Abstract
Purpose
This study compared the efficacy and safety of hyaluronic acid (HA)-trehalose, a new eyedrop containing trehalose (a natural bioprotectant) and HA, to an established formulation containing only HA.
Methods
This was a phase III, randomized, active-controlled, investigator-masked, multicenter study in France and Tunisia. In all, 105 adult patients (≥18 years) with moderate to severe dry eye disease (DED) received either HA-trehalose (n = 52) or HA (n = 53) 3-6 times per day for 84 days. The primary efficacy variable was the Oxford grading score at day 35. A questionnaire on dry eye and symptoms, Schirmer test, tear break-up time, conjunctival hyperemia, and global performance were assessed as secondary efficacy criteria at baseline, day 35, and day 84. Safety assessments were standard.
Results
Noninferiority of HA-trehalose to HA for keratoconjunctivitis sicca assessed by Oxford grading score was demonstrated at day 35. For the secondary efficacy parameters, reductions in dry eye questionnaire classes of none or mild at day 84, dry eye symptoms of stinging, itching, and blurred vision at day 35, and investigator (days 35 and 84) and patient assessments (day 35) of global performance were significantly better for HA-trehalose. There were no clinically meaningful differences between groups for the other secondary criteria. Both treatments were well-tolerated, and there were fewer ocular symptoms upon instillation and fewer adverse events for HA-trehalose than for HA.
Conclusions
Hyaluronic acid-trehalose is effective and safe, with better patient satisfaction, than existing HA-only eyedrops particularly from the first month of treatment, and offers a therapeutic advancement in the treatment of moderate to severe DED.
Introduction
Dry eye disease (DED) is characterized by symptoms of ocular discomfort, visual disturbance (e.g., blurry or foggy vision that clears temporarily on blinking), and tear film instability that is often associated with tear film hyperosmolarity and can lead to damage of the ocular surface. It has a multifactorial etiology, including insufficient tear production and/or excessive tear evaporation linked to multiple conditions: aging, menopause, Meibomian gland deficiency, Sjögren syndrome, conjunctival fibrotic disease, and disturbance of the ocular surface by systemic or topical medications (1, 2). Insufficient tear production or excessive evaporation can lead to tear hyperosmolarity, which can trigger a chain of self-perpetuating events that constitute a vicious circle of DED (3). In this cycle, tear hyperosmolarity can result in cell morphologic changes that lead to inflammatory cascades and further cell death, leading to further tear film instability and tear hyperosmolarity, which continues the circle (3, 4).
Current management of DED is largely by the suppression of contributing factors (e.g., medications) and prescription of a wide range of artificial tears (eyedrops, fluid solutions, or ophthalmic gels). Sodium hyaluronate solutions are considered the gold standard artificial tear treatment (5–6–7).
However, the recent introduction of trehalose, a natural bioprotectant, which promotes bioprotection of ocular epithelial cells against inflammation and apoptosis triggered by osmotic stress (3, 8–9–10–11–12), can help to manage tear hyperosmolarity and so promote an exit from the vicious circle of DED physiopathology described earlier. Trehalose has been used for many years in therapeutic products and in medical applications such as cryopreservation and organ transplant (13), and as a bioprotectant it has an important role in anhydrobiosis. It helps to restore osmotic balance to the ocular surface (8, 10) as well as preventing denaturation of cell membrane lipid bilayers and proteins and activating autophagy (14–15–16).
A new eyedrop formulation that contains both sodium hyaluronate and trehalose (HA-trehalose) has been developed to capitalize on the bioprotectant properties of trehalose. This new formulation is preservative-free and available in a multidose presentation, allowing frequent use for extended periods without causing harm to the ocular surface (17).
This study was therefore performed to compare the efficacy of HA-trehalose vs a commonly used HA ophthalmic solution in terms of deterioration in the ocular surface integrity caused by keratoconjunctivitis sicca assessed by the Oxford grading score (18), symptom relief, and other standard ophthalmic parameters in patients with moderate to severe DED, as well as to assess its tolerability following frequent administration for 35 and 84 days.
