Abstract
Since squamous cell carcinoma antigen (SCC-Ag) testing became commercially available on the Architect platform, the previously established method on the Abbott IMx platform has been progressively replaced. Aim of this work was to compare SCC-Ag values obtained with the 2 methods. Clinical and laboratory data of 188 patients for whom SCC-Ag determination was requested, were reviewed. IMx was used to determine the levels of SCC-Ag from June 2007 to May 2009, while the Architect system was used from June 2009 to April 2011. Only patients consistently diagnosed with no evidence of disease, for whom at least 2 determinations with each analyzer were available were used. Comparison of the results obtained with the 2 systems was then performed. Mean values for SCC-Ag were 0.56 ng/mL (Standard Error (SE): 0.08) with the IMx method, and 1.08 ng/mL (SE 0.10) with Architect (p<0.0001). False positive results were found in 4.8% of patients with the IMx method and in 9.5% of patients with Architect (p=0.049). The values of SCC-Ag determined on the Architect platform are higher than those obtained on the IMx, with a higher percentage of false positive results.
Introduction
Cervical cancer is still one of the major causes of cancer-related death in women. Despite the awareness of its association with persistent infection by high-risk HPV types, and the organization of screening programs (mainly based on Pap smear), in the United States alone cervical cancer was diagnosed in about 12,400 women and caused around 4,000 deaths (1, 2). Early stages of the disease are often asymptomatic, while in advanced stages symptoms related to the cancerous mass are usually reported. After diagnosis and appropriate treatment, patients are followed up with different surveillance programs, all aimed at early detection of disease recurrence, which should be more susceptible to treatment. In this context, tumor markers may play a role. As approximately 80%-85% of cervical cancers are of squamous cell type, the marker of choice for the follow-up of this cancer is the squamous cell carcinoma antigen (SCC-Ag) (3). Described for the first time by Kato and Tarigore in 1977 as one of the 14 sub-fractions of antigens associated with tumors (TA-4), the SCC-Ag is a glycoprotein with a molecular weight of 48 kDa. The SCC-Ag belongs to the serine proteinase inhibitor family, encoded by 2 high homogeneous genes, SCCA1 and SCCA2 (4, 5). SCCA1 and SCCA2 play a key role in the process of tumor development, suppressing apoptosis in human tumor cells (6, 7). Their expression is enhanced in all squamous cell carcinomas (uterine cervix, lung, head and neck, esophagus and anal canal) as well as in several non-malignant skin disorders and renal failure (8–13). Different studies showed that serum SCC-Ag concentrations correlate with tumor stage and reflect the extent of the disease. Moreover, the marker is useful to predict the response to therapy and to detect disease recurrence or progression (14–20). The use of SCC-Ag during regular monitoring of patients requires that the system used for its determination is stable over time, with low analytical imprecision. Moreover, evaluating the comparability of the 2 systems under consideration is of utmost importance in case of a method change.
An automated assay for SCC-Ag determination, based on a chemiluminescent magnetic microparticle immunoassay (CMIA) technology, was introduced for the Architect i2000 platform (Abbott Laboratories, Abbott Park, IL, USA).
The objective of this study was to compare the results generated by the Abbott Architect i2000 assay with those generated by the previously used Abbott IMx method (Abbott Laboratories, Abbott Park, IL, USA). This analysis allows evaluating the comparability of the SCC-Ag measurements obtained with 2 different analytical systems, as well as evaluating the concordance of the SCC-Ag values determined by both systems with the actual clinical state.
Materials and Methods
Patients
We reviewed clinical and laboratory data of all consecutive patients managed between June 2007 and May 2011 at the European Institute of Oncology in Milan (Italy) for whom an SCC-Ag evaluation was requested for diagnosis or follow-up of cervical gynecological cancer. In June 2009, the method for measuring the SCC-Ag was changed, therefore while all samples from June 2007 to May 2009 were tested using the IMx platform (first period), the samples from June 2009 to May 2011 were tested using the Architect i2000 system (second period). For the analysis we selected women with gynecological squamous cell carcinoma having at least 2 determinations during both the first and the second period, for a total of at least 4 SCC-Ag results, with no evidence of disease (NED), as assessed by clinical and imaging evaluation. A comparison of the results obtained in the 2 periods was then performed. The study was notified to the European Institute of Oncology Ethics Committee. All patients gave written informed consent, in accordance with hospital protocols.
Serum SCC-Ag assays
Blood specimens were allowed to clot before being centrifuged at 1100 g for 10 minutes. Serum SCC-Ag measurements were performed using the following analyzers: IMx and Architect i2000 (Abbott Laboratories, Abbott Park, IL, USA). All analyses were performed according to the manufacturer instructions. The cutoff value used was >1.5 ng/mL for both the instrumentations, as suggested by the manufacturer. The analytical sensitivity of the Architect i2000 system and the IMx analyzer were both ≤0.1 ng/mL.
