Abstract
Objectives
This study aims to evaluate and compare cytokines in gingival crevicular fluid (GCF) and saliva of patients with aggressive periodontitis (AP) before and after treatment.
Methods
Forty AP patients and 40 healthy volunteers were enrolled in this study. Clinical parameters included probing depth and sulcus bleeding index. GCF and saliva were collected from both groups. The levels of IL-1β, IL-2, IL-4, IL-6, IFN-γ and TNF-α were measured using ELISA.
Results
The probing depth in AP patients was significantly deeper before treatment than after treatment. The concentrations of cytokines in GCF and saliva were significantly higher in AP patients than in the control group and decreased after periodontal treatment. Positive relationships were found between cytokine levels in GCF and clinical parameters. The reliability of cytokines in GCF and saliva was assessed by Cronbach's alpha analysis, which could be considered satisfactory.
Conclusion
Cytokine levels in GCF and saliva correlated well with clinical parameters and AP. Measurements of cytokines in saliva may be regarded as a noninvasive and quick method for monitoring periodontal disease activity.
Introduction
Periodontal disease is a disease of periodontal tissues characterized by loss of connective tissue attachment and bone around the teeth in conjunction with the formation of periodontal pockets (1, 2). The tooth supporting collagen fibers of the periodontal ligament and bone are broken down, mainly due to the development of an exacerbated immune inflammatory response to a bacterial dental biofilm. The local ecological conditions of the gingival sulcus and host response can influence the overgrowth of periodontal bacteria (3, 4) and promote destructive periodontitis, resulting in irreversible loss of connective tissue and bony attachment.
Traditional characteristics of aggressive periodontitis (AP) involve rapid periodontal destruction; this generally occurs in young patients with no concomitant systemic diseases. Familial aggregation can also occur (1). Activation of the innate immune system by periodontal pathogens results in excessive production of proinflammatory cytokines, which play a major role in the progression of periodontitis. Furthermore, the acquired immune system activated by periodontal pathogens would further contribute to disease progression. Therefore, ample recent periodontal research studies have focused on finding and testing the cytokine profiles of periodontal disease activity. Cytokines and chemokines produced by immune cells play an important role in immune and inflammation responses. The different profiles of cytokines are normally determined by the subsets of T helper (Th) cells. Different subsets of Th cells are characterized by different cytokine profiles. Th1 cells produce mainly interleukin (IL) 1β, IL-2, IL-12, interferon gamma (IFN-γ), and tumor-necrosis factor alpha (TNF-α), which induce cellular immune responses. IL-4, −5, −6, −10, and −13 are mainly produced by Th2 cells and induce the humoral immune response (5).
The levels of cytokines released by the tissues and cells adjacent to the teeth, measured in the gingival crevicular fluid (GCF), were investigated by several previous studies, but their results are incomplete and controversial (6-9). For instance, a higher IL-1β/IL-10 ratio was found in AP patients than in periodontally healthy volunteers, suggesting an imbalance between pro- and antiinflammatory cytokines in AP (8, 9). Elevated levels of IL-2, IFN-γ (9) and TNF-α (6) in GCF samples were observed in AP patients compared with healthy individuals. Lower Levels of IL-4 were measured in GCF samples of patients with chronic and aggressive periodontal disease (6). Conversely, Zong et al (10) reported that the level of IL-4 in patients with chronic periodontitis was significantly higher than the control group. However, Tsai et al (11) suggested the IL-4 to IFN-γ ratio rather than IL-4 as such to be associated with periodontal status.
GCF is an inflammatory exudate collected within the gingival crevice or at the orifice of the crevice, which has been applied to the diagnosis of oral and systemic diseases (12). Another oral fluid of interest for clinical diagnosis is saliva. The present study aims to comparatively evaluate different cytokine profiles in GCF and saliva, and to detect whether salivary biomarkers reflect periodontal status over time in AP patients who received localized periodontal therapy.