Methods
Study design and participants
This was a phase III, randomized, active-controlled, investigator-masked, multicenter study with a run-in period, conducted in France (14 active centers) and Tunisia (4 centers), comparing HA-trehalose and HA eyedrops (ClinicalTrials.gov identifier: NCT02023268). The study was conducted in accordance with Good Clinical Practice and applicable International Conference on Harmonisation and the European directive 2001/20/CE guidance documents and practices, and in compliance with the ethical principles of the Declaration of Helsinki (2004) and local regulations (including independent ethics committee approvals before starting the study). Prior to enrollment, written informed consent was obtained from each participant. The study took place between October 2013 and December 2014.
Male or female patients were aged ≥18 years and had moderate to severe DED, defined by an Ocular Surface Disease Index (OSDI) score ≥18 (19) and with either Schirmer test ≥3 mm/5 min and ≤9 mm/5 min or the sum of 3 tear break-up times (TBUT) ≤30 seconds in at least one eye. Presence of a notable dry eye-related keratoconjunctivitis was required (corneal and conjunctival ocular staining by the Oxford grading scheme ≥4 and ≤9). Dry eye disease was required not to be controlled by conventional tear substitution treatment within the 3 months before selection. The main study-specific exclusion criteria were best-corrected far visual acuity <1/10; severe blepharitis; eyelid malposition, corneal dystrophy, ocular neoplasia, filamentous keratitis, corneal neovascularization, or orbital radiotherapy; and ocular trauma, ocular infection, ocular inflammation, ocular allergy, or ocular herpes in the preceding 3 months. The following treatments were prohibited: cataract or corneal surgery (from 12 months prior to and during the study); isotretinoin, calcineurin inhibitors; lachrymal plugs; any change in systemic medication; and contact lenses. Any nonstudy ocular medications were prohibited during the run-in and study.
Between selection and inclusion (day 0), patients were only allowed to use Hydrabak® (preservative-free 0.9% sodium chloride artificial tears) as ocular treatment. No instillation during the 6 hours preceding inclusion was allowed. At inclusion, patients were randomly assigned to receive either the bioprotective trehalose (3%) solution (HA-trehalose; Thealoz Duo®/Théalose®, Laboratoires Théa, Clermont-Ferrand, France) or the sodium hyaluronate solution (HA; Vismed®, Horus Pharma, Saint-Laurent-du-Var, France). Each patient was instructed to instill 1 drop in each eye 3 to 6 times daily for 84 days, was to be contacted by telephone on day 7, and was to visit the study site on days 35 and 84.
Efficacy assessments
Primary performance variable: Oxford grading score
The Oxford 0-15 grading score (18) was used at days 0 (baseline), 35, and 84 to assess global ocular staining in the corneal area by fluorescein coloration (keratitis severity) and in the temporal and nasal conjunctival areas by lissamine green coloration (severity of conjunctival impairment). The assessment in the eye with the worse score at day 0 was used in the analysis (if the same in both eyes, the eye with the worse Schirmer score was used; if the same, the eye with the worse TBUT score was used; if the same, the right eye was used).
Secondary performance variables
The OSDI (19), Schirmer test, TBUT (recorded as the mean of 3 measurements made the first minute after instillation of 1 drop of fluorescein), conjunctival hyperemia (using the McMonnies scale (20)), and global ocular fluorescein staining using the van Bijsterveld score (21, 22) were assessed at days 0 (baseline), 35, and 84. The severity of dry eye symptoms (ocular burning, stinging, itching, dryness feeling, foreign body sensation, light sensitivity, blurred vision, and pain) was recorded at days 0, 7, 35, and 84, and a global efficacy assessment was done by both the patient and the investigator at days 35 and 84 (evaluation of the product as very satisfactory, satisfactory, not very satisfactory, or unsatisfactory).
Safety assessments
Ocular symptoms (irritation/burning/stinging, photophobia, foreign body sensation, eye dryness feeling, pain, and other symptoms) and global tolerability were assessed at days 35 and 84. Ocular symptoms were assessed by asking if any sensation was felt upon instillation; if yes, an assessment of duration and severity was completed. Global tolerability was assessed by the investigator as very satisfactory, satisfactory, not very satisfactory, or unsatisfactory.
Adverse event (AE) data were collected at days 0, 7, 35, and 84. All AEs observed by the investigator or reported by patients were recorded, along with their severity and potential relationship to study treatment.
Best-corrected visual acuity (BCVA) was measured at the screening visit (baseline) and days 35 and 84 with the patient's best correction using a Snellen chart.
