et al reported on 82 patients in whom 483 tandem PE/hemodialysis treatments were performed over a 16-year period (8). The components of the dialyzer (polysulfone membrane) and plasma filter were serially connected by a continuous arteriovenous hemofiltration (CAVH) system. In extracorporeal circulation, using a blood pump, the patient's blood was led first to the plasma filter and then into the dialyzer. The substitute connection was located behind the plasma filter and before the dialyzer. They emphasized that, at the beginning, it is obligatory to carry out an inspection of the tubing system to check for leakages. Afterwards, the system is flushed with a heparinized (5000 IE) sodium-chloride solution, which is removed thereafter. During treatment, a blood flow of 150 ml/min to 200 ml/min is possible. In each case, plasma-filtration and ultrafiltration should not exceed 25% of blood flow. The total procedure did not take longer than a routine hemodialysis session (3 to 4 h). They found that none of the patients had volume disturbances caused by plasma shifts, and that imbalances of electrolytes and acid-base balance were immediately equalized. There were no episodes of hypotension or bleeding. Back-filtration did not occur. Hence, they concluded that this serial procedure was safe and efficient for saving time.
More recently, Pérez-Sáez et al reported on 36 patients who were treated with 287 tandem PE and HD sessions (mean 7.97±5.6 per patient). PE was connected 30 min after HD was started. The mean HD blood flow was 313.7 ± 44 ml/min, the mean PE blood flow was 141 ± 25 ml/min, and the duration of the tandem procedure was no longer than 240 min. The heparin dose was similar to that used for a standard HD procedure. Of the 287 tandem sessions performed, 10.45% experienced minor complications. There were significant changes in mean blood pressure after connection of the PE system. However, these differences were not clinically relevant as patients remained asymptomatic and did not require saline infusion. The authors concluded that this tandem treatment decreased exposure to an extracorporeal circuit, thereby reducing the risks that are associated with anticoagulation agents and it improved the patients’ comfort (9).
As compared to previous reports (7–9), our technique is much more flexible in that each of its components, IA and HD, are not dependent on each other, and therefore one can be stopped (if needed) while the other can continue. Moreover, this kind of procedure saves considerable nursing time because the tandem IA + HD takes only ∼6 h as compared to ∼10 h: 30 min when the HD session is performed after the IA session. From the patient's perspective, the tandem procedure improves quality of life by reducing the time he/she has to spend in hospital attached to a machine. In addition, when the IA procedure is performed alone it is associated with a net weight gain of around 1 kg: this may cause cardiac disorders in hemodialysis patients in whom fluid overload is poorly tolerated. Conversely, when IA is performed in tandem with HD, the patient loses weight and total weight loss can then be easily determined by the dialysis machine.
At a time when there are drastic budget cuts in hospitals, significant cost savings can be made by saving nursing time. By using this procedure, in which dialysis is only needed for half a day (and not an entire day) and HD is conducted in parallel with IA, it enables twice as many patients to be treated per day, thereby increasing the overall efficiency of the dialysis unit.
With respect to the dialysis patients, because desensitization is performed on an outpatient basis and because transportation costs are paid by the health-care system, considerable cost savings are achieved by having the same patient only need 1 treatment session rather than 2. Moreover, with regards to quality of life, the patient has to spend less time and make fewer visits to the hospital.
In conclusion, this is the first report on a large series of patients undergoing immunoadsorption performed in tandem in living kidney-transplant candidates who require pretransplant desensitization because of ABO incompatibility (with or without HLA incompatibilities). We found that the tandem procedure was as efficient as IA followed by HD and, additionally, was associated with a large reduction in nursing time and costs as well as improving patient comfort.
Footnotes
Financial support: The work was supported by Fresenius Medical Care and by AOL de Toulouse.
Conflict of interest: None to report.
