Abstract
The present study investigated whether tumor markers such as cell proliferation and steroid receptor status, which have been shown to have relevance for important endpoints (relapse-free and overall survival), can also predict axillary disease in elderly patients with breast cancer. We evaluated 351 consecutive elderly women with breast cancer ≥70 years of age with estrogen receptor (ER)-positive tumors with no palpable axillary nodes, for whom information on cell proliferation determined by the 3H-thymidine labeling index (TLI) and progesterone receptor (PgR) was available. Patients underwent quadrantectomy (70.1%) or quadrantectomy plus radiotherapy (29.9%) without axillary node dissection, followed by adjuvant tamoxifen for at least 2 years. Univariable (cumulative incidence curves) and multivariable analyses (Fine and Gray models) were carried out. After a median follow-up of 16 years, ipsilateral axillary relapse was not related to PgR status but was strongly associated with tumor cell proliferation in both small (pT1) and large (pT2-4b) tumors. Axillary relapse cumulative incidence increased from 1% in patients with low-TLI (≤3%), PgR-positive and pT1 tumors to a maximum of 20% in patients with high-TLI, PgR-negative and pT2-4b tumors. Tumor cell proliferation, determined by TLI at primary surgery, is an important predictor of axillary relapse in elderly ER-positive breast cancer patients and could help to identify patients who should undergo axillary surgery.
Introduction
Axillary lymph node status is no longer considered the most important prognostic factor in the management of breast cancer, as the biological profile of the primary tumor also provides important and reliable prognostic and predictive indications for clinical treatment (1, 2). Sentinel node biopsy is now widely used to stage the axilla in patients with a clinically clear axilla, and axillary clearance is performed only if the sentinel node is metastatic (3-5). However, there is an increasing tendency not to perform axillary clearance in carefully selected patients even when the sentinel node is positive (6). Furthermore, in older patients with early, hormone-responsive breast cancer and a clinically clear axilla, neither axillary dissection nor sentinel node biopsy appears justified (7, 8). The 5-year results from our randomized trial comparing primary surgery with or without axillary dissection in T1N0 breast cancer patients over 65 years of age showed that axillary surgery can be omitted without affecting breast cancer mortality or overall survival (9). Similarly, in our prospective nonrandomized study on a consecutive series of conservatively treated breast cancer patients over 70 years of age with a clinically clear axilla, who did or did not receive axillary dissection, we observed low rates of local-regional failure overall and we found that omission of axillary dissection had no adverse effect on breast cancer mortality after 16 years of follow-up (10). In both studies, over 90% of cancers were estrogen receptor (ER) positive and postoperative tamoxifen was administered to all patients for at least 2 years.
Since the 1980s, biological markers such as hormone receptor status, cell proliferation indices and genome alterations have been investigated for their ability to predict breast cancer outcomes (11) and, more recently, to predict response to systemic treatment (12, 13). High-throughput molecular methods, particularly gene expression profiling, are also showing promise as prognostic and predictive factors in breast cancer (14). However, genomic studies have only occasionally been carried out on tumors from elderly patients (15). Conventional prognostic factors including hormone receptor status, tumor grade and tumor cell proliferation indices are mainly used to plan treatment in older patients (16, 17).
In the present study, we investigated whether cell proliferation evaluated by 3 H-thymidine labeling index (TLI) and progesterone receptor (PgR) status can predict the risk of axillary relapse in a series of elderly patients with ER-positive breast cancer and a clinically clear axilla. We made use of a series of patients prospectively recruited to a nonrandomized study in the period 1987-1992 for whom information on TLI and PgR status was available at the time of surgery. Our aim was to determine whether the 2 markers, singly or together, are capable of predicting axillary relapse in order to rationalize axillary surgery.
Materials and Methods
Patients
The present study was conducted on 351 patients aged ≥70 years with primary invasive breast cancer and a clinically clear axilla treated by conservative surgery without axillary dissection and with adjuvant tamoxifen for at least 2 years. The patients, recruited from January 1987 to December 1992 at the Istituto Nazionale dei Tumori (INT) of Milan, constituted the subset of ER-positive tumor cases with available TLI and PgR information within the group (499 patients) not given axillary dissection in our prospective nonrandomized study to assess the role of axillary dissection in elderly breast cancer patients (18). Recruited patients gave their informed consent, and the study was approved by the INT Institutional Review Board.
Postoperative radiotherapy to the breast, and not to the axilla, was administered to 105/351 patients and started within 4 weeks of surgery. A cobalt unit or a 6-MeV linear accelerator was used to deliver a total of 50 Gy (daily target dose of 2 Gy) to the residual breast parenchyma in 2 opposing tangential fields over 5 weeks, followed by a 10 Gy boost to the tumor bed. Whether or not radiotherapy was given depended on patient preference and the opinion of the treating physician. All patients were prescribed tamoxifen, 20 mg/day, starting within 2 weeks of surgery and continuing for at least 2 years.
