Abstract
Purpose
The intra-aortic balloon pump (IABP) provides circulatory support through counterpulsation. The hemodynamic effects of the IABP may vary with assisting frequency and depend on IAB inflation/deflation timing. We aimed to assess in vivo the IABP benefits on coronary, aortic, and left ventricular hemodynamics at different assistance frequencies and trigger timings.
Methods
Six healthy, anesthetized, open-chest sheep received IABP support at 5 timing modes (EC, LC, CC, CE, CL, corresponding to early/late/conventional/conventional/conventional inflation and conventional/conventional/conventional/early/late deflation, respectively) with frequency 1:3 and 1:1. Aortic (Qao) and coronary (Qcor) flow, and aortic (Pao) and left ventricular (PLV) pressure were recorded simultaneously, with and without IABP support. Integrating systolic Qao yielded stroke volume (SV).
Results
EC at 1:1 produced the lowest end-diastolic Pao (59.5 ± 7.8 mmHg [EC], 63.4 ± 11.1 mmHg [CC]), CC at 1:1 the lowest systolic PLV (69.1 ± 6.5 mmHg [CC], 76.4 ± 6.5 mmHg [control]), CC at 1:1 the highest SV (88.5 ± 34.4 ml [CC], 76.6 ± 31.9 ml [control]) and CC at 1:3 the highest diastolic Qcor (187.2 ± 25.0 ml/min [CC], 149.9 ± 16.6 ml/min [control]). Diastolic Pao augmentation was enhanced by both assistance frequencies alike, and optimal timings were EC for 1:3 (10.4 ± 2.8 mmHg [EC], 6.7 ± 3.8 mmHg [CC]) and CC for 1:1 (10.8 ± 6.7 mmHg [CC], −3.0 ± 3.8 mmHg [control]).
Conclusions
In our experiments, neither a single frequency nor a single inflation/deflation timing, including conventional IAB timing, has shown superiority by uniformly benefiting all studied hemodynamic parameters. A choice of optimal frequency and IAB timing might need to be made based on individual patient hemodynamic needs rather than as a generalized protocol.
Introduction
The intra-aortic balloon pump (IABP) provides circulatory support to the failing heart through counterpulsation (1, 2). Positioned in the descending thoracic aorta, the intra-aortic balloon (IAB) is inflated in early diastole, enhancing coronary blood flow through an increase in diastolic aortic pressure. IAB deflation in late diastole reduces end-diastolic aortic pressure and consequently left ventricular (LV) afterload, wall tension and myocardial oxygen demand.
For efficient IABP support, the correct timing of IAB inflation and deflation is vital (3). Current convention is for the onset of inflation to coincide with aortic valve closure, as land-marked by the dicrotic notch, while deflation must start shortly before the onset of the following systole. Without a suitable visual marker to guide the latter setting, the onset of deflation is usually fine-tuned to generate the largest reduction in end-diastolic aortic pressure. Due to the sheer number of vascular, ventricular, and coronary parameters of interest, thorough in vivo studies of the hemodynamic consequences of non-conventional IAB trigger timing are challenging to conduct (4). Earlier reports focus on a subset of hemodynamic parameters and inflation/deflation trigger timings (3, 5, 6). Nonetheless, it is generally thought that early inflation and late deflation can be detrimental because they disrupt LV ejection, during the preceding late systole and the following early systole, respectively. Instead, late inflation and early deflation are considered suboptimal in terms of therapeutic benefit, but not hazardous.
Assistance frequency is another time-related IABP operating parameter. During IABP weaning, the operator can select the assistance frequency delivered to the patient and can opt for switching from full assistance to partial assistance (support provided every heart beat (1:1) or every second/third/fourth (1:2/1:3/1:4) beat, respectively). It is generally assumed that the hemodynamic benefits of full and partial support per supported beat are alike. Namely, that a single beat from a 1:1 sequence and the supported beat from a 1:2/1:3/1:4 sequence provide equivalent assistance to the patient, and thus that the latter sequences are simply a “diluted” (over time) form of 1:1, consisting of a supported beat equivalent to those found in 1:1 and a series of 1/2/3 unassisted beats. However, coronary autoregulatory controls and baroreceptor reflexes might respond differently to different assistance modes and evidence has emerged lately that full support is superior (5, 7). Nevertheless, a comparison between the benefits of isolated supported beats at different frequencies, and the effect of support frequency in conjunction with the effect of trigger timing have not been studied before.