Statistical analyses
The primary objective of this study was to demonstrate the noninferiority of HA-trehalose to HA in terms of the change from baseline at day 35 of global ocular staining according to the Oxford grading score. The noninferiority test was based on the bilateral 95% confidence interval (CI) for the difference between groups (HA-trehalose minus HA). A 2-way analysis of covariance (ANCOVA) model was constructed using main effects of product (HA-trehalose or HA) and baseline score as covariates. The 95% CI was computed for the difference and if the upper bound was no higher than 2 points it was to be concluded that HA-trehalose was noninferior to HA. The estimations of the noninferiority limit and standard deviations were based on previous published studies.
A total of 76 patients (38 per group) would have provided at least 90% power for the noninferiority calculation on a 1-sided 2-sample t test with α 2.5%. An estimated attrition rate of 15% meant that it was planned to enroll a total of 90 patients.
Other quantitative variables were analyzed using the same ANCOVA model for group comparison. Ordinal variables were analyzed using a 2-tailed Wilcoxon signed-ranked test or chi-square test. Missing data were handled using a last available assessment carried forward approach. Comparisons were performed 2-sided at the 5% α level of significance.
A subanalysis for OSDI was performed at day 35 and day 84 for the percentage of patients with scores 0-13, 14-23, 24-33, and 34-100, corresponding to OSDI classes of none, mild, moderate, and severe, respectively (23). Another subanalysis was performed at day 35 and day 84 to compare the evolution (improvement, no improvement) of each ocular symptom (burning, pain, stinging, itching, dryness feeling, foreign body sensation, light sensitivity, and blurred vision) between groups.
Efficacy analyses were performed in the full analysis set (FAS; all randomized patients who received at least 1 dose and had at least 1 postbaseline efficacy evaluation) and per protocol set (PPS; patients in the FAS who had no major protocol deviation) used as the main population for the primary efficacy evaluation. The safety population (used for safety evaluations) was defined as all randomized patients who received at least 1 dose.
Results
Participants studied
Of 112 patients initially selected, 105 patients were included at day 0 and randomized to receive HA-trehalose (n = 52) or HA (n = 53) (Fig. 1). Of these, 52 and 49 patients completed the study, with 46 and 45 patients being included in the PPS.
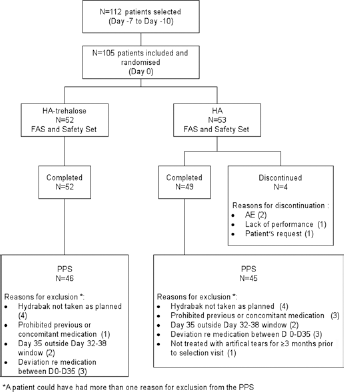
Patient disposition.
Demographic and baseline (day 0) assessments are presented in Table I. More female than male patients were recruited in both groups, and both groups were similar in terms of age. The time since DED diagnosis, DED etiologies, and baseline Oxford score, OSDI score, total ocular symptom score, van Bijsterveld score, Schirmer score, and TBUT were similar in both groups. Additionally, ocular medical and surgical histories were similar in each group, the most common being cataracts (14.3% overall) and lens extraction (16.2% overall), respectively. Systemic medical and surgical history was also similar in each group, with no clinically important differences.
Demographic and baseline (day 0) assessments (full analysis set)
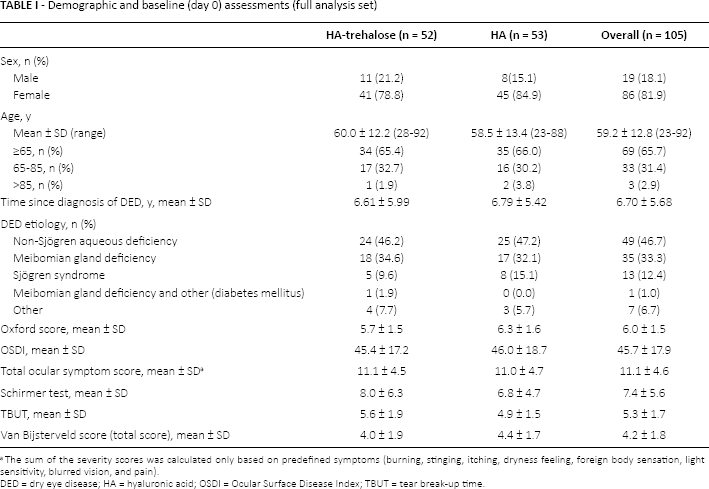
DED = dry eye disease; HA = hyaluronic acid; OSDI = Ocular Surface Disease Index; TBUT = tear break-up time.
