Abstract
In colorectal cancer, CD133+ cells from fresh biopsies proved to be more tumorigenic than their CD133– counterparts. Nevertheless, the function of CD133 protein in tumorigenic cells seems only marginal. Moreover, CD133 expression alone is insufficient to isolate true cancer stem cells, since only 1 out of 262 CD133+ cells actually displays stem-cell capacity. Thus, new markers for colorectal cancer stem cells are needed. Here, we show the extensive characterization of CD133+ cells in 5 different colon carcinoma continuous cell lines (HT29, HCT116, Caco2, GEO and LS174T), each representing a different maturation level of colorectal cancer cells. Markers associated with stemness, tumorigenesis and metastatic potential were selected. We identified 6 molecules consistently present on CD133+ cells: CD9, CD29, CD49b, CD59, CD151, and CD326. By contrast, CD24, CD26, CD54, CD66c, CD81, CD90, CD99, CD112, CD164, CD166, and CD200 showed a discontinuous behavior, which led us to identify cell type-specific surface antigen mosaics. Finally, some antigens, e.g. CD227, indicated the possibility of classifying the CD133+ cells into 2 subsets likely exhibiting specific features. This study reports, for the first time, an extended characterization of the CD133+ cells in colon carcinoma cell lines and provides a “dictionary” of antigens to be used in colorectal cancer research.
Introduction
The theory of cancer stem cells has recently provided new insights into tumorigenesis and metastatic processes as well as novel explanations for cancer recurrences (1). This theory states that only a small subset of cancer cells is able to indefinitely self-renew without differentiating, and to support the continuous growth of the tumor (1). The so-called cancer stem cells show different phenotypes in different types of cancers: in colon carcinomas they seem to be CD133+/Ep-CAMhigh/CD166+/CD44+ (2–4). Tumors arising from epithelia share a partially common phenotype characterized by CD133 expression. This molecule, also called prominin 1, is a transmembrane glycoprotein firstly found on CD34+ hematopoietic stem cells (5). In a growing number of tissues CD133 is used, alone or in combination with other markers, to separate the stem cell fraction in both normal (6) and cancer specimens (3). Although little is known about the biological function of CD133 (7) its role as stem cell marker has been commonly recognized. Nevertheless, recent findings seem to question the utility of CD133. Its modulation by oxygen levels and mycoplasma infections (8, 9), the ability of CD133– colon cancer cells to initiate tumors in mice (2, 10), the low percentage of CD133+ cells capable of forming spheres (3), and the scarce effect of RNA interference silencing of CD133 on stem cell-like properties (11) all suggest that the use of CD133 alone as stem cell marker has to be reassessed and, moreover, highlight the need for additional markers. It has been demonstrated that also cancer-derived continuous cell lines contain a stem-like compartment and are suitable for analysis of cancer stem cells phenotype and function (12–14).
In this paper we report, for the first time, an extensive characterization of putative cancer stem cells (CD133+) in 5 different colon adenocarcinoma continuous cell lines, i.e. HT29, HCT116, Caco2, GEO and LS174T. For this characterization we used a panel of 33 antigens, selected from markers that are potentially associated with stemness, tumorigenesis and metastasis, along with epithelial specificities. We evaluated, by a polychromatic analysis, the expression of all these markers on CD133+ cells. The final goal of this work was to find new features characterizing the CD133+ fraction in colorectal tumors, in order to provide the scientific community with information about novel molecules, possibly functional, on colorectal cancer stem cells.
Materials and Methods
Cell cultures
Five human colon carcinoma cell lines were used in this study: HT29, HCT116, Caco2, GEO and LS174T. HT29, HCT116 and Caco2 cell lines were purchased from the DSMZ cell bank, while GEO and LS174T cell lines were authenticated and provided by the CEINGE cell bank. HT29 and HCT116 cell lines were propagated in McCoy'S 5A medium (SIGMA-Aldrich, Oakville, ON, USA) supplemented with 10% of fetal bovine serum (FBS, GIBCO, Carlsbad, CA, USA) and 1% ultraglutammine (Cambrex, East Rutherford, NJ, USA). Caco2 and LS174T cell lines were grown in essential minimum Eagle's medium (EMEM, SIGMA-Aldrich, Oakville, ON, USA) supplemented with 10% FBS, 1% ultraglutammine, 1% non essential amino acids and 1% sodium pyruvate. The GEO cell line was maintained in Dulbecco's modified Eagle's medium (DMEM, SIGMA-Aldrich, Oakville, ON, USA) enriched with 10% FBS and 1% ultraglutammine. Cultures were grown at 37°C in a humidified 5% CO2 incubator and split when 80%-90% confluent. Adherent cells were detached using Trypsin-EDTA solution (SIGMA-Aldrich, Oakville, ON, USA), then floating cells were collected and trypsin-EDTA was inactivated using complete culture medium.