Materials and Methods
Patient selection criteria
A total of 80 subjects including 40 patients and 40 healthy volunteers were enrolled in this study. Informed consent was obtained from all subjects, and the study protocol was approved by the Ethics Committee of West China Hospital of Stomatology, Sichuan University. The patient group included 13 females and 27 males aged 19 to 29 years (mean age ± standard deviation 24.43±2.61 years) with moderate to advanced AP. Subjects from the patient group were recruited from West China Hospital of Stomatology. The healthy group included 15 females and 25 males between the ages of 19 and 29 (mean age 24.33±2.70 years) who were selected from local colleges. Individuals from the healthy group were invited for a full oral examination at West China Hospital of Stomatology.
The diagnosis of periodontal disease was based on the criteria defined by the American Academy of Periodontology in 1999 (13). Subjects were regarded as healthy when they presented no signs of periodontal disease with the absence of gingival recession, clinical attachment loss (CAL) and sites with probing depth (PD) >3 mm. All candidates were confirmed to meet the study criterion of being free of systemic diseases such as rheumatoid arthritis, diabetes, hypertension and cardiovascular disease. Additionally, the participants could not be regular drinkers or smokers. Female subjects could not be menstruating, pregnant, or breastfeeding during the time of the oral examination and during treatment. All subjects were also free of oral soft tissue abnormalities and severe dental caries. Subjects did not receive local periodontal treatment or antibiotics for 3 months prior to the start of diagnostic procedures.
Clinical parameters
An intraoral examination of periodontal conditions including supragingival/subgingival calculus, gingival recession, bleeding on probing (BOP), PD, CAL, gingival recession and tooth mobility was performed.
Sample collection
Samples were collected before and 30 days after the periodontal treatment. Before GCF and saliva samples were taken, subjects received normal saline for an oral rinse. After the collection of the saliva samples with the Greiner Bio-One Saliva Collection System (Greiner Bio-One GmbH, Frickenhausen, Germany), GCF samples were taken from 6 sites (mesiobuccal, buccal-central, buccal-distal, mesio-lingual, lingual-central, and lingual-distal). The periodontal probe had to be first applied to measure the depth of the sulcus in each subject with a device that was designed for this study. A syringe with a needle pore size of approximately 150 μm and 2 cm in length was placed on the occlusal surface of the teeth during the aspiration of GCF. The device was inserted at a depth of approximately 80% into the sulcus for drawing the GCF. All samples were stored for further biomarker examinations.
Measurement of cytokine profiles in GCF and saliva
The levels of IL-1β, IL-2, IL-4, IL-6, IFN-γ and TNF-α in GCF and saliva were measured by ELISA according to the manufacturer's instructions (Diaclone, France). The procedures were as follows: 100 μL standards, GCF and saliva of each subject were pipetted into a 96-well plate. The plate was incubated for 2-3 hours at 350 rpm and washed with washing buffer 3 times. Two hundred microliters of substrate tetramethylbenzidine was added into each well for 20 minutes in the dark at room temperature. The plate was read at 450 nm wavelength using a Sunrise universal microplate reader (Tecan, Grödig, Austria). The levels of cytokines in the samples were obtained by comparison with the standard curve generated from standards supplied by the manufacturer.
Statistical analysis
The values obtained are presented as range, mean, and standard deviation. The ANOVA technique was used for multiple group comparisons followed by the Mann-Whitney test for groupwise comparisons. A p value of 0.05 or less was considered statistically significant. The homogeneity or internal consistency of the Assessment of cytokines in saliva and GCF was assessed using Cronbach's alpha. Cronbach's alpha between 0.70 and 0.95 was considered satisfactory (14).
Results
Clinical parameters in response to therapy
The demographic and clinical characteristics of the enrolled subjects are described in Table I. Treatment led to significant reductions of PD and BOP in AP patients when baseline values were compared with posttreatment values (p<0.05) (Tab. II). CAL also improved from a baseline value of 5.93±1.12 to a posttreatment value of 5.85±1.05, although the change was not significant (p>0.05) (Tab. II).