Patient follow-up involved clinical assessment every 6 months for the first 5 years, and yearly thereafter. Chest X-ray and mammography were carried out annually, and bone scan was performed every 2 years. Median follow-up was 191 months (interquartile range [IQR] 169-203). At the close of follow-up (June 2006), 81 patients were alive, 49 had died from breast cancer, 15 from another malignancy and 206 from an unrelated condition. First unfavorable events were as follows: 24 ipsilateral axillary relapses, 28 ipsilateral breast tumor recurrences, 31 distant metastases, 6 contralateral tumors, and 20 other malignancies.
Biological marker determinations
TLI was determined prospectively on the primary tumor at the time of surgery. The Reporting Recommendations for Tumor Marker Prognostic Studies (REMARK) (19) were followed since this ancillary cell proliferation study had already been detailed in the trial protocol as well as in the patient informed consent and was approved by the INT Institutional Review Board and the Independent Ethics Committee. Immediately after surgery, fresh tumor samples were sent to the laboratory, and part of the material was processed immediately for TLI determination and some was frozen in liquid nitrogen and stored at −80°C for ER and PgR analyses.
TLI was determined with a commercial kit (Euroframe, Asti, Italy) using a widely used procedure at that time, which had produced consistent results in several studies conducted in different laboratories (20-22). Briefly, tumor specimens were cut into fragments of a few mm3 and incubated in a medium containing 3 H thymidine for 1 hour at 37°C under normal oxygen conditions. The fragments were then fixed in neutral formalin and embedded in paraffin, and 4-μm-thick sections were cut and stained with hematoxylin and eosin at 4°C. Autoradiography was performed by the stripping-film technique with a 10-day exposure time at 4°C. Over 3,000 cells in different parts of the tumor were scored. TLI was expressed as the ratio of cells with overlying silver grains to the total number of cancer cells (11). Counts were performed by 2 independent observers. Tumors with TLI >3% were considered rapidly proliferating and those with TLI ≤3% slowly proliferating (22). Quality control procedures were applied periodically in compliance with the National Quality Control Program of the Italian Society of Basic and Applied Cell Kinetics (SICCAB) and the Italian National Research Council (23-25) to ensure the intercenter reproducibility of subjective evaluations.
ER and PgR were assayed by the dextran-coated charcoal method according to the European Organization for Research and Treatment of Cancer standards (26). The quality control procedures used were those recommended by Ronchi et al (27). Receptor levels were expressed as fmol/mg cytosol protein. Tumors with ER ≥10 or with PgR ≥25 fmol/mg cytosol protein were considered ER and PgR positive, respectively.
Statistical methods
The SAS statistical package (28) and R software (29) were used to carry out the analyses.
The relation between PgR status (positive versus [vs] negative) and TLI (≤3% vs >3%) was assessed by the chi-square test. Spearman's correlation coefficient was used to assess the association between PgR and TLI as continuous variables. The main study endpoint was axillary relapse, whereas breast cancer mortality was analyzed as the secondary endpoint. Date of surgery was taken as time 0 for all analyses; in the absence of any event, time was censored at June 30, 2006. Analyses of the effects of study variables on the endpoints took account of competing risks; for axillary relapse, competing events were ipsilateral breast tumor recurrence, distant metastasis, contralateral tumor, other malignancy, and death from unrelated conditions; for breast cancer mortality, competing events were death from other malignancy or death from unrelated conditions (30). Crude (unadjusted) cumulative incidences of axillary relapse and breast cancer mortality were estimated in relation to TLI and PgR status (binary variables). We also estimated the cumulative incidence curves in relation to a binary variable set up for analyzing combined information on TLI and PgR as follows: TLI >3% and PgR-negative vs the other 3 combinations between PgR and TLI categories (i.e., TLI >3% and PgR-positive, TLI <3% and PgR-positive/negative). The Gray test was used to compare cumulative incidence curves (31).
Multivariable Fine and Gray modeling (32) was also used to investigate the independent effects of TLI and PgR on the cumulative incidence of axillary relapse and breast cancer death, adjusting for postoperative radiotherapy (performed vs not performed), pathological tumor size (pT1 vs pT2-4b) and histology (lobular/other vs ductal). Different models were fitted including the above described binary variable combining information on TLI and PgR, together with the 3 adjustment variables. Significance was determined using the 2-sided Wald test.
To characterize the prognostic potential of TLI we used the approach of Heagerty et al (33), implemented in the R (29) library survivalROC. The approach allows evaluation of time-dependent accuracy measures (sensitivity, specificity, and area under the receiver operating curve [ROC]), which are extensions to survival data of the accuracy measures typically applied to binary data.