Relevant features of the selected cell lines
The cell lines used in this study have different characteristics. HT29 and HCT116 are quite undifferentiated with a high expression of CD133 (100% and 80% respectively, data not shown) and high tumorigenic and metastatic potentials. GEO and LS174T cell lines are well differentiated cell lines with low expression of CD133 (2% and 1% respectively, data not shown) and a reduced metastatic potential. Caco2 is a cell line with intermediate characteristics, as it is spontaneously differentiating in normal culture conditions, it highly expresses CD133 (100%, data not shown), is tumorigenic but hasn't a metastatic behavior in vivo (15–22).
Immunophenotyping and flow cytometry experiments
Cells were detached by trypsin-EDTA solution, then washed and reconstituted to a final concentration of 1×10 6 cells/mL in 2% FBS/PBS. Cells were subdivided in 5 mL polystyrene tubes (Falcon, Becton Dickinson, San Jose, CA, USA). We incubated 50 μL of cell suspensions with 5 μL of each antibody for 30 minutes at 4°C. Cells were then washed with 1 mL of 2% FBS/PBS, re-suspended in 500 μL of 2% FBS/PBS and analyzed by flow cytometry. We repeated each characterization 3 times to validate the results observed. We used the BD FACSAria cell sorter (Becton Dickinson, San Jose, CA, USA) for the analysis. The cell sorter was equipped with 4 excitation channels (633 nm, 488 nm, 407 nm, and 375 nm lasers). FITC (fluorescein isothiocyanate), PE (phycoerythrin), Per-CP (peridinin chlorophyll protein), PE-Cy7 (phycoerythrin-cyanine 7) were excited by the 488 nm channel while APC was excited by the 633 nm laser. The antigens chosen for the present study are reported in Table I. The antibodies used in this study included CD133-PE and CD133-APC (AC133 clone; Miltenyi Biotec, Auburn, CA, USA), CD9-APC, CD10-APC, CD13-APC, CD24-FITC, CD26-FITC, CD29-APC, CD34-PE, CD43-FITC, CD44-APC, CD47-FITC, CD49b-FITC, CD49f-FITC, CD54-APC, CD55-PE, CD56-PECy7, CD59-PE, CD66b-FITC, CD66c-PE, CD81-APC, CD90-FITC, CD99-PE, CD112-PE, CD117-PerCP, CD151-PE, CD164-FITC, CD165-FITC, CD166-PE, CD200-PE, CD221-PE, CD227-FITC, CD324-FITC, CD326-APC (BD Biosciences and BD Pharmingen, San Jose, CA, USA), and CDw338-APC (R&D Systems Minneapolis, MN, USA).
Antigens Analyzed in the Study
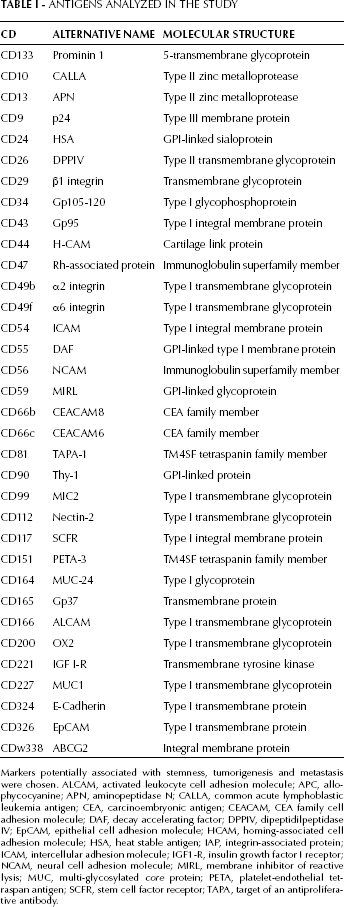
Markers potentially associated with stemness, tumorigenesis and metastasis were chosen. ALCAM, activated leukocyte cell adhesion molecule; APC, allophycocyanine; APN, aminopeptidase N; CALLA, common acute lymphoblastic leukemia antigen; CEA, carcinoembryonic antigen; CEACAM, CEA family cell adhesion molecule; DAF, decay accelerating factor; DPPIV, dipeptidilpeptidase IV; EpCAM, epithelial cell adhesion molecule; HCAM, homing-associated cell adhesion molecule; HSA, heat stable antigen; IAP, integrin-associated protein; ICAM, intercellular adhesion molecule; IGF1-R, insulin growth factor I receptor; NCAM, neural cell adhesion molecule; MIRL, membrane inhibitor of reactive lysis; MUC, multi-glycosylated core protein; PETA, platelet-endothelial tetraspan antigen; SCFR, stem cell factor receptor; TAPA, target of an antiproliferative antibody.
