Abstract
Purposes
This substudy aimed to examine the changes in biomarkers for cardiac injury in patients who received neoadjuvant 5-fluorouracil, epirubicin, cyclophosphamide with concurrent celecoxib (FEC-C).
Methods
Thirty-four female patients with histologically confirmed locally advanced breast cancer preoperatively received 3 cycles of FEC-C (500 mg/m2, 75 mg/m2, 500 mg/m2) with concurrent celecoxib (400 mg bid). Blood samples were drawn from patients on day (D) 0, D3, D21, D42, and D63 (end of therapy), and the serum levels of lactate dehydrogenase (LDH) and plasma levels of cardiac troponin I (cTnI) and N-terminal prohormone brain-type natriuretic peptide (NT-proBNP) were measured with commercially available test kits.
Results
All patients tolerated this regimen well. Neither life-threatening toxicity nor clinical symptoms of cardiac damage were observed. Serum LDH increased significantly from baseline after 3 cycles of FEC-C (p<0.0001), but the change was possibly brought about by chemotherapy-induced liver derangement. However, NT-proBNP decreased significantly (p=0.009), while cTnI increased nonsignificantly (p=0.078) after 3 cycles of FEC-C compared to baseline, although this increase was still regarded as normal.
Conclusions
Short-term use of the FEC-C regimen has proven to be effective in locally advanced breast cancer, with an acceptable cardiac safety profile.
Introduction
Anthracyclines have proven to be more successful in the treatment of various cancers than other classes of chemotherapy and are the frontline chemotherapy treatment for breast cancer. Anthracyclines are widely used in the neoadjuvant setting for treatment of locally advanced breast cancer (LABC) in order to downstage the disease and allow a chance for breast conservation at surgery (1-3). Despite the demonstrated anticancer effects of these chemotherapeutic agents, a major adverse effect that accompanies anthracyclines is cardiotoxicity. In some cases, anthracyclines can lead to congestive heart failure and left ventricular dysfunction. The former is dose dependent, while the latter occurs more frequently in patients with a history of cardiac disease. Therefore dose- and drug-induced cardiotoxicity is a major concern in treating cancer patients with chemotherapy, possibly impairing their prognosis (4, 5). Long-term patient follow-up is needed as the negative cardiac effects of anthracyclines are more notable many years after receiving treatment.
The use of 5-fluorouracil, epirubicin and cyclophosphamide (FEC) as neoadjuvant breast cancer chemotherapy has been shown to be well tolerated and efficacious. In vivo testing of cyclooxygenase-2 (COX-2) inhibition has demonstrated the chemopreventative and chemotherapeutic properties in rodent models of breast diseases (6, 7). Additionally, breast conservation was made possible for some LABC patients whose tumors shrunk to a size of 3 cm or less with the application of FEC chemotherapy regimen before surgery (8, 9).
Cyclooxygenase enzymes are pivotal for the conversion of arachidonic acid to prostaglandins. Typically, the COX-2 enzyme is overexpressed in breast cancer, and its expression correlates with more aggressive disease. Inhibiting COX-2 activity would reduce antiangiogenic and proapoptotic effects and may therefore be of benefit in the prevention and treatment of some cancers, including breast cancer.
Celecoxib, a selective COX-2 inhibitor, has been demonstrated to be useful in counteracting angiogenesis and tumor progression in preclinical and clinical settings (10-13). In patients with familial adenomatous polyposis, 6 months of twice-daily treatment with 400 mg of celecoxib was found to lead to a significant reduction in the number of colorectal polyps (14). In our previous study, a higher pathological response was observed in patients receiving FEC with concurrent celecoxib of 400 mg twice daily compared to patients receiving FEC alone (15). However, the potential cardiovascular risk of prolonged use of nonsteroidal antiinflammatory drugs has posed a question on the use of COX-2 inhibitors in cancer treatment (16-19). The present substudy aimed to detect cardiac injury in LABC patients after receiving combination chemotherapy with celecoxib. For this purpose we determined the changes in cardiac biomarkers.
