Abstract
Background
Despite advances in management and care, Duchenne muscular dystrophy (DMD) remains universally fatal. The objective of this study was to estimate the mortality cost of DMD.
Methods
We estimated the mean total national annual mortality cost associated with DMD by simulating the mean number of patients who would have been alive in 2012 in the absence of DMD using data on the mean DMD incidence, mean number of live male births, mean life expectancy at birth in DMD and the male general population, and a societal willingness-to-pay (WTP) for a life-year. We attributed each patient who would have been alive in 2012 a lost life-year. Finally, to estimate the mean mortality cost of DMD, we multiplied the estimated mean number of life-years lost with a societal WTP for a life-year of €75,000.
Results
The mean total number of patients who would have been alive in 2012 in the absence of DMD was estimated at 4470 (95% bootstrapped CI: 4449-4492) in Germany, 3313 (3297-3329) in Italy, 3564 (3547-3581) in the UK, and 16,105 (16,029-16,186) in the USA. The corresponding mean mortality cost (in millions) was estimated at €335 (€334-€337) for Germany, €248 (€247-€250) for Italy, €267 (€266-€269) for the UK, and €1,208 (€1,202-€1,214) for the USA.
Conclusions
We show that DMD is associated with a considerable mortality cost and a substantial total economic burden to society. Our findings serve as important intelligence input to health economic policy decisions, allocation of funds for research, and cost-effective care delivery systems.
Introduction
Although most would agree that it is not possible to quantify the value of a life in monetary terms, policy-makers rely on assumptions of society's willingness-to-pay (WTP) for a life or life-year to inform decisions regarding allocation of public funds. Examples include health economic evaluations of new expensive drugs shown to prolong life expectancy or whether or not to implement a screening programme at the population level for early detection of a rare, life-threating illness. The societal WTP for a life-year varies between settings and jurisdictions but in health economic evaluations, cited values for a life-year in full health often lie between approx. €50,000 and €100,000 (1).
Duchenne muscular dystrophy (DMD) is a rare, X-linked, neuromuscular disease characterised by progressive muscle degeneration and serious, multi-system complications (2, 3). Despite advances in management and care, and the prospect of new therapies, DMD remains universally fatal and few patients live beyond their third decade (4).
We have previously reported the economic burden of DMD in Germany, Italy, the UK, and the USA, but that study excluded the cost to society associated with premature death (5). The objective of this study was to estimate the annual mortality cost of DMD.
Methods
To estimate the mean annual mortality cost of DMD in Germany, Italy, the UK, and the USA in 2012, a year for which we previously have estimated the DMD cost of illness in a cross-sectional observational study (5), we simulated the mean number of patients who would have been alive in 2012 in the absence of DMD. Each patient who would have been alive was assigned a lost life-year, and the country-specific mean total number of life-years lost due to DMD was subsequently calculated. Finally, to estimate the mean mortality cost, we multiplied the estimated mean number of life-years lost with a societal WTP for a life-year.
In detail, our simulation involved five data components: (i) the mean incidence of DMD, estimated at between one in 3802-6291 live male births (6), (ii) the country-specific number of live male births (per year, 1940 through 2012) (7, 8), (iii) the mean DMD life expectancy at birth, estimated at 14.4 years (95% CI: 11.9-16.8) for patients born in the 1960s, 19.3 years (18.6-20.0) for the 1970s and 1980s, and 25.3 years (23.1-26.6) for the 1990s (4), (iv) the country-specific mean life expectancy at birth in the male general population (per year, 1940 through 2012) (7–9), and (v) a societal WTP for a life-year. We collated available data on DMD incidence (total number of subjects screened and number of identified cases) from previous research (6) and estimated the pooled incidence at one in 5021 live male births (proportion: 0.0002; 95% CI: 0-0.0013). In the absence of data, we extrapolated the mean DMD life expectancy in the 1960s to the 1950s and 1940s, and the DMD life expectancy in the 1990s to the 2000s and 2010s. In accordance with the central limit theorem, we assumed that the sampling distribution of mean incidence of DMD, mean DMD life expectancy, and mean life expectancy in the male general population followed a normal distribution (standard errors were derived from published confidence intervals).