Gating strategies
Cells were first gated on physical parameters, FSC (forward scatter) and SSC (side scatter), to exclude the majority of dead and apoptotic cells. Then, the FSC-Area versus FSC-Height profile was used to identify single cells and exclude doublets. Putative cancer stem cells were gated in a CD133 vs “empty channel” dot plot. For the HCT116, GEO and LS174T cell lines we followed the spontaneous separation of a subset of CD133+ cells from more differentiated cells. For the other 2 cell lines (Caco2 and HT29) no gate on CD133 positivity was imposed since cells were entirely CD133+.
The background level for each fluorochrome was determined taking one channel empty at a time (the so called “fluorescence-minus-one” technique). This helped us to evaluate the spillover of other fluorochromes in the empty channel. Were defined completely positive (+) those antigens for which more than 99,6% of cells analyzed resulted positive, partially expressed (±) were those antigens for which the positive subset was lower than 99,6%, and not expressed (-) those antigens that were negative at least on 99,6% of the cell population analyzed.
Results
Classification of the surface antigens included in the study
All the antigens analyzed in this study are reported in Table I. In Table II we reported the immunophenotyping results for the 5 cell lines analyzed. Surface antigens were classified in 4 groups:
Antigens homogeneously and consistently positive in the 5 cell lines (CD9, CD29, CD49b, CD59, CD151, CD326). These antigens were positive on 100% of the CD133+ cells in all cell lines analyzed.
Antigens heterogeneously positive (CD44, CD47, CD49f, CD55, CD221, CD227, CD324). These antigens were operationally considered positive, although in some cases the percentage did not reach 100%. CD227 is a paradigmatic example: it was expressed in every type of cell line utilized, but on a variable fraction of CD133+ cells.
Antigens with discontinuous behavior (CD24, CD26, CD54, CD66c, CD81, CD90, CD99, CD112, CD164, CD166, CD200). These antigens were positive in some cell lines and negative in others. Thus, on basis of their expression, it was possible to derive a specific pattern for each cell line.
Always negative antigens (CD10, CD13, CD34, CD43, CD56, CD66b, CD117, CD165, CDw338).
Classification of the Surface Antigens on Basis of Their Expression on CD133+ Cells in 5 Colorectal Carcinoma Cell Lines
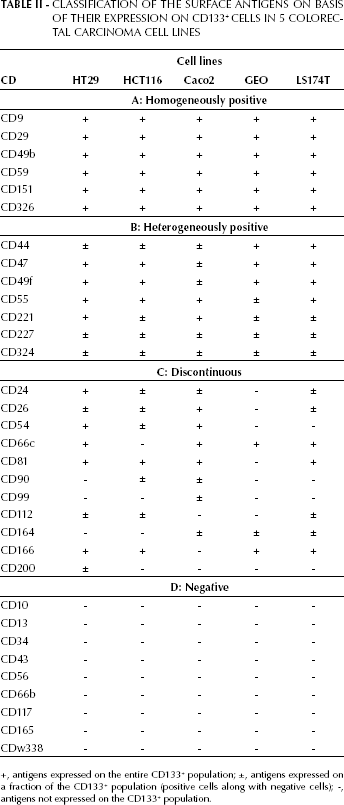
+, antigens expressed on the entire CD133+ population; ±, antigens expressed on a fraction of the CD133+ population (positive cells along with negative cells); -, antigens not expressed on the CD133+ population.
Flow cytometric characterization of the undifferentiated cell line HT29
Vertical reading of Table II allows the obtaining of the phenotype of each cell line. HT29 CD133+ cells were characterized by the expression of CD9, CD29, CD49b, CD59, CD151, CD326, CD47, CD49f, CD55, CD221, CD24, CD66c, CD54, CD81, and CD166. CD44, CD227, CD324, CD26, CD112, and CD200, partially expressed, distinguished the CD133+ population into 2 subsets, depending on the positive or negative expression of each of the markers. The other antigens analyzed (CD90, CD99, CD164, CD10, CD13, CD34, CD43, CD56, CD66b, CD117, CD165, CD324, and CDw338) were completely absent.