The main aim of the present study was to investigate in healthy sheep the combined effect of varying assistance frequency and IAB inflation/deflation timing on coronary, aortic, and LV hemodynamics. We hypothesized that an assistance frequency of 1:1 will be associated with superior indices of hemodynamic function. We also hypothesized that non-conventional IAB trigger timings might yield better hemodynamic benefits compared to conventional timing.
Materials and Methods
Animal model
Acute animal studies were performed on six adult, Q-fevernegative, Dorset sheep (70 ± 15 kg, 4 male). This model was selected because its cardiovascular dimensions and baseline hemodynamic values are similar to those of human adults, and it has been successfully used for assessment of cardiac assist device performance in heart failure (8).
All animals received humane care in accordance with the Guide for Care and Use of Laboratory Animals (NIH publication 85–23, revised 1996). The protocol was approved by the Institutional Animal Care and Use Committee of the University of Louisville.
Animal preparation and instrumentation
Each pre-fasted animal was pre-medicated with xylazine (0.02–0.03 mg/kg, IM) before surgery. Anesthesia was induced with valium (0.2–0.4 mg/kg IV) and ketamine (4–8 mg/kg, IV) through the auricular vein. Following intubation, general anesthesia was maintained with isoflurane inhalation (0.5–3%) in 100% oxygen throughout the procedure, following standard protocol at the Cardiovascular Innovation Institute (CII). Vital signs were continuously monitored and hourly blood gas analysis guided ventilator management.
With the animal in the recumbent position, the right femoral vein and left femoral artery were exposed. A fluid-filled catheter was inserted into the right femoral vein and the left femoral artery was isolated for later insertion of the IAB catheter.
After repositioning in lateral recumbency, the heart, aortic arch, and descending thoracic aorta were exposed with a left lateral thoracotomy at the fourth intercostal space. A high fidelity 5 Fr dual-pressure-sensor catheter (Millar Instruments, Houston, TX, USA) was inserted into the ascending aorta and advanced retrograde across the aortic valve until the tip transducer was inside the LV and the proximal transducer in the ascending aorta. Transit-time ultrasonic perivascular flow probes (Transonic Systems, Ithaca, NY, USA) were securely fitted around the ascending aorta and the left anterior descending artery. A second high-fidelity 5 Fr pressure-sensor catheter (Millar Instruments) was inserted into the distal end of the aortic arch, with the transducer positioned where the IAB catheter tip was anticipated to extend.
Following an IV bolus injection of heparin (225 IU/kg), a Fidelity 9.5 Fr 40 cc IAB catheter (Datascope, Fairfield, NJ, USA) was introduced in the left femoral artery. Under fluoroscopy, the IAB catheter was advanced towards the aortic arch until its tip was about 2 cm distal to the subclavian artery, while radiopaque dye injection through the innominate trunk confirmed that the balloon was situated in its entirety in the descending thoracic aorta. The IAB catheter was connected to a Datascope 98XT IABP console (Datascope, Fairfield, NJ, USA).
Measurements and protocol
Aortic (Pao) and LV pressure (PLV), aortic (Qao) and coronary flow (Qcor), and the ECG were recorded at 500 Hz, as well as IAB catheter shuttle gas pressure (Pshuttle) (provided by the IABP console output). The aortic pressure waveform recorded close to the IAB tip guided IABP operation (9).
The animals were randomly divided into two groups: ‘1:1’ group (n = 3) and ‘1:3’ group (n = 3), receiving 1:1 and 1:3 IABP support, respectively. For each animal, the protocol consisted of a series of measurements during counterpulsation, followed by measurements with the IABP at standby (baseline), and was repeated three times for the 1:3 group and once for the 1:1 group. The counterpulsation measurements comprised a randomized sequence of approximately 30-second segments, each segment corresponding to different IABP inflation and deflation timing. Apart from conventional onset of inflation and deflation, earlier- and later-than-conventional onsets were produced through the IABP controls. In total 3 to 5 inflation and 3 to 5 deflation timings and various combinations between them were tested. The ventilator was turned off during data recording.