Materials and Methods
Patient population
Thirty-four patients aged 33-56 years (mean age 45.5 years) were included in this subanalysis. Patients enrolled in the study were histologically proven to have LABC with a tumor size of 3 cm or greater. Additional inclusion criteria were female gender, age older than 18 years, ECOG performance status ≤3 or Karnofsky performance status ≥70%, acceptable liver function (bilirubin <50 μmol/L), acceptable renal function with serum creatinine <150 μmol/L and GFR >40 mL/min, and ability to give written informed consent and follow prescription instructions reasonably well. The exclusion criteria included known sensitivity to celecoxib, major cardiac disease or left ventricular ejection fraction <50%, coronary artery disease, active liver disease, renal impairment, and prior history of other malignancy within 5 years of study entry except basal cell carcinoma of the skin or carcinoma in situ of the uterine cervix, and seropositivity for either HbsAg or anti-HCV or HIV antibodies.
All eligible patients were explained the nature and purpose of the clinical study and consented to inclusion in the trial. Approval was received from the local ethics committee, and the study was conducted at the Division of Breast Surgery, The University of Hong Kong Medical Centre.
Study design and treatment
The 34 LABC patients enrolled in the study received neoadjuvant chemotherapy consisting of 5-fluorouracil 500 mg/m2, epirubicin 75 mg/m2 and cyclophosphamide 500 mg/m2 (FEC) concurrently with the COX-2 inhibitor celecoxib 400 mg bid (FEC-C) as 3-weekly cycles (21 days/cycle). Each patient received 3 cycles of therapy and surgery was performed after the third cycle. For patients who refused to undergo surgery, the last endpoint assessment for inclusion in the study was the 63rd day after commencement of treatment. Three additional cycles of therapy were given after surgery.
Blood samples were drawn from the patients on day (D) 0 (baseline), D3 (cycle 1 D3), D21 (cycle 2 D0), D42 (cycle 2 D21), and D63 (end of therapy) to measure the levels of serum lactate dehydrogenase (LDH), plasma cardiac troponin I (cTnI) and plasma N-terminal proB-type natriuretic peptide (NT-proBNP). The blood samples were collected in plain tubes for serum and in tubes containing ethylenediaminetetraacetic acid for plasma. cTnI was determined using immunoenzymatic fluorescent assay (Stratus II; Dade International Inc., Miami, FL, USA) and its reference values are <0.4 ng/mL (20). NT-proBNP was measured using electrochemiluminescence immunoassay (Elecsys System 1010/2010, ProBNP Kit, Roche, Mannheim, Germany). The measurement of serum LDH was obtained from the local institutional blood biochemistry laboratory.
Statistical analyses
Data in this study are presented as means and standard error of means unless otherwise stated. Statistical analysis was performed using the SPSS version 13.0 software to compare the parameters, and statistical comparisons were performed by the paired t-test. Results were considered statistically significant when p was <0.05.
Results
The 34 LABC patients were consecutively accrued (Tab. I). All patients received neoadjuvant FEC-C for 3 cycles and symptomatic cardiotoxicity was not clinically observed. After 3 cycles of combination therapy with FEC-C, 4 (11.8%) and 20 (58.9%) patients achieved complete and partial clinical responses, respectively. The primary tumor shrunk by an average of 60% in terms of tumor area. Over 80% of patients underwent modified radical mastectomy. Pathological complete response was observed in 3 (8.8%) patients. The average residual tumor size on pathological examination was 2.7±2.2 cm.