Statistical analysis
Categorical patients' characteristics for the whole sample and for the NED subset were tabulated using counts and percentages. Age at baseline was summarized as: counts (N), mean ± standard deviation (SD), median, min and max. Individual profiles of serum SCC-Ag values, divided by analyzer type, were plotted against the variable “time from the first sampling”. The repeated measurements analysis was a multivariable hierarchical mixed model growth curve with analyzer nested within time and was conducted on the NED patients subset only. Analyzer, International Federation of Gynecology and Obstetrics (FIGO) staging, and age entered the model both as main effects and first-order interactions. Patients' intercepts entered the model as mixed effects. A heterogeneous autoregressive (ARH[1]) covariance structure was specified. Models selection was based on the Akaike Information Criteria (AIC) and the best explanatory model was considered the one with the lowest AIC value. False positive rates by analyzer were calculated as the number of serum SCC-Ag values exceeding the 1.5 ng/mL threshold at least once during the follow-up and compared by the McNemar test. Percent of false positive rate by analyzer, on a per patient basis, was also calculated and compared by the Signed rank-sum test. All tests were 2-sided and considered significant at the 5% level. All analyses were performed using SAS 9.2 (N.C, Cary, USA).
Results
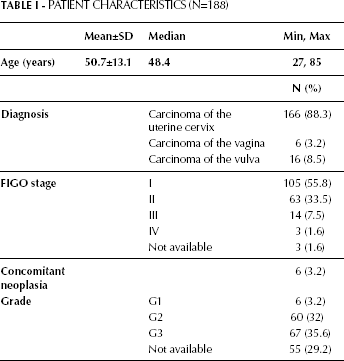
From the prospectively maintained laboratory database 3,549 SCC-Ag values, obtained from a total of 1,238 patients, were extracted (1,811 tested from June 2007 to May 2009 and 1,738 from June 2009 to May 2011). Of these, 237 patients had at least 2 SCC-Ag determinations available during both periods, 14 declined to give informed consent, and of the remaining 223 patients we selected those with a gynecological squamous cell carcinoma (197 patients) with NED. One hundred and eighty-eight patients fulfilled the inclusion criteria and represent the overall study population (Fig. 1). The median age at the time of SCC-Ag measurement was 48 years (range: 27-85 years). One hundred and sixty-six (88.3%) patients had a squamous cell carcinoma of the uterine cervix, 6 (3.2%) had a vaginal carcinoma, and 16 (8.5%) patients had a squamous cell carcinoma of the vulva. Clinical staging according to the FIGO system was as follows: 105 (55.8%) patients had a stage I cancer, 63 (33.5%) stage II, 14 (7.5%) stage III and 3 (1.6%) stage IV; FIGO staging was not available for 1.6% of the patients. Other clinicopathological characteristics are listed in Table I.
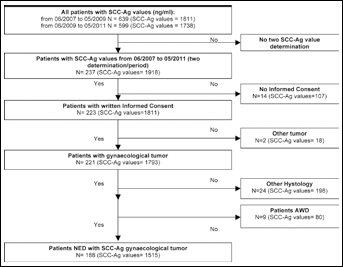
Flow-chart for SCC-Ag and patients' selection.
Patient Characteristics (N=188)
The final dataset analyzed included a total of 1,515 SCC-Ag measurements (814 on the IMx and 701 on the Architect i2000 platform), with a comparable mean number of SCC-Ag samples per patient between instruments: 4.7 (range 2-15) on IMx and 4.2 (range 2-10) and on Architect i2000 (p=0.290).
Figure 2 shows the distribution of the SCC-Ag values obtained with both instruments: on the left side are depicted the values of the tests performed with the IMx system (first period) and on the right side those performed with the Architect system (second period). The mean value for IMx was 0.56 ng/mL (SE 0.08), significantly lower than the mean value determined with the Architect system (1.08 ng/mL, SE 0.10) (p<0.0001).
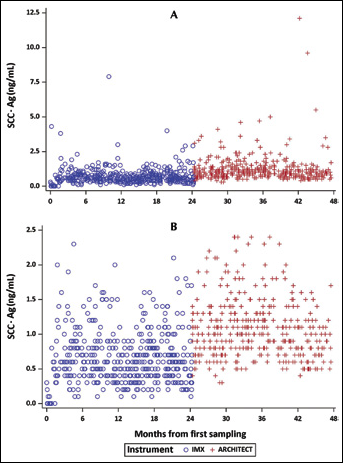
Distribution of SCC-Ag values obtained with the IMX and Architect systems during the 2 periods of observation: A, all values; B, values between 0 ng/mL and 2.5 ng/mL.