Data analysis
All data processing was done in Matlab (version R2011b, MathWorks, Natick, MA, USA). Pao and Qao were smoothed using a 3rd order Savitzky-Golay filter (10). Aortic flow velocity (Uao) was derived from Qao by dividing with the nominal area of the aortic flow probe. Hardware-induced time lags between Pao and Uao were corrected using the PU-loop method (11).
Two representative and non-consecutive assisted beats (assisted beats) were selected from each 30-second segment and from each baseline (control beats) for further analysis. For the 1:3 group the beats following the assisted beats (following beats) were also interrogated. Simultaneous inspection of Pao, Pshuttle, and ECG for each assisted beat enabled the quantification of onset of inflation and deflation, with respect to the preceding and the following R-wave, respectively. These times were normalized to the duration of the beat and grouped as early (E), late (L), and conventional (C) inflation and deflation. Five inflation-deflation trigger timing combinations were investigated further: C-C (CC), E-C (EC), L-C (LC), C-E (CE) and C-L (CL). Table I presents the mean normalized inflation and deflation times and the total number of analyzed beats for each of the 5 interventions.
Times and aortic pressure parameters
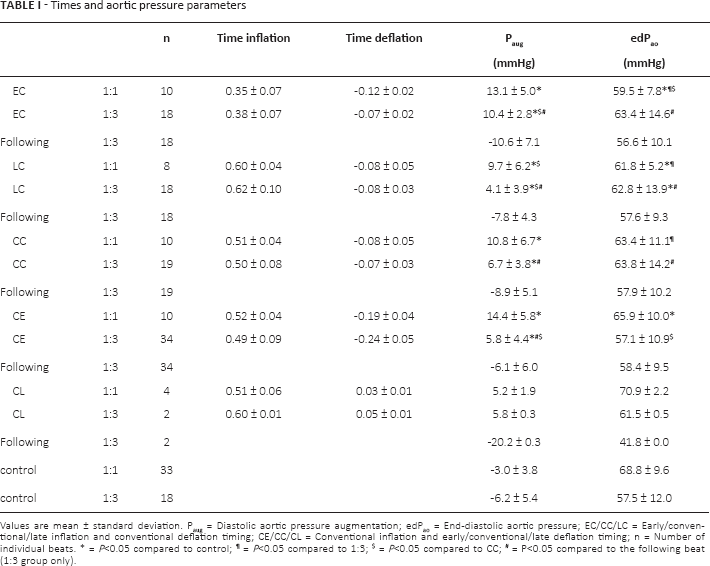
Values are mean ± standard deviation. Paug = Diastolic aortic pressure augmentation; edPao = End-diastolic aortic pressure; EC/CC/LC = Early/conventional/late inflation and conventional deflation timing; CE/CC/CL = Conventional inflation and early/conventional/late deflation timing; n = Number of individual beats.
= P<0.05 compared to control;
= P<0.05 compared to 1:3;
= P<0.05 compared to CC;
= P<0.05 compared to the following beat (1:3 group only).
For each analyzed beat, averages per beat as well as systolic (5) and diastolic (D) averages were derived for Pao, Qao, PLV, and Qcor, Diastolic aortic pressure augmentation (Paug) was determined by subtracting maximum systolic Pao from maximum diastolic Pao (Fig. 1B). End-diastolic Pao (ed Pao) was defined as Pao immediately prior to aortic valve opening (Fig. 1B). Stroke volume (SV) was calculated from Qao by integrating the positive flow from aortic valve opening until flow reversal at the end of systole (Fig. 1A). Integration of the subsequent negative aortic flow until flow reversal or until a second acceleration phase in negative flow (whichever occurred first), yielded a measure of initial aortic backflow surge (Fig. 1A).