PATIENT DEMOGRAPHICS
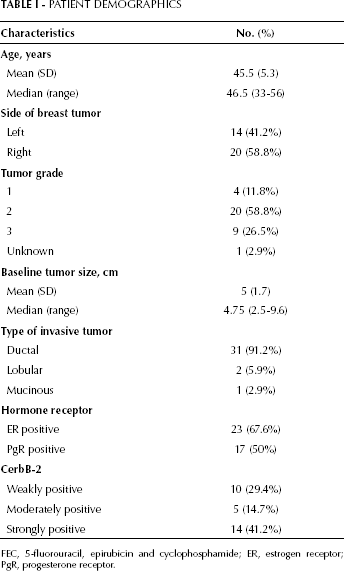
FEC, 5-fluorouracil, epirubicin and cyclophosphamide; ER, estrogen receptor; PgR, progesterone receptor.
Blood samples of the patients were obtained before, during and after receiving treatment. The change in cardiac markers across time points is illustrated in Table II. Across the time points of assessment, the mean cTnI (Fig. 1), NT-proBNP (Fig. 2) and LDH (Fig. 3) levels did not deviate from normal values, which were defined as <0.2 ng/mL, <250 fmol/mL and 313-618 U/L, respectively. Both cTnI and NT-proBNP did not show a significantly increasing trend during treatment, and a significant drop in the mean NT-proBNP level from baseline (240.51 fmol/mL ± 1.97) to FEC-C day 63 (208.02 fmol/mL ± 11.32) was observed (p=0.009). A significant increase in mean LDH from baseline (357.32±21.10) to FEC-C day 63 (521.06±21.19) was observed (p<0.0001). Notably, except for LDH, these specific cardiac markers were still within normal ranges.
CHANGE OF CARDIAC MARKERS ACROSS TIME POINTS
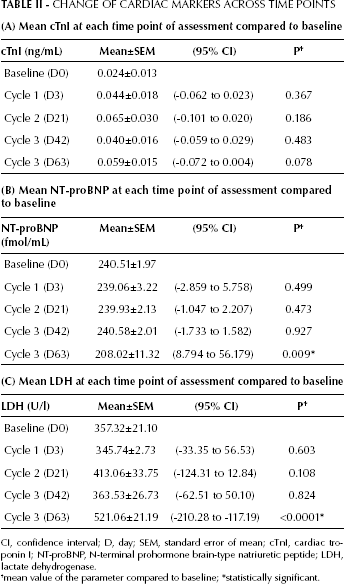
CI, confidence interval; D, day; SEM, standard error of mean; cTnI, cardiac troponin I; NT-proBNP, N-terminal prohormone brain-type natriuretic peptide; LDH, lactate dehydrogenase.
mean value of the parameter compared to baseline;
statistically significant.
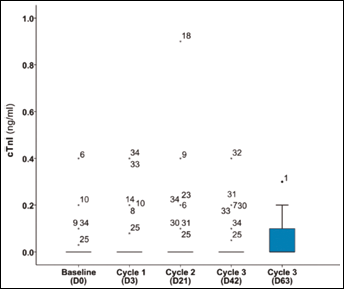
Change of plasma cTnI across time points during neoadjuvant therapy.
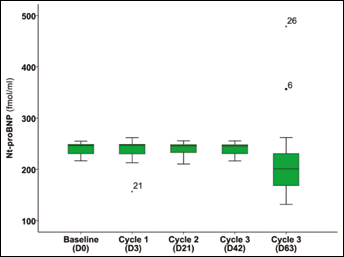
Change of plasma NT-proBNP across time points during neoadjuvant therapy.
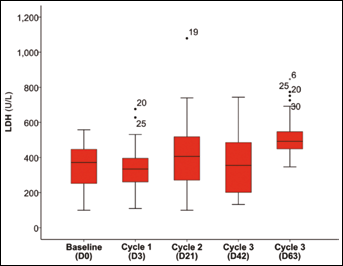
Change of serum LDH across time-point during neoadjuvant therapy.
Discussion
LDH has been used to help diagnose and monitor heart attacks. Following a heart attack, peaked levels of LDH can be observed at 3-4 days and can remain elevated for up to 10 days. Measuring the level of LDH in patients with complaints of several days of chest pain may help doctors to diagnose an episode of heart attack. However, this has now been replaced by more specific markers such as troponin and brain natriuretic peptides. In this study, LDH was also determined as a reference in clinical practice.