P values <0.05 were considered significant.
Results
Patient and tumor characteristics
Baseline characteristics of the 351 ER-positive elderly breast cancer patients are shown in Table I. Median age was 77 years (range, 74-81); 70.1% of the patients underwent quadrantectomy alone and about 30% also had postoperative radiotherapy to the residual breast. Fifty-six per cent of patients had pT1 tumors and 68% had infiltrating ductal carcinoma. About 3 quarters (74.4%) of patients had PgR-positive tumors and 1 quarter (25.6%) had PgR-negative disease. Moreover, 54% of patients had a low proliferative activity tumor and 46% a high proliferative activity tumor. The correlation between PgR and TLI as continuous variables was very weak (correlation coefficient −0.11). The proportion of patients who received radiotherapy was similar in the TLI ≤3% (58/190, 30.5%) and TLI >3% groups (47/161, 29.2%), as well as in the PgR-positive (80/261, 30.7%) and PgR-negative (25/90, 27.8%) groups.
PATIENT AND TUMOR CHARACTERISTICS IN 351 ER-POSITIVE ELDERLY BREAST CANCER PATIENTS
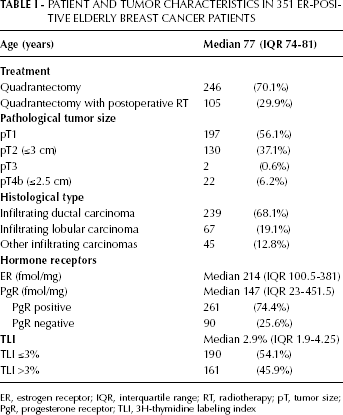
ER, estrogen receptor; IQR, interquartile range; RT, radiotherapy; pT, tumor size;
PgR, progesterone receptor; TLI, 3H-thymidine labeling index
Primary endpoint: axillary relapse
The 16-year crude cumulative incidence of axillary relapse as first event in the TLI >3% group (11.8%, 95% confidence interval [CI] 7.7%-18.0%) was about 6-fold that of the TLI ≤3% group (2.1%, CI 0.8%-5.6%, p=0.0002). In both groups, axillary relapse occurred during the first 7 years of follow-up (Fig. 1). The area under the receiver operating curve (AUC) evaluated at 16 years showed a good discriminative accuracy of TLI (AUC=73.3%) with sensitivity and specificity of 80.0% and 57.3%, respectively, at the 3% cutoff value. ROC data also indicated that the conventionally used 3% cutoff produced the best compromise between sensitivity and specificity (data not shown).
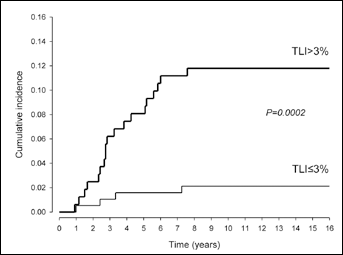
Crude cumulative incidence of axillary relapse in 351 elderly ER+ breast cancer patients according to tumor proliferation rate (TLI ≤3% vs TLI >3%).
As illustrated in Figure 2, TLI had a marked effect on axillary relapse rates irrespective of tumor size. In particular, for pT1 disease, the 16-year crude cumulative incidence of axillary relapse in tumors with TLI >3% was more than 10-fold that of TLI ≤3% tumors: 9.3% (CI 4.8%-18.1%) vs 0.9% (CI 0.1%-6.4%, p=0.003). For pT2-4b disease, the cumulative incidence of axillary relapse was still significantly higher (about 4-fold) for TLI >3% than TLI ≤3% tumors: 14.7% (CI 8.5%-25.4%) vs 3.9% (CI 1.3%-11.8%, p=0.019). Furthermore, among pT1 patients with TLI ≤3% disease, axillary relapse as first event occurred only once and within the first year of follow-up, whereas with TLI >3%, 21 axillary relapses occurred during the first 6 years. In patients with pT2-4b disease, axillary relapse was observed during the first 7 years in both cell proliferation tumor subgroups.
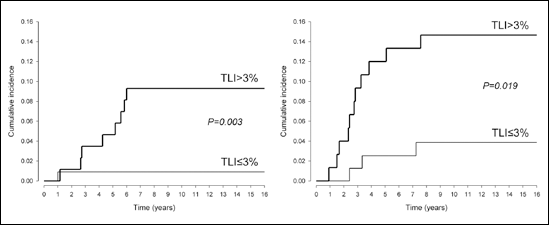
Crude cumulative incidence of axillary relapse in elderly ER+ breast cancer patients with pT1 disease (left panel) and pT2-4b disease (right panel) according to tumor proliferation rate (TLI ≤3% vs TLI >3%).