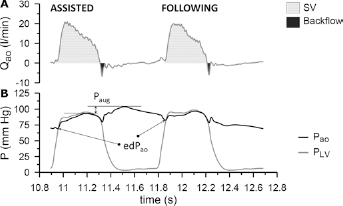
Hemodynamic signals for a beat with conventional IABP timing and its following beat at assistance frequency 1:3.
With reference to the 1:3 group, throughout this study we consistently use the term “assisted beat” to refer to a heart cycle during the diastole of which the IABP operates and the term “following beat” to refer to the beat that succeeds it. However, strictly speaking, during 1:3 support it is only the diastole of the assisted beat that is assisted (through inflation), since during the preceding systole the IABP was not operating yet. The effect of counterpulsation on primarily systolic parameters (such as PLV, SPLV, SV, and SQcor) will be therefore revealed on the following beat (through deflation). To reflect this and contrary to the strict use of the terms “assisted beat” and “following beat”, we reserve the terms “with IABP support” and “without IABP support” to be used without any presumption about their association to assisted or following beats.
Statistical analysis
All statistical analysis was performed in MLwiN (version 2.27, Centre for Multilevel Modelling, University of Bristol, Bristol, UK) (12). The values reported are mean ± standard deviation. For all parameters of interest, we compared: 1) values with IABP support and control values (separately for the 1:1 and 1:3 groups); 2) values with and without IABP support in successive beats (1:3 group only); 3) values with IABP support at CC with EC, LC and CE (separately for the 1:1 and 1:3 groups); and 4) values with IABP support between the 1:1 and 1:3 groups, after adjusting for possible baseline variations between the two groups. The statistical comparisons were performed with mixed model analysis (12). Statistical significance was assumed at P<0.05.
CL trigger timing, with late deflation defined as onset of deflation after the R-wave, was achieved only in half of the animals and no statistical testing could be performed.
Results
Hemodynamic signals
Figure 2 shows typical examples, from a single animal of the 1:3 group, of the Pao, Qao, PLV, and Qcor contours and their divergence between C, E, and L inflation (Fig. 2A) and between C, E and L deflation (Fig. 2B). The effect of the various trigger interventions is noticeable in the shape of the Pao waveform, which drastically changes according to the time diastolic pressure starts to increase and decrease due to IAB inflation and deflation, respectively. The Qcor signal also provides visual evidence of an increase in DQcor at all timing interventions. Differences in the PLV and Qao shapes between the different conditions are more subtle.
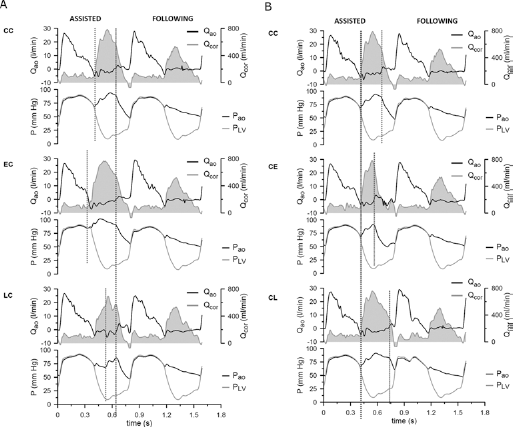
Hemodynamic data collected during 1:3 support for an assisted beat and its following beat for different IABP inflation (A) and deflation trigger timings (B). Top panels: Aortic flow (Qao, black) and coronary flow (Qcor, filled grey); Bottom panels: Aortic pressure (Pao, black) and left ventricular pressure (PLV, grey). EC/CC/LC: Early/conventional/late inflation and conventional deflation timing; CE/CC/CL: Conventional inflation and early/conventional/late deflation timing.
Hemodynamic parameters
Tables I and II summarize the mean values of the various hemodynamic parameters that were studied. A complete overview of the results of the statistical analysis is also provided in the tables.