DEMOGRAPHIC AND CLINICAL CHARACTERISTICS OF STUDY GROUPS
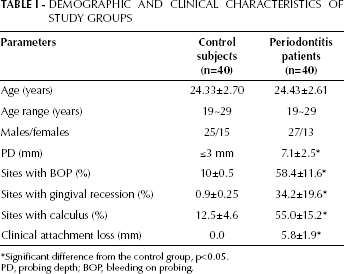
Significant difference from the control group, p<0.05.
PD, probing depth; BOP, bleeding on probing.
CLINICAL PARAMETERS OF PATIENTS WITH AGGRESSIVE PERIODONTITIS
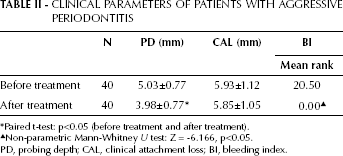
Paired t-test: p<0.05 (before treatment and after treatment).
Non-parametric Mann-Whitney U test: Z = −6.166, p<0.05.
PD, probing depth; CAL, clinical attachment loss; BI, bleeding index.
Cytokine concentrations in GCF in response to therapy
The detected concentrations of various cytokines in GCF are presented in Table III. The levels of IL-1β, IL-2, IL-4, IL-6, IFN-γ and TNF-α were higher in AP patients than in healthy individuals. After treatment there were significant reductions in the levels of IL-1β, IL-2 and IFN-γ (p<0.05), which decreased most significantly with respect to baseline values (2.22-, 2.86- and 2.22-fold, respectively).
CYTOKINE CONCENTRATIONS IN GINGIVAL CREVICULAR FLUID
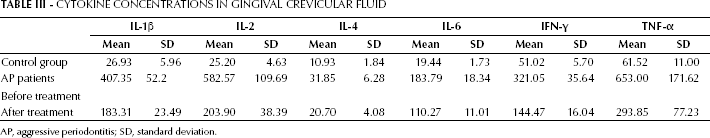
AP, aggressive periodontitis; SD, standard deviation.
Concentrations of cytokines in saliva in response to therapy
IL-1β, IL-2, IL-4, IL-6, IFN-γ and TNF-α concentrations were higher in saliva of AP patients than in saliva of healthy subjects (Tab. IV). Compared with baseline concentrations, the levels of cytokines significantly decreased throughout the course of treatment (p<0.05). IL-1β, IL-2 and IFN-γ decreased most significantly compared to baseline values (2.22-, 2.50- and 2.22-fold, respectively).
CYTOKINE CONCENTRATIONS IN SALIVA
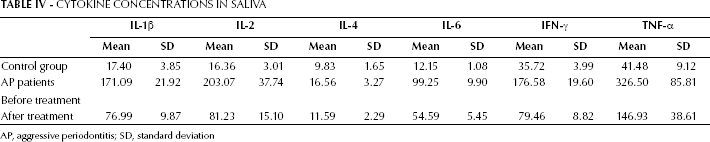
AP, aggressive periodontitis; SD, standard deviation
Saliva was chosen for this study because it can be handled relatively quickly and easily. Cronbach's alphas for cytokines in GCF and saliva were 0.82 for IL-1β, 0.75 for IL-2, 0.78 for IL-4, 0.90 for IL-6, 0.90 for IFN-γ, and 0.88 for TNF-α, which can be regarded as satisfactory.