The sum of the severity scores was calculated only based on predefined symptoms (burning, stinging, itching, dryness feeling, foreign body sensation, light sensitivity, blurred vision, and pain).
The mean number of daily instillations per patient for days 0-35 and days 35-84, respectively, was 4.0 ± 1.0 and 3.9 ± 0.9 for the HA-trehalose group and 3.9 ± 0.9 and 4.0 ± 0.9 for the HA group.
Efficacy
For the HA-trehalose and HA groups, the mean reduction in global Oxford score from day 0 to 35 was -2.5 ± 2.0 and -2.7 ± 1.7, respectively, in the PPS (Tab. II) (data supported by FAS analysis). Noninferiority of HA-trehalose to HA was demonstrated at day 35 as the upper bound of the 95% CI of the difference between groups (0.79) and did not exceed the noninferiority margin of 2 points. The score continued to decrease to day 84 (Tab. II), with reductions being similar for HA-trehalose and HA from day 0 to day 84 (-4.0 ± 2.2 and -3.9 ± 2.3). For the separate areas (temporal bulbar conjunctival staining, corneal staining, and nasal bulbar conjunctival staining), there were no group differences in Oxford score (reductions in corneal staining from baseline were -0.8 and -1.0 at day 35 and -1.3 and -1.2 at day 84, for HA-trehalose and HA, respectively).
Oxford score (global ocular staining 0-15) in the worse eye at day 35 (primary efficacy criterion) and day 84 (per protocol set)
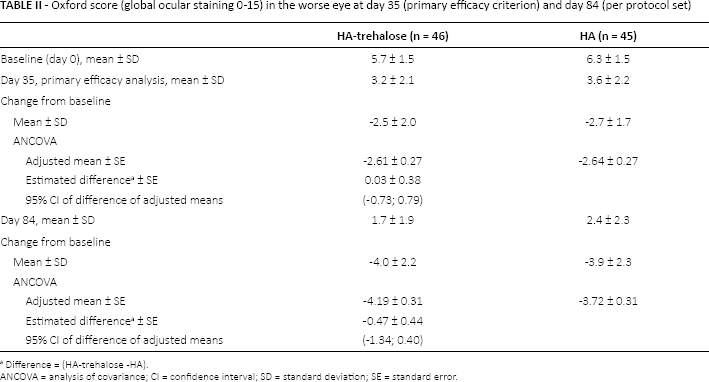
ANCOVA = analysis of covariance; CI = confidence interval; SD = standard deviation; SE = standard error.
Difference = (HA-trehalose -HA).
The OSDI was reduced from baseline in both groups at days 35 and 84 (FAS), with a trend for a greater effect in the HA-trehalose group vs the HA group at day 84 (day 35: -20.6 ± 17.2 vs -17.6 ± 16.2, p = 0.240; day 84: -30.2 ± 18.7 vs -25.6 ± 19.5, p = 0.060). The subanalysis that grouped patients based on OSDI class (Fig. 2) shows a significantly increased number of patients with an OSDI class of none or mild in both groups through the study, with this effect being more pronounced for the HA-trehalose group (p = 0.044 at day 84).
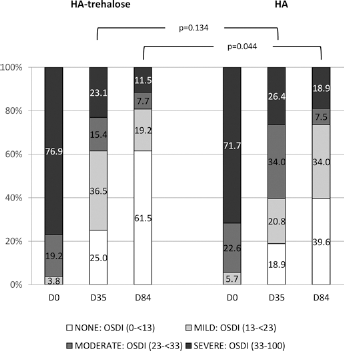
Percentage of patients in each Ocular Surface Disease Index class (full analysis set).
The reductions in severity for each symptom at day 35 are presented in Figure 3. A statistically significant better improvement was achieved at day 35 with HA-trehalose for 2 symptoms of ocular discomfort linked to dry eye, i.e., stinging (p = 0.024) and itching (0.012), and for one symptom of visual disturbance, i.e., blurred vision (p = 0.024). No between-group difference was shown at day 84. A reduction in the global ocular symptom severity was shown in both groups at day 35 (-5.6 ± 4.5 and -4 ± 3.9 for the HA-trehalose and HA groups, respectively, p = 0.085) and day 84 (-7.4 ± 6.0 and -6.5 ± 5.0, respectively, p = 356).