Troponin I is an experimental tool in the early diagnosis of cardiac toxicity (18). cTnI is a cardiac regulatory protein and has not been identified outside the myocardium. An increase in the cTnI level is an early sensitive and specific marker of myocardial injury and is predictive of damage in left ventricular function (19). The potential role of cTnI as a predictive factor was studied in 204 patients treated with high-dose chemotherapy for aggressive cancers. Plasma cTnI levels were checked and echocardiography was done after each chemotherapy cycle. It was observed that in patients with normal cTnI levels, left ventricular ejection fraction decreased progressively but then normalized, whereas in patients with high cTnI levels the decrease was greater and persisted longer, up to the end of follow-up (23).
Brain natriuretic peptide (BNP) is the early biomarker of left ventricular dysfunction developing with doxorubicin cardiotoxicity (24). NT-proBNP is a neurohormone synthesized in the cardiac ventricles in response to increased wall tension. An increase in NT-proBNP levels indicates asymptomatic and symptomatic heart failure, as ventricular cells secrete BNP in response to high filling pressures. NT-proBNP is now recognized as a more sensitive marker than BNP (25). In addition, persistently elevated NT-proBNP levels in patients during the course of not-high-dose anthracycline-based chemotherapy is predictive of impaired left ventricular function (26). In this study, during the course of neoadjuvant chemotherapy, the level of NT-proBNP did not increase at all, but a significant drop was observed after patients received chemotherapy treatment. This might not be valid to affirm a cardioprotective effect, but at least no cardiotoxicity was observed clinically along with the steady level of the cardiac marker during chemotherapy. Serial measurement of NT-proBNP during chemotherapy might be useful for early detection of cardiotoxicity, serving as a foundation for preventive or therapeutic management of cardiac dysfunction.
The adverse effects of COX-2 inhibition on the cardiovascular system remain unclear. The selective inhibition of COX-2 under certain conditions may have fatal effects on the risk of thrombosis, while in other circumstances it may actually decrease ischemic events (27). In this study, the combination of anthracycline-based chemotherapy and a COX-2 inhibitor was anticipated to pose a certain level of cardiac risk, which would be reduced in a short-term use situation, e.g. neoadjuvant therapy. The treatment did not change the cardiac biomarkers significantly except for LDH. Both cTnI and NT-proBNP did not show a statistically increasing trend, which was consistent with clinical observations. LDH, however, showed an increasing trend during the course of treatment and was significantly higher at D63 compared to baseline. Such a change should not be a surprise because LDH itself is not specific: it is present in many organ systems and may change under many different conditions. Chemotherapy-induced hepatotoxicity might also cause increased serum LDH levels and, therefore, the change in LDH should be regarded as a reference for clinical practice but not an indicator of cardiac damage.
Neoadjuvant FEC-C is an active and well-tolerated regimen. The short-term use of anthracycline-based chemotherapy with celecoxib, a selective COX-2 inhibitor, for LABC patients before surgery resulted in some changes in cardiac markers. The changes, however, did not outrange the normal values and did not indicate any cardiac damage. Long-term follow-up would be needed to establish whether patients may experience subclinical or clinical toxicity many years after receiving treatment. Large-scale clinical trials need to be conducted to give a full clinical picture of the balance between the anticancer and cardiovascular adverse effects of selective COX-2 inhibition. A phase II study on the neoadjuvant use of FEC followed by docetaxel with concurrent celecoxib in LABC patients was conducted to investigate both the safety and efficacy of the regimen (28). Serial cardiac monitoring is also carried out to examine the left ventricular ejection function and serum cardiac markers (29).
Conclusions
The short-term addition of a COX-2 inhibitor to chemotherapy is clinically feasible with an acceptable cardiac safety profile and potential anticancer effects. Further research into the FEC-C regimen for the treatment of breast cancer in the neoadjuvant setting is warranted.