The results of the multivariable analysis showed that the only variable significantly affecting the SCC-Ag values is the instrument (p<0.0001). Age (p=0.836), FIGO staging (p=0.752), time (p=0.070), and the interaction of age with FIGO staging (p=0.238) were not significantly associated with the SCC-Ag values.
Considering the false-positive rate, we examined the number of patients showing at least one SCC-Ag above 1.5 ng/mL either during the first or the second period: 9 patients had at least 1 pathological value determined with the IMx system (4.8%), while 18 had a high value determined with the Architect system (9.5%) (p=0.049). Considering the percentage of measurements per patient with SCC-Ag >1.5 ng/mL, significantly higher values were found using the Architect system compared to the IMX system (11.2% and 3.7% respectively, p=0.003).
Discussion
The monitoring of patients with cancer constitutes the most common clinical use of serum tumor markers. SCC-Ag is currently recognized as the marker of choice for the follow-up of squamous cell carcinomas of the uterine cervix, and the most useful in the management of cancer patients according to the European Group of Tumor Markers guidelines (3). Increasing levels of SCC-Ag are reported to precede the clinical detection of relapse by 2-7.8 months (20, 21), and SCC-Ag level changes are strongly correlated with the clinical course of the disease (18). Despite all this, it is still a matter of debate whether earlier detection of relapse may affect the overall survival of the patients, as different results have been reported by different authors (22–24).
During follow-up SCC-Ag is tested at pre-definite intervals of time, to detect potential variations in the marker concentration: it is therefore important to be aware of the performance of the specific assay used, especially in the interpretation of either an abnormal value or changes (increase or decrease) during serial samplings. In fact, methods may differ in terms of sensitivity, specificity, and cutoff, the latter being usually determined in healthy subjects, a definitely different cohort from the one in which the marker is used.
After the introduction, in 2005, of a new assay for SCC-Ag determination based on the CMIA technology of the fully automated system Architect i2000, the previously established method (Abbott IMx system), has been progressively replaced. However, no studies aimed at the evaluation of possible biases between the 2 methods have been performed.
The present study was undertaken with the aim of evaluating if the 2 methods (IMx and Architect i2000, Abbott Diagnostic) are comparable or rather exist differences that may have an impact on the management of the patients. To this aim, we matched the SCC-Ag measurements recorded in the same patients with the 2 analytical systems in 2 consecutive periods of time, including only patients cured and without any evidence of disease.
This study shows that the Architect system gives higher values compared to the IMx system: this is suggested by both the higher overall median SCC-Ag value determined with the Architect system and the finding that very few determinations with Architect gave a result <0.5 ng/mL. The only possible explanation for this discrepancy resides in differences in the formulation of the 2 assays, with a definitely higher background in case of the automated Architect system. As a consequence, this rises some questions regarding the cutoff value, identical for the 2 methods, which leads to more patients presenting with pathological SCC-Ag levels. Apart from subjects with an impairment of renal function, a condition known to increase SCC-Ag concentrations (in our population accounting for only 3 patients, 1 with SCC-Ag determined in both periods and 2 with SCC-Ag determined only in the second period) (9), we could not find any other reliable reason to justify the false positives results. The percentage of patients with elevated SCC-Ag levels detected with the Architect system (9.5%) is higher, but with a borderline significance, than those detected with the IMx system (4.8%). It is important to note that, excluding patients with renal impairment, IMx showed persistent increased levels of SCC-Ag in only 1 case (out of 8), and Architect in 5 cases (out of 20): none of these patients had any evidence of disease at follow-up.
This study has some limitations. First, to properly check the comparability of the results obtained with the Architect and the IMx platforms we should have tested the same samples with both methods at the same time. Nevertheless, the large number of determinations analyzed, equally subdivided into the 2 periods examined, allowed us to obtain an appropriate evaluation of the comparability of the 2 systems. Moreover, this was not a prospective study, but it was based on a review of laboratory and clinical data. However, this approach has the benefit to reliably check the clinical outcome of the patients: in fact, we could effectively select only those subjects free of disease for which no increases in SCC-Ag should have been detected.
We believe that these findings should be a stimulus to further evaluate if the proposed cutoff for the new system is adequate, or if a revision must be performed with an appropriate protocol. This approach seems of utmost importance as, if confirmed, a potential overestimation of the new assay might be problematic for clinicians in the follow-up of the patients. Specifically a potential overestimation may induce anxiety, as well as increase the request for new examinations in the search of a relapse of the disease.