There was no difference in Paug between the two assistance frequencies and a performance superior to CC was achieved only by EC at 1:3 (10.4 ± 2.8 mmHg [EC], 6.7 ± 3.8 mmHg [CC]). Support frequency 1:1 outperformed 1:3 at several timing combinations in terms of edPao reduction, and during 1:1 support EC provided lower edPao than CC (59.5 ± 7.8 mmHg [EC], 63.4 ± 11.1 mmHg [CC]). Assistance frequency 1:1 reduced SPLV more than 1:3 at all timing combinations except CE, and there was no trigger timing at 1:1 that provided lower SPLV than CC (69.1 ± 6.5 mmHg [CC], 76.4 ± 6.5 mmHg [control]). Findings for average per beat PLV were qualitatively identical to SPLV, while DPLV scarcely presented statistically significant changes, overall (Tab. II and Fig. 3A).
Flow and left ventricular pressure parameters
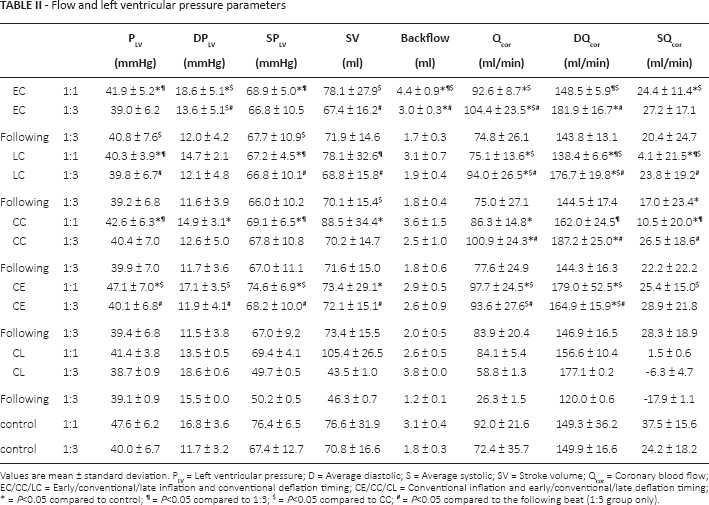
Values are mean ± standard deviation. PLV = Left ventricular pressure; D = Average diastolic; S = Average systolic; SV = Stroke volume; Qcor = Coronary blood flow; EC/CC/LC = Early/conventional/late inflation and conventional deflation timing; CE/CC/CL = Conventional inflation and early/conventional/late deflation timing;
= P<0.05 compared to control;
= P<0.05 compared to 1:3;
= P<0.05 compared to CC;
= P<0.05 compared to the following beat (1:3 group only).
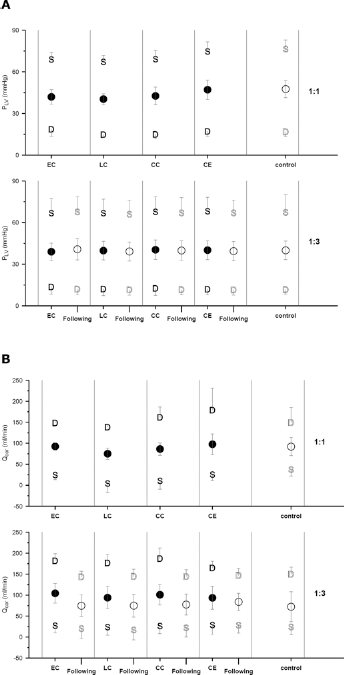
Average systolic (S), average diastolic (D) and average per beat (•) left ventricular pressure (PLV) (A) and coronary blood flow (Qcor) (B). The top panels correspond to the 1:1 group and the bottom panels to the 1:3 group. Data are presented as overall means for the various IABP trigger timings, for the corresponding following beats (1:3 group only) and for the control beats. The error bars represent standard deviations.
An increase in stroke volume compared to control took place only during 1:1 support at CC, although 1:3 support did show some efficiency compared to values without IABP. No trigger timing provided greater increase in SV than CC at 1:1 (88.5 ± 34.4 ml [CC], 76.6 ± 31.9 ml [control]). Backflow increased significantly with both support frequencies at EC compared to control and to the following beat, but this increase was significantly higher with 1:3 (67% [1:3], 42% [1:1]). 1:3 showed superior efficiency over 1:1 in terms of DQcor augmentation (except for CE) and at 1:3 no trigger timing produced higher DQcor than CC (187.2 ± 25.0 ml/min [CC], 149.9 ± 16.6 ml/min [control]). The results for Qcor are similar to those of DQcor, with small differences due to variations in SQcor (Tab. II, Fig. 3B).