Flow cytometric characterization of the poorly differentiated cell line HCT116
Table II summarizes the expression of the selected antigens on HCT116 CD133+ cells. These cells were characterized by the homogeneous expression of CD9, CD29, CD49b, CD59, CD151, CD326, CD47, CD49f, CD55, CD81, and CD166. Furthermore, the antigens CD44, CD221, CD227, CD324, CD24, CD26, CD54, CD90, CD112, resulted partially expressed. The remaining markers (CD66c, CD164, CD99, CD200, CD10, CD13, CD34, CD43, CD56, CD66b, CD117, CD165, CD324, and CDw338) were completely absent on this cell line.
Flow cytometric characterization of the spontaneously differentiating cell line Caco2
In Table II the expression of the selected antigens on Caco2 CD133+ cells is also reported. The Caco2 cell line is defined by a homogenous expression of CD9, CD29, CD49b, CD59, CD151, CD326, CD55, CD221, CD26, CD54, CD66c and CD81. A number of antigens (CD44, CD47, CD49f, CD227, CD324, CD24, CD90, CD99, and CD164) allowed for the distinction of these cells into 2 subsets, depending on the positive or negative expression of each of the markers. The remaining markers CD112, CD166, CD200, CD10, CD13, CD34, CD43, CD56, CD66b, CD117, CD165, CD324 and, CDw338) were completely absent on this cell line.
Flow cytometric characterization of the well differentiated cell line GEO
Table II shows the expression of the 33 selected antigens on the GEO cell line. CD133+ cells were defined by a uniform expression of CD9, CD29, CD49b, CD59, CD151, CD326, CD44, CD47, CD49f, CD66c and CD166. A number of antigens (CD55, CD221, CD227, CD324 and CD164) distinguished the CD133+ population into 2 subsets, defined by their positivity or negativity. The remaining markers (CD24, CD26, CD54, CD81, CD90, CD99, CD112, CD200, CD10, CD13, CD34, CD43, CD56, CD66b, CD117, CD165, CD324, and CDw338) were completely negative on this cell line.
Flow cytometric characterization of the highly differentiated cell line LS174T
The last column of Table II reports the phenotype of the CD133+ population of the LS174T cell line. These cells were all positive for CD9, CD29, CD49b, CD59, CD151, CD326, CD44, CD47, CD49f, CD55, CD66c, CD81, and CD166 antigens. A number of antigens (CD221, CD227, CD324, CD24, CD26, CD112, and CD164) distinguished the CD133+ population into 2 subsets defined by their positivity or negativity. The remaining markers (CD54, CD90, CD99, CD200, CD10, CD13, CD34, CD56, CD66b, CD117, CD165, and CDw338) were completely negative on this cell line.
Discussion
This study presents an extended cytometric analysis of the CD133+ compartment in 5 different continuous cell lines obtained from colon adenocarcinoma patients. To our knowledge, this is the first time that CD133+ cells have been characterized in such a comprehensive way. The 5 cell lines represent different developmental stages of colorectal cells: in fact, HT29 and HCT116 are considered rather undifferentiated, Caco2 is described as spontaneously differentiating, while GEO and LS174T are considered to be well differentiated (15–22). The analyzed cell lines display also different behaviors with regard to the tumorigenic and metastatic potentials in vivo (15). Some of the antigens identified may be also functionally linked to each other: in fact CD9, CD151, and CD81 are known to be components of the so called “tetraspanins enriched microdomains” (TEM). The TEM domains have an important role in regulating motility and interactions of cancerous cells with their microenvironment, thank to their ability to organize other transmembrane proteins such as cell adhesion molecules, growth factors and proteases (23, 24). Known interactors of tetraspanins in the TEM domains are integrins (CD49b, CD49f, CD29), CD44, CD326, as well as other analyzed molecules that could be organized in such TEM signaling platforms (i.e. the peptidase CD26) (23).
Common core phenotype
The human CD133+ cells in all the cell lines analyzed in this study shared a common phenotype, including the expression of CD9, CD29, CD49b, CD59, CD151, CD326 on the entire population. All these common antigens may represent the basic characteristics shared by cancer stem cells. CD326, as well as CD29 and CD49b integrins, seem to be involved in migration and invasiveness of cancerous cells (25, 26). Also the tetraspanins CD9 and CD151 are involved in cell motility and adhesion processes during cancer development, as already demonstrated by experiments on CD9 engagement and CD151 blockade (27, 28). CD59 can be related to other properties of cancerous cells, since it is a membrane complement inhibitor and was found to have a role in the resistance to complement-related toxicity (29).