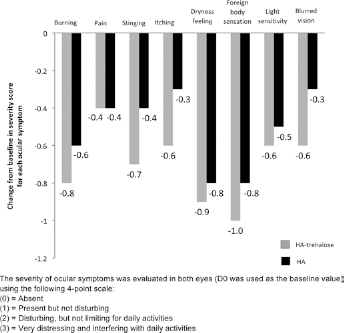
Reduction in severity of dry eye symptoms on day 35 (full analysis set). The severity of ocular symptoms was evaluated in both eyes (D0 was used as the baseline value) using the following 4-point scale: (0) absent; (1) present but not disturbing; (2) disturbing, but not limiting for daily activities; (3) very distressing and interfering with daily activities.
All assessed secondary objective parameters were similarly improved throughout the study in both groups: Schirmer test (mean increase of 2.2 mm/5 min [both groups] at day 35 and 3.0 mm/5 min [HA-trehalose] to 3.7 mm/5 min [HA] at day 84), TBUT (mean increase of 2.0 seconds [both groups] at day 35 and 2.7 seconds [HA-trehalose] to 2.6 seconds [HA] at day 84), van Bijsterveld score (mean reduction of -1.9 [HA-trehalose] to -1.7 [HA] and -2.8 [HA-trehalose] to -2.5 [HA] at days 35 and 84), and conjunctival hyperemia (mean reduction of -1.0 [both groups] at day 35 and -1.5 [HA-trehalose] and -1.4 [HA] at day 84).
The global efficacy assessments by the investigator and patient are shown in Figure 4 (FAS). Both the investigator and patient assessments, and at both days 35 and 84, were better for HA-trehalose than for HA; these differences were significant on both days for the patient assessments (p = 0.045 and p = 0.015 on days 35 and 84) but not for the investigator assessments (p = 0.053 and p = 0.145 on days 35 and 84).
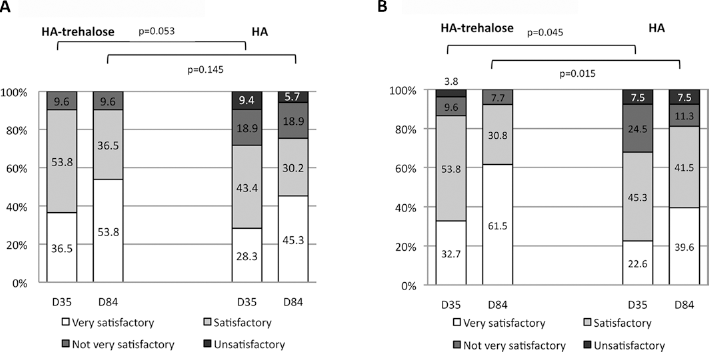
Global efficacy assessments by investigator and patient (full analysis set). (
Safety and tolerability
Overall, for ocular symptoms upon instillation, both the incidence and the severity were lower for HA-trehalose than for HA. The severity score of symptoms was statistically significantly lower in the HA-trehalose group at day 35 (p = 0.03) (Fig. 5); there was no statistically significant difference at day 84.
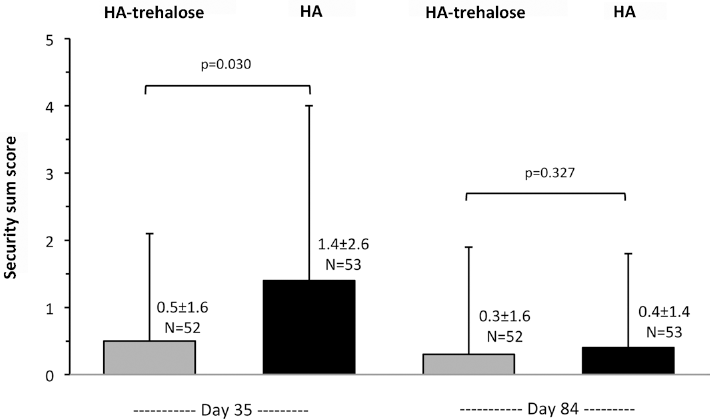
Severity score of ocular symptoms upon instillation (full analysis set). The severity of ocular symptoms was evaluated in both eyes using the following 4-point scale: (0) absent; (1) present but not disturbing; (2) disturbing, but not limiting for daily activities; (3) very distressing and interfering with daily activities. The sum score was based on 5 predefined ocular symptoms: irritation/burning/stinging, photophobia, foreign body sensation, eye dryness feeling, and pain.