Discussion
Summary of findings
The effect of varying IABP assistance frequency and trigger timing on various quantities derived from pressure and flow measurements in the ascending aorta of healthy sheep was investigated. Paug was enhanced by both assistance frequencies alike. Assistance frequency 1:1 was more beneficial in reducing edPao and SPLV and increasing SV, while frequency 1:3 was more beneficial in increasing DQcor. The trigger timings that resulted in best IABP performance at the above frequencies were EC and CC for Paug (at frequency 1:3 and 1:1, respectively), EC for edPao, and CC for SPLV, SV and DQcor.
Hemodynamic effects of assistance frequency and timing
The most commonly quoted benefit of IAB inflation is Paug, leading to increased DQcor in non-stenotic or moderately stenotic coronary vessels, due to the increased aortic-coronary pressure gradient (1, 13–15). Despite enhanced diastolic Pao always achieved with IABP support in our healthy animal population (Tab. I), this increase was not always accompanied by an increase in DQcor. It is likely that the native autoregulatory mechanisms that govern myocardial perfusion precluded a simple linear relationship between Paug and DQcor (16).
There was no timing combination that yielded both Paug and DQcor better than CC at a single assistance frequency in this study. During 1:3 support EC timing improved Paug compared to CC, while DQcor was augmented more at CC. During 1:1 support CC timing was optimal for Paug, whereas DQcor was increased more with CE timing. Despite the apparent benefit of EC timing on Paug, with early inflation starting while the aortic valve is still open, the IABP may displace blood directly into the LV, interrupting the phase of late ejection. During the systole that immediately precedes early IAB inflation at 1:3 support, a reduction in SV from 70.8 ± 16.6 ml at control down to 67.4 ± 16.2 ml (P<0.0005), directly followed by increased backflow from 1.8 ± 0.3 ml at control up to 3.0 ± 0.3 ml (P<0.0001), provide further evidence of a detrimental effect of early inflation on LV mechanics. The beneficial effect of CE timing on DQcor indicates that early deflation did not lead to coronary blood flow stealing in our experiments.
Although DQcor overall benefited more by 1:3, this frequency enhances DQcor only every third beat, whereas 1:1 increases myocardial perfusion every beat. Therefore, during 1:1 support the DQcor enhancement provided by both CC (13 ml/min) and CE (30 ml/min) could match or significantly exceed, respectively, the benefit of 1:3 (37 ml/min) (Tab. II).
A decrease in myocardial oxygen demand, generated through edPao reduction and thus reduction in afterload, is a widely reported outcome of IAB deflation (2). A statistically significant reduction in edPao with IABP support was observed under most timing interventions, but only at assistance frequency 1:1 (Tab. I). A resulting drop in SPLV, indicating a decrease in myocardial metabolic demand, was also observed at 1:1 but not 1:3 (Fig. 3A). This superiority of 1:1 was confirmed statistically at several triggering timings for both edPao and SPLV, with 1:3 either not affecting or even stimulating increases in these parameters. This apparent inferiority of 1:3 could be due to population differences between the 1:1 and 1:3 groups (such as baseline Pao).
Clinical implications
All hemodynamic findings from the animal cohort that we studied indicate that there is no single assistance frequency that shows superiority to other frequencies by improving uniformly all investigated hemodynamic parameters. For instance, DQcor appeared to benefit more by 1:3 support, while 1:1 support was more beneficial for parameters related to afterload reduction and myocardial metabolic demand. Furthermore, even though there were unconventional IABP trigger timings that benefited some hemodynamic parameters more than CC, there was not a single timing that demonstrated superiority to CC across all parameters. Hence, despite not maximizing the benefit for all hemodynamic parameters, CC could nonetheless still be the favored choice for balanced support.