Heterogeneously positive antigens
A number of antigens showed a heterogeneous behavior despite the fact that they were consistently present on the CD133+ population. CD227 was expressed on a variable subset of CD133+ cells. This antigen has been found in colorectal cancers in an abnormal hypo-glycosylated form that elicits an immune response in patients; a CD227-based vaccine was successfully used in a preclinical model of colon cancer (30).
CD47 has been recently linked to cancer stem cells in acute myeloid leukemia, where it is able to distinguish normal stem cells from cancerous stem cells in the CD34+/CD38+ population (31). It is intriguing to observe that CD47 was maximally expressed on the undifferentiated HT29 cell line. Similarly to CD59, also CD55 plays a role in the resistance to complement-mediated toxicity (30). CD221, the insulin-like growth factor receptor, showed a higher expression associated with CD133/CD44 expression in hepatocellular carcinomas (32).
Discontinuous antigens
The third class of antigens analyzed had variable expression patterns in the cell lines used for this study and resulted alternatively positive or negative. Some of these discontinuously expressed antigens showed patterns linked to the differentiation grade of the cells. CD24, CD112, and CD200 resulted more expressed in HT29 CD133+ cells than in particular CD200 was in the CD133+ populations of the other cell lines. Among these antigens, CD24 has been suggested to be a specific marker for colon cancer stem cells and, despite the fact that its function is still unexplored, it has been observed that CD24 is rapidly downregulated in colon-sphere differentiation (3). An interesting role could be attributed to CD200 expression: this molecule has an immunomodulatory activity and could be involved in the immunosuppression elicited by cancer cells. CD200 has recently been found to be expressed on cancer stem cells in solid tumors (33).
CD26, CD54, and CD81 were expressed in HT29, HCT116 and Caco2 cell lines while they were negative in the differentiated GEO cell line. It is known that CD26 plays a role in cancer cells malignant behavior, is a peptidase involved in tumor-stroma interactions and has been recently associated with the metastatic potential of colon cancer stem cells (34). Tetraspanin CD81 and the cell adhesion molecule CD54 have both been related to the invasive and metastatic potentials of cancer cells (35, 36). Interestingly, CD164 behaves according to an opposite trend, being expressed in Caco2, GEO and LS174T, CD133+ cells but negative in HT29 and HCT116, CD133+ cells; its downregulation seems to be related to an enhanced metastatic potential in colon carcinomas (37). Unexpectedly, CD66c was expressed by all cell lines but HCT116. CD66c, a member of the CEA antigen family, has proved to play a role in invasiveness and metastatic potential of pancreatic cancer cells and has been evaluated as potential therapeutic target in animal primate models (38); moreover, we found that CD66c is a reliable colon cancer stem cell marker in fresh cancer tissues (7).
Conclusion
Our extended characterization of 5 different colon cancer cell lines has highlighted the existence of a common phenotype for the CD133+ cells characterized by the presence of CD9, CD29, CD49b, CD59, CD151, and CD326. The comparison between the undifferentiated HT29, and HCT116 the spontaneously differentiating Caco2 and the differentiated GEO and LS174T cell lines also highlighted a number of markers associated with the stage of maturation of these colon cells' progenitors. Expression changes in some of these markers could correspond to differences in the surface membrane composition between the CD133+ stem cell and progenitor cell compartments. Our findings are coherent with the hypothesis that an undifferentiated and more aggressive cell line can be similar to a fresh cancer-derived stem cell. In fact, the HT29 and HCT116 cell lines characteristics highlight that, in these highly undifferentiated cell lines, the surface phenotype of the CD133+ cells is characterized by a pattern of expression linked to the invasive and aggressive characteristics of cancer cells (i.e. CD24, CD47, CD49f, CD166, CD112, and CD200 expression). Conversely, more differentiated Caco2, GEO and LS174T, although with different extents, cells had a reduced expression of the same markers on the CD133+ cell compartment (data not shown).
We here provided a sort of “legend” to the cell surface characteristics of cancer stem5 cells that could help researchers choosing the best cell model for their experiments and thus dissecting the complex issue of the surface phenotypic signature of cancer stem cells from fresh samples of colon tumor. Altogether this information could help in the selection of multi-parametric antibody panels to be used for screening the stem cell content of different samples, such as primary tumors or metastases, in order to find new cancer stem cell-associated antigens with a prognostic value. Moreover this information could be used in the analysis of complex issues, such as the analysis of circulating cancer stem cells, whose identification is largely affected by the scarcity of specific markers (39).
