Abstract

To the Editor,
Prostate cancer is the second leading cause of cancer-related death in men in most Western countries. Advanced and metastatic prostate adenocarcinoma has been traditionally managed with androgen-deprivation therapy. However, the disease eventually becomes resistant to castration and evolves into castration-refractory prostate cancer (CRPC), which is associated with a poor prognosis. In this setting, docetaxel has been the first drug to be proven to have survival benefit; other drugs such as estramustine or mitoxantrone have shown a palliative effect on bone pain but have not improved survival.
The efficacy of docetaxel has been investigated in some phase III studies. In 2004, 2 large randomized trials comparing chemotherapy treatments in CRPC (1, 2) showed that survival and quality of life are significantly improved by docetaxel and prednisone in comparison with older regimens. Therefore, docetaxel has become the first-line standard of care, with prostate-specific antigen (PSA) responses of about 50%, median survival of usually <20 months, and progression-free survival that does not exceed 7 months.
More recently, some docetaxel-based combinations have been investigated. In 2011, a randomized phase II trial by Sonpavde et al (3) compared the addition of AT-101, a small- molecule oral inhibitor of the Bcl-2 family, with 3-weekly docetaxel plus continuous prednisone with 3-weekly docetaxel plus prednisone alone. The overall survival (OS; primary endpoint) in 221 evaluable patients was not significantly different between the 2 groups and neither were secondary endpoints. In 2012, Kelly et al (4) compared the combination of docetaxel plus prednisone and bevacizumab with standard therapy in a randomized, double-blind, placebo-controlled phase III trial. A total of 1,050 men with metastatic CRPC were involved. The median OS (primary endpoint) was not significantly different in the group receiving docetaxel and bevacizumab (plus continuous P) or the group receiving standard docetaxel and prednisone. However, a greater proportion of patients in the experimental arm achieved a PSA response and an objective response by reduction of measurable disease than patients in the control arm.
Although most clinical trials (5, 6) in prostate cancer have used an improvement of ≥50% in the serum PSA as a marker of response and this is also commonly used to guide treatment decisions in individual patients, median OS (defined as the median time from random assignment or first treatment to death) remains a universally accepted endpoint for phase III trials and for the evaluation of treatment efficacy in cancer patients. The identification of a surrogate marker for OS in CRPC patients treated with docetaxel-based therapy would represent an important advance in the early identification of active therapy, particularly now that the available alternative therapeutic options have increased.
In order to identify a surrogate marker for OS in CRPC patients, Armstrong et al (7) examined various degrees of PSA decline and pain response as surrogate markers for the survival benefit observed in the TAX327 trial. The analyses found that patients with minimal pain had longer median survival than symptomatic patients. PSA decline (the optimal cutoff was identified as a ≥30% decline over 3 months) was also associated with improved survival, but neither pain nor PSA endpoints are considered to be suitable surrogates for OS in phase III trials. Also a retrospective analysis of the SWOG 9916 trial performed by Petrylak et al (8) confirmed the PSA decline of ≥30% within 3 months of chemotherapy as a surrogate for OS. However, prospective validation of these potential surrogates is required. Other intermediate surrogates have been identified, such as an early change in circulating tumor cells (9) or biochemical markers of bone metabolism (10) but, although their use as prognostic factors is more clear, there is uncertainty as to their use as surrogate markers for OS.
On the basis of these findings, it would seem reasonable to extend the analyses performed by Armstrong et al and Petrylak et al to other docetaxel-based first-line treatments of CRPC. Particularly, as a surrogate for OS, we opted for PSA response rate (RR) as a decrease of ≥50% in the PSA concentration from the pretreatment baseline value. Considering this as a primary endpoint would allow studies of shorter duration with consequent earlier results and lower costs.
Therefore, in this letter we present the preliminary results of an ongoing revision performed by our group. We carried out a literature search for controlled or randomized studies of docetaxel-based first-line treatment of patients affected by metastatic CRPC with at least 100 patients enrolled and reporting both PSA RR and median OS. We used MEDLINE to search for controlled trials containing the keywords “Docetaxel Prostate cancer” while we searched manually for abstracts of the American Society of Clinical Oncology and the European Society of Medical Oncology annual meetings. Six studies comparing 14 arms of different docetaxel-based chemotherapies were chosen (Tab. I) (1–4, 11, 12). For trials with multiple arms, we compared the control arm with a randomly chosen experimental arm.
Trials Included in the Analysis
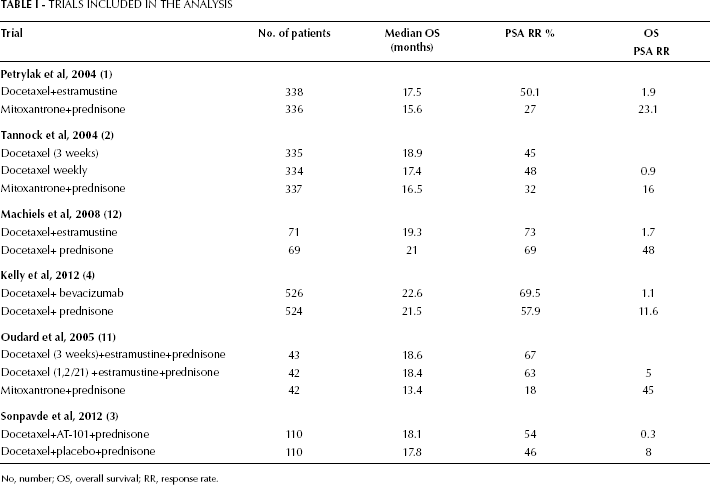
No, number; OS, overall survival; RR, response rate.
The nonparametric Spearman rank correlation test (ρ) was used to search for relations between PSA RR and OS. We also established Δ PSA RR and Δ OS by subtracting the value in the control arm from the value in the experimental arm. StatIC software, version 2012, was used for the analysis.
Although the limited sample size does not allow definitive conclusions, our preliminary results show a strong relation between PSA RR and OS, with ρ=0.909 (p≤0.0001). However, when tested for correlation between Δ PSA RR and Δ OS, ρ decreased to 0.771 and proved nonsignificant (p=0.07).
CRPC is a heterogeneous disease, with wide variation in clinical response to hormone manipulation and chemotherapy. Although docetaxel plus prednisone remains the optimal first-line chemotherapy for patients with metastatic CRPC, it is clear that this treatment is not suitable for administration in a prolonged number of cycles, particularly considering the recent increase in available therapeutic options after first-line docetaxel-based treatment. Therefore, surrogate endpoints and predictive biomarkers of response are required to help physicians identify in good time patients who do not respond to docetaxel.
In conclusion, although the issue about the correlation between PSA RR and OS for metastatic CRPC patients treated with first-line docetaxel-based chemotherapy remains unresolved, our results suggest that a revision of the role of PSA RR as a surrogate marker for median OS is required. Meanwhile, we believe that OS should remain the primary endpoint until specific studies will validate other surrogate endpoints. However, PSA RR should not be ignored for the early identification of patients who do not respond to docetaxel and who would benefit from new therapeutic alternatives.