The simulation encompassed 1000 iterations for each country and year (1940 through 2012). In each iteration, we simulated 1000 point estimates representing the mean total contribution to the number of life-years lost in 2012 due to DMD (based on the simulated number of new DMD cases, the simulated DMD life expectancy, and the simulated general population life expectancy) (243,000 simulated data points in total per country). Specifically, for each country and year, we first simulated the total number of new DMD cases using data on the total number of live male births and the incidence of DMD. Next, we simulated the number of these cases that would have been alive in 2012 in the absence of disease using DMD and general population mortality data. Next, to estimate the total mortality cost of DMD, we multiplied the estimated mean number of life-years lost with a societal WTP for a life-year of €75,000 [in-between the two most commonly cited estimates for a life-year in full health (1)]. We explored a WTP of €50,000 and €100,000, respectively, in sensitivity analysis. We ran a set of test simulations varying the number of iterations for different parameters and outcomes (e.g., number of new DMD cases) and then compared the means of the sampled mean values from these outputs and found that 1000 iterations were sufficient to capture the underlying variability.
The simulations were carried out using Visual Basic for Applications (Microsoft) and the statistical analyses using Stata 12 (StataCorp).
Results
The mean number of life-years lost due to DMD in 2012 is presented in Table I. On average, in Germany, the UK, and the USA, patients born between 1948 and 1989 contributed to the mean number of life-years lost in 2012. The corresponding interval in Italy was 1950 to 1989. Earlier birth cohorts (e.g., those born before 1948 in the UK) would have died before 2012 in accordance with the mean general population life expectancy, and later cohorts (e.g., those born after 1989 in the UK) would still be alive in 2012 following the mean DMD life expectancy.
Contribution to the mean number of life-years lost due to Duchenne muscular dystrophy (DMD) in 2012, by country and decade
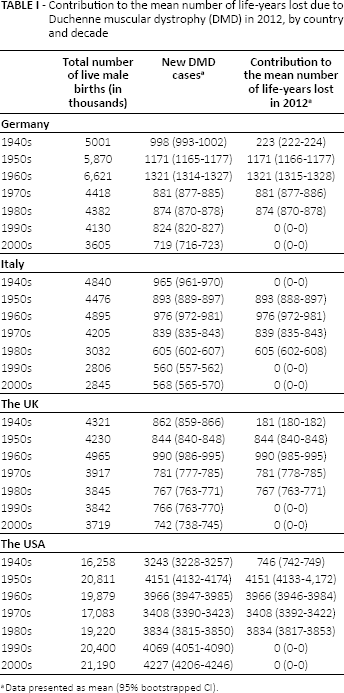
Data presented as mean (95% bootstrapped CI).
Adding the mortality cost of DMD (Tab. II) to our previously published results on the burden of illness (5), the mean annual total societal economic burden of DMD was estimated at €613 million (95% bootstrapped CI: €591-€640 million) for Germany, €403 million (€385-€420 million) for Italy, €468 million (€450-€491 million) for the UK, and €2425 million (€2330-€2525 million) for the USA. Per patient, based on our previous estimates, this corresponds to a mean total burden of €240,000, €209,000, €278,000, and €241,000, respectively. The different cost components of the burden of DMD are presented in Figure 1. Using a WTP for a life-year of €50,000, the mean annual total societal economic burden of DMD was estimated at between €166 million and €805 million, and using a WTP of €100,000 between €331 million and €1611 million, across the four countries.
The mortality cost of Duchenne muscular dystrophy (DMD) in 2012
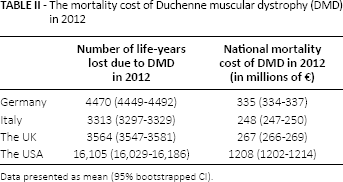
Data presented as mean (95% bootstrapped CI).
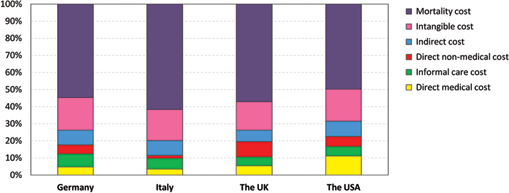
Components of the total economic burden of Duchenne muscular dystrophy (DMD). Note: Direct medical costs include DMD-related hospital admissions, visits to physicians and other healthcare professionals, medical tests and assessments, medications, and emergency and respite care. Informal care costs include paid and unpaid informal care by the primary caregiver. Direct non-medical costs include non-medical aids, devices, and investments (including investments and reconstructions of the home), as well as non-medical community services (e.g., transportation services and home help). Indirect costs include production losses for the patient and primary caregiver, as well as productivity losses due to absenteeism and impaired productivity while working. Intangible costs (costs due to pain, anxiety, social handicap, etc.) include the monetary value of the loss in quality of life for the patient and primary caregiver valued at approx. €75,000 per QALY (5).