There was no difference between groups in global tolerability at day 35 (treatment considered satisfactory of very satisfactory for 88.5% [HA-trehalose] and 86.8% [HA] of patients) or day 84 (94.1% and 93.9%) as assessed by the investigator.
Table III summarizes all ocular treatment-emergent AEs. The overall incidence of ocular AEs was lower for HA-trehalose (1 patient, 1.9%) than for HA (7 patients, 13.2%). The incidence of nonocular treatment-emergent AEs was similar for HA-trehalose (7.7%) and HA (11.3%). Overall, few AEs were considered to be related to the study product (none in the HA-trehalose group and 1 AE of pruritus in the HA group) and most were mild in severity. No patients in the HA-trehalose group were withdrawn prematurely due to an AE; in the HA group, 3 patients were withdrawn, due to AEs of keratitis (due to lack of efficacy of the treatment), conjunctivitis, and productive cough. No serious AEs were reported.
Summary of ocular treatment-emergent adverse events (AE) (all causalities) (safety set)

System organ class and preferred term coding according to the Medical Dictionary for Regulatory Activities Version 16.1 (September 2013).
For BCVA, there was no clinically important change from baseline in either group at either day 35 (mean change of 0.1 ± 0.7 for HA-trehalose and 0.0 ± 0.4 for HA) or day 84 (0.1 ± 0.6 and 0.1 ± 0.8).
Discussion
Trehalose is a naturally occurring bioprotective disaccharide that is present in numerous nonmammalian species, allowing survival in osmotically unfavorable environments by playing a major role in anhydrobiosis, desiccation, and protection against oxidative and other stresses. It acts as an osmoprotectant that is associated with a reduced concentration of intracellular inorganic salts (24), helping to control the osmotic gradient between the extracellular and intracellular environments (25). Trehalose is also a bioprotectant that helps maintain the integrity of cell membranes, protects proteins, promotes homeostasis, and activates autophagy (14–15–16), reducing cell death associated with apoptosis and inflammation. As such, it has numerous medical applications, such as cryopreservation and organ transplant, as described earlier (13), as well as others, such as improving antibody stability. In vitro and in vivo studies have shown trehalose to protect corneal cells from desiccation (26), as well as protecting corneal and conjunctival cells against apoptosis (8), corneal cells against ultraviolet-induced oxidative damage (accelerating corneal healing) (27), and reducing conjunctival inflammatory cytokines in a murine model of DED (10). The bioprotection offered by trehalose is now being applied to DED and could be an important means to counter tear hyperosmolarity, cellular damage, apoptosis, and inflammation (key steps in DED pathophysiology), leading to symptomatic relief.
This study aimed to establish noninferiority in terms of the Oxford score of a new eyedrop formulation (HA-trehalose) containing both trehalose (as a bioprotectant) and hyaluronate (as a promoter of corneal healing with a water-containing network and mucomimetic properties) compared to a preexisting formulation containing only HA. All included patients were diagnosed with moderate to severe DED and presented with highly symptomatic dry eye and notable corneal impairment (i.e., severe keratoconjunctivitis sicca). The baseline profile of the patients was as expected in this DED population with a high incidence of elderly women in each group (menopause is often cited as an etiologic factor). The primary efficacy criterion was met, with HA-trehalose improving keratoconjunctivitis sicca (Oxford score) after 1 month (day 35) and being noninferior to HA; a similar descriptive result was demonstrated at day 84. Together with the secondary efficacy criteria, this shows that the addition of trehalose to a hyaluronate solution does maintain a similar efficacy. In fact, for some secondary efficacy parameters, the results for HA-trehalose were favorable to those for HA, notably for OSDI scores, ocular symptoms of stinging, itching, and blurred vision, for both the investigator and the patient global efficacy assessments. This could be related to the effect of the bioprotectant in HA-trehalose reducing epithelial damage and helping to restore the ocular surface. Together, these results indicate an overall advantage to patients of the HA-trehalose formulation compared to a formulation containing only HA. A similar result for the global efficacy assessments has recently been reported elsewhere (28), and other clinical studies have also demonstrated the value of the addition of trehalose in the treatment of DED (29, 30) and in increasing tear film thickness in patients with DED (31), further supporting its benefits. While there were no group differences for the remaining secondary efficacy parameters, all improved with treatment.