Discussion
The main goal of the present study was to evaluate and compare cytokines in GCF and saliva and to develop a noninvasive and quick method for assessing cytokine levels in AP patients. GCF, collected from the gingival crevice surrounding the teeth, exists as either a serum transudate or more commonly as an inflammatory exudate. It contains substances from the host such as molecules from blood, contributions from cells and substances from microorganisms in the subgingival and supragingival plaque. The traditional and most common clinical method to collect GCF is the use of micropipettes or precut methylcellulose filter paper strips to isolate and collect fluid from the gingival margin. The fluid will be absorbed on the strips, then eluted and analyzed further. However, this approach is complicated by the need to collect multiple samples throughout the dentition to fully assess the inflammatory response when applied to AP diagnosis. Concern has also been raised about the GCF volume collected by strips. It has been reported that both plaque and saliva on the test strip can influence the volume of fluid that is collected (15), and the GCF volume varies from tooth site to tooth site. Hence, attention was placed on developing and standardizing the collection of GCF (16, 17). In the present study, the periodontal probe technique was modified and the probe was placed on the occlusal surface for the aspiration of GCF. The probing device was inserted at a depth of approximately 80% into the sulcus for drawing the GCF.
Recently, salivary diagnostic tests have come to serve as another diagnostic tool and are being recognized as a central player in a wide variety of oral and systemic diseases, because saliva contains specific soluble biomarkers and is easy to collect and store (18, 19). The inflammatory mediators in whole saliva are derived not only from the periodontium via influx of GCF but also from the mucosa if inflammation is present. Although saliva contains a diluted concentration of biomarkers that may be difficult to detect, attention is placed on evaluating the role of salivary biomarkers for monitoring periodontal health.
Cytokines in GCF and saliva were also evaluated in this study as values of Cronbach's alpha, varying from 0.75 to 0.90. This indicated the reliability of the 2 methods to detect cytokine profiles in GCF and saliva.
Measuring cytokine levels of host mediators is considered time-consuming and technique-sensitive as it will predominantly be influenced by GCF collection. However, cytokines in GCF are considered important biomarkers for AP, because longitudinal clinical trials have identified that the host response plays an important role in the progression of periodontal health (20, 21). Many studies have suggested that proinflammatory cytokines may play an integral role in the etiology of periodontal disease (9, 22). One theory, relative to T-cell function, explains that the host response in periodontal tissues is the balance between the protective role of Th1 cells in conjunction with functional specific antibodies and destructive Th2 profiles, leading to B cell expansion with nonprotector antibodies in lesions (23, 24). Several investigators have analyzed the Th1/Th2 profile in periodontal disease using different methods.
Some studies have shown an increased Th1 response in periodontitis; increased Th2 responses have also been reported and high levels of Th1 cytokines (IL-2, IL-1β, TNF-α and IFN-γ) have been observed (6, 9, 25, 26). In this study, a consistent observation was found in GCF and saliva. The concentrations of IL-1β, IL-2, TNF-α and IFN-γ were statistically significantly higher in AP patients compared with healthy subjects (p<0.05). Furthermore, the levels of inflammatory cytokines were responsive to periodontal therapy.
The Th2 cytokines IL-4 and IL-10 are implicated in suppressing the destructive actions of Th1 and other cell-mediated inflammatory responses (23). Both cytokines can target macrophages and inhibit the release of IL-1, TNF-α, reactive oxygen intermediates and nitric oxide, but they are also growth and differentiation factors for activated B cells, found in increased proportions in progressive periodontal lesions (24). Several studies (6, 27) reported an inverse relationship between the antiinflammatory cytokine IL-4 and the status of periodontal disease. Bastos et al (6) reported that the concentration of IL-4 was higher in the assessed sites of control subjects than in those of AP individuals. In contrast, the levels of IL-4, detected in both GCF and saliva, were higher in patients with AP compared with healthy individuals in this study. In view of these results, it was recently suggested that the dominance of B cells in periodontitis lesions cannot entirely be explained by enhanced Th2 functions (28). Longer-term clinical studies involving a larger population are warranted to confirm the significance of specific GCF and saliva markers for monitoring periodontitis and other lesions in the oral cavity.
In conclusion, this study demonstrated that inflammatory cytokines such as IL-1β, TNF-α, IL-2, IL-6 and IFN-γ dominated in the response to periodontopathogenic bacteria in GCF and saliva from AP patients. Measurement of cytokines in saliva thus may be regarded as a noninvasive and quick method for monitoring periodontal disease activity.