Discussion
The objective of this study was to estimate the annual mortality cost of DMD, a rare, severely disabling neuromuscular illness. Despite substantial improvements in survival in recent decades, mortality in DMD remains high, and although several novel therapeutic modalities are currently being tested in trials, the trajectory to better prognosis appears to be long. In addition, poor access and compliance to adequate DMD care, as described in our previous work (10), may compromise treatment outcomes for many patient groups, in particular in developing countries, resulting in further reduced life expectancy.
Across the studied countries, we estimated the mean total national annual mortality cost of DMD at between €248 million and €1208 million, between 50% and 62% of our previous estimates of the total burden of illness (5). Thus, the cost associated with excess mortality constitutes the largest cost component of the disease burden, followed by costs associated with impaired quality of life.
To the best of our knowledge, this study is the first to estimate the mortality cost of any disease using societal WTP estimates for a life-year as utilised by decision-makers in cost-effectiveness analyses and health economic evaluations. The lack of previous research could be related to the fact that most cost-effectiveness evaluations are based on mathematical disease models in which mortality is explicitly incorporated and where life-years gained is a primary model outcome. For this reason, to estimate the cost of premature death separately may have been regarded as redundant as these estimates cannot be populated directly into the typical cost-effectiveness model framework. Many cost of illness studies instead regard the mortality cost as an indirect cost, valued at the loss in production to society in accordance with the Human Capital Approach. However, this approach leads to an underestimation since it fails to account for the fact that society puts an explicit value on the life-year itself (through the practice of employing WTP thresholds in cost-effectiveness evaluations), irrespective of the loss in contribution to production, or other costs.
It should be emphasised that the estimates generated in this study constitute the mortality costs as valued by health policy decision-makers in the standard health technology assessment framework. Our estimates thus complement our previous work (5) on the cost of DMD, which included direct medical costs (e.g., DMD-related hospital admissions, visits to physicians and other healthcare professionals, medical tests and assessments, medications, and emergency and respite care), informal care costs (i.e., paid and unpaid informal care by the primary caregiver), direct non-medical costs (i.e., costs associated with non-medical aids, devices, and investments), non-medical community services (e.g., transportation services and home help), indirect costs (i.e., production losses for the patient and primary caregiver, as well as productivity losses due to absenteeism and impaired productivity while working), and intangible costs (i.e., costs due to impaired quality of life) estimated for patients who were alive at different ages and stages of disease.
Cost of illness studies are important as they can help inform policy-decision at both the micro level (e.g., cost-effectiveness evaluations of new interventions) and the macro level (e.g., allocation of funds for research and prioritisation of diseases to target with preventive interventions) (11, 12). For diseases with no or low excess mortality, recordings of costs accrued for patients who are alive constitute a satisfactory estimate of their economic burden to society. However, for disease with high mortality, such as DMD, despite a considerable per-patient burden, the total aggregated national cost will be relatively low due to the low disease prevalence. Put differently, because deceased patients do not accrue costs, diseases with high mortality will be regarded as less costly to society, although the life-years lost are attributed considerable value. Accordingly, to allow for meaningful comparison of costs across diseases, it is important to also consider mortality costs in the burden analysis to inform optimum policy.
An alternative method to estimate the number of deaths due to a disease in a specific year would be to conduct a registry study using, e.g., causes of death data. However, for DMD, this approach is not feasible because of the fact that DMD does not have a unique classification code (in e.g., ICD-9 or ICD-10). It is, as a consequence, not possible to identify DMD patients within the standard registry framework, underscoring the utility of disease-specific registries, such as the TREAT-NMD (13), for DMD research.
The main limitation of our study concerns the lack of country-specific DMD incidence data. Previous studies report an incidence between one in 3802 and 6291 live male births based on research from the USA, New Zealand, the UK, Germany, Canada, France, Cyprus and Belgium (6). In addition to possible geographical variations, the heterogeneity in published rates could be attributed to differences in diagnostic testing methods, as well as other factors relating to study design and sample size. In this study, we used a pooled incidence estimate derived from previously published data, and consequently it is uncertain to which settings this estimate is externally valid. However, given that we incorporated the full variability around the mean DMD incidence observed in previous research in our simulation, the impact of the data limitation on the robustness of our results is expected to be minor. However, observed or unexplained heterogeneity attributed to characteristics of patients or study design may still bias our findings.
Moreover, in absence of data, we also applied UK DMD mortality estimates to Germany, Italy, and the USA, which resulted in an under/over estimation of morality costs given lower/higher survival in these counties from the 1960s and onwards.
In summary, we have shown that DMD is associated with a considerable mortality cost and a substantial total economic burden to society. Our results represent the final piece of information to the description of the previously unknown economic context of a rare neuromuscular disease such as DMD and serve as important intelligence input to health economic policy decisions, allocation of funds for research, and cost-effective care delivery systems.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