The HA-trehalose formulation was well-tolerated, with significantly fewer and less severe ocular symptoms at instillation, and fewer ocular adverse events compared with the standard HA formulation. These improvements associated with the HA-trehalose formulation, particularly by day 35, demonstrate a clear advantage of the HA-trehalose solution and a clear benefit to patients, reinforcing the advantages seen in the efficacy results, which also show an earlier beneficial effect than the HA formulation. Negative effects of DED have been documented on reading, using a computer, and poor visual performance when driving (32, 33) as well as on anxiety and depression (34) and work productivity (35). In this context, the benefits of HA-trehalose after as little as 1 month are particularly relevant for the improvement of patients’ quality of life. Furthermore, this may translate into better patient compliance with the prescribed treatment and improved effectiveness in the longer term.
Some limitations of this study include a small patient population size and a lack of blinding of the patients to their treatment allocation. However, the small population size was validated statistically for the purposes of this study, and an investigator-masked design was used rather than a double-masked study design since the latter was not possible due to differences in the presentation of the 2 products. Notably, the primary criterion was assessed by the investigator masked to treatment, limiting bias regarding the primary analysis.
Overall these data demonstrate that HA-trehalose is at least as efficacious as HA which, together with other eyedrops containing 0.18% sodium hyaluronate (36), has proven efficacy in the treatment of dry eye syndrome (37). Additionally, HA-trehalose is better appreciated by both the investigator and the patient, and is better tolerated than HA, with its advantageous effects on DED symptoms (both symptoms of ocular discomfort and visual disturbance linked to dry eye) being evident as early as day 35. This confirms that the addition of trehalose improves tolerability and efficacy in terms of symptomatic relief (better improvement of OSDI score and certain ocular symptoms), compared to the instillation of a product that includes only hyaluronate, and demonstrates the beneficial bioprotective effect of trehalose.
The short-term (day 35) improvements with HA-trehalose compared to HA were more marked than in the longer term (day 84), suggesting that HA-trehalose could be considered a therapeutic advancement in the treatment of moderate to severe dry eye syndrome, particularly in the first month of treatment, improving patients’ quality of life and promoting longer term treatment compliance.
Appendix - The HA-trehalose Study Group
Dr. Emmanuel Alliot (Cabinet Medical, Cluses, France); Prof. Tristan Bourcier (Nouvel Hôpital Civil, Strasbourg, France); Dr. Anne Chretien-Malinconi (Cabinet Médical, Marseilles, France); Prof. Béatrice Cochener (CGU Morvan, Brest, France); Dr. Zsuzsanna Etedi-Gaygi (Centre Hospitalier Emile Roux, Le Puy en Velay, France); Prof. Brigitte Girard (Hôpital Tenon, Paris, France); Prof. Pierre-Yves Robert (CHU de Limoges-Hôpital Dupuytren, France); Dr. Anne Sabadel (Centre Médical Croix de Neyrat, Clermont-Ferrand, France); Dr. Marie-Claude Veschambre (Centre Médical Croix de Neyrat, Clermont-Ferrand, France); Dr. Jean-Paul Vounatsos (Cabinet Médical, Bordeaux, France); Prof. Jean-Jacques Gicquel (Centre Hospitalier Saint Louis, Saint Jean d'Angély, France); Prof. Louis Hoffart (CHU Timone Adultes, Marseilles, France); Prof. Raja Gmar-Zhioua (Hôpital Charles Nicolle, Tunis, Tunisia).
Footnotes
Acknowledgment
The authors thank the patients for their participation in this study; all study site personnel; Marielle Estrade for managing the study; Claire Billard and Sylvie Nisslé for statistical analyses; and Dr. Andrew Lane (Lane Medical Writing) and Hervé Chabanon, who provided professional medical writing assistance in the preparation and development of this manuscript in accordance with the European Medical Writers Association guidelines and Good Publication Practice.
Financial support: Supported by Laboratoires Théa, Clermont-Ferrand, France.
Conflict of interest: S.D. has received consulting fees from Laboratoires Théa. C.B. has received consulting and investigator fees from Alcon, Allergan, Santen, and Laboratoires Théa.
Meeting presentation: The data included in this article were presented at the European Association for Vision and Eye Research (EVER) annual congress, Nice, France, October 7-10, 2015 (Poster T085).