However, the improved performance of some unconventional trigger timings compared to CC warrants further investigation. CC timing appears to have been established early on, based on theoretical indications and lack of evidence against it, and has never been challenged through extensive and thorough testing. Our study provides indication that CC timing might well not be the ultimate timing mode for every patient. For example, a patient receiving IABP support as a prophylactic measure during a high-risk coronary procedure might benefit more by inflation (enhancement in coronary blood flow), while a patient in cardiogenic shock might benefit more by deflation (reduction in afterload). Our study has provided evidence that the optimal timing for the above 2 possible patient groups might be different and CC might not be optimal for either of them, calling altogether for open-mindedness about investigating alternatives to the established timing. It thus promotes the idea of personalized rather than generalized patient therapy for the achievement of higher IABP therapeutic efficiency, with a choice of inflation/deflation timing that prioritizes the recovery of those hemodynamic indices that are more in need of support in the unassisted circulation.
In current practice, both frequency weaning and volume weaning are legitimate options for withdrawal from full 1:1 IABP support, prescribing a gradual reduction in support frequency and IAB augmentation volume, respectively (17, 18). In this work, a large number of parameters benefited more by 1:1 than 1:3, in agreement with other studies (7), suggesting that the support provided per assisted cardiac cycle during 1:3 does not meet the therapeutic effects provided during 1:1. Further investigation is warranted to explore whether the above findings will evolve into a clear advantage of volume weaning within the context of a clinical trial.
The onset of inflation and deflation with respect to other events during the cardiac cycle are unquestionably critical for IABP performance, but the resulting duration of inflation could also be of relevance. Trigger timing with onset of inflation or deflation that are considered unsafe according to the current practise, for example EC and CL, might still yield positive outcomes because they accommodate longer duration of inflation compared to CC. Likewise, trigger timings that result in shortened duration of inflation compared to CC, such as LC and CE, might lead to adverse outcomes despite generally considered not harmful.
The clinical significance of small (but statistically significant) hemodynamic changes induced by the IABP warrants further investigation.
Limitations
The critically ill patients supported with the IABP are typically old, with impaired cardiac function, irregular heart rate, compromised coronary autoregulatory mechanisms, and atherosclerotic vessels. These pathophysiological abnormalities are not replicated by our animal model and may have an impact on the performance of the IABP. This choice was made in order to avoid introducing pathophysiological variability in a small animal cohort.
The most desired outcome of IABP support is a reduction in oxygen demand coupled with improved oxygen delivery. The impact of our study would therefore be enhanced with measures of myocardial oxygen supply and demand during frequency and timing manipulations.
A large number of statistical tests were conducted in the present study (22 for each variable of interest) and no P-value adjustment for multiple testing was made. Multiple comparisons can result in an increased risk of type I errors, but on the other hand P-value adjustments increase the chance of making type II errors. Because of the exploratory nature of the present study, the possibility of investigating leads that might turn out to be false due to type I errors was deemed preferable to potentially missing important findings due to type II errors.
The present work does not provide definitive clinical recommendations for alternative timings that might deliver better hemodynamic support than CC, since we tested a small number of animals that were healthy, as we have already acknowledged. However, our results suggest that based on the investigated parameters, the currently established trigger timing does not appear to be optimal and further investigation is encouraged.
Conclusions
No single IABP assistance frequency showed superiority to other frequencies by uniformly benefiting all investigated hemodynamic parameters. Likewise, conventional trigger timing did not aid evenly all investigated parameters more than unconventional trigger timings. These findings suggest that a choice of optimal frequency and timing might need to be made based on individualized hemodynamic needs rather than as a generalized protocol.
Footnotes
Acknowledgements
The authors would like to thank Datascope Corporation (currently Maquet Cardiac Assist) for making the data available to AWK. We also acknowledge Dr. Steven Koenig and the research staff of the CM.
Financial Support: CK is supported by the British Heart Foundation (PG/12/73/29730).
Conflict of interest: GMP was sponsored by Datascope Corporation (currently Maquet Cardiac Assist) for the animal experiments at the CII.