PgR status was not significantly related to the cumulative incidence of axillary relapse in the overall series or in the 2 subgroups defined by pathological tumor size (Tab. II).
SIXTEEN-YEAR CRUDE CUMULATIVE INCIDENCE OF AXILLARY RELAPSE ACCORDING TO PgR STATUS AND pT OF THE PRIMARY TUMOR
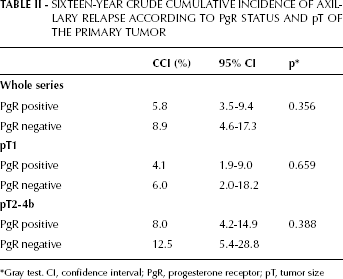
Gray test. CI, confidence interval; PgR, progesterone receptor; pT, tumor size
TLI predicted axillary relapse in both PgR-positive and negative disease (Tab. III). In particular, the axillary relapse rate was about 5-fold higher in the TLI >3% group than in the TLI ≤3% group and appeared independent of PgR status. When the association of TLI and PgR with axillary relapse was investigated according to tumor size (Tab. III), the axillary relapse rate for pT1 cancers was very low (0% and 1%) when TLI was ≤3% and significantly higher in TLI >3% groups (9.1% and 9.7%). Again, axillary relapse was independent of PgR status. For pT2-4b tumors, the axillary relapse rate was lower (3.5%) in PgR-positive TLI ≤3% cases; 5.0% for those with PgR-negative TLI ≤3% disease, around 13% in PgR-positive TLI >3%, and 20% in those with PgR-negative TLI >3% disease. Nevertheless, 95% CI for all these cumulative incidence estimates were wide, and differences between the groups were not significant.
SIXTEEN -YEAR CRUDE CUMULATIVE INCIDENCE OF AXILLARY RELAPSE ACCORDING TO TLI, PgR AND pT OF THE PRIMARY TUMOR
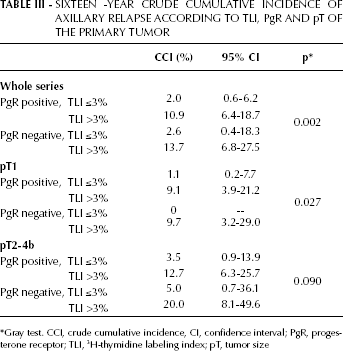
Gray test. CCI, crude cumulative incidence, CI, confidence interval; PgR, progesterone receptor; TLI, 3 H-thymidine labeling index; pT, tumor size
In the multivariable Fine and Gray model, TLI was a significant independent predictor of axillary relapse (p=0.002), whereas PgR (p 0.425), radiotherapy (p=0.072), pT (p=0.916) and histology (p=0.103) were not.
Secondary endpoint: breast cancer mortality
Both TLI and PgR as independent predictors were not significantly associated with breast cancer mortality. The 16-year crude cumulative incidence of breast cancer mortality was 16.2% (CI 11.4%-23.1%) in TLI >3% cases compared with 11.2% (CI 7.5%-16.8%) in TLI ≤3% cases (p=0.130). Regarding PgR status, the 16-year crude cumulative incidence was 16.9% (CI 10.6%-26.9%) in PgR-negative compared with 12.4% (CI 8.9%-17.2%) in PgR-positive cases (p=0.247). Such a result was confirmed at multivariable analysis by Fine and Gray modeling with adjustment for radiotherapy, tumor size and histology, in which neither TLI nor PgR were significantly related to breast cancer mortality (data not shown).
Combined analysis of TLI and PgR showed that TLI >3% and PgR-negative cancers were associated with a higher incidence of breast cancer mortality (23.8%, CI 14.4%-39.5%) than cancers of the other 3 TLI and PgR combinations taken together (11.8%, CI 8.6%-16.1%, p=0.021). The multivariable analysis including the binary variable distinguishing the 2 categories TLI >3%/PgR-negative vs others confirmed the above results showing that mortality was significantly worse in the former category (p=0.028).
Discussion
In our retrospective series of elderly patients with ER-positive breast cancer and clinically clear axilla treated by conservative surgery without axillary dissection and tamoxifen for at least 2 years, we found that high tumor cell proliferation was significantly associated with a high incidence of ipsilateral axillary relapse after 16 years of follow-up, i.e., 11.8% compared with 2.1% (p=0.0002) in patients with slowly proliferating tumors. Accuracy measures determined by ROC analysis (AUC=73.3%, sensitivity 80.0%, specificity 57.3% at the 3% cutoff) also suggested that TLI could be useful in predicting ipsilateral axillary relapse. Furthermore, by multivariable modeling, high TLI emerged as an independent predictor of axillary relapse after adjustment for tumor size, histological type, PgR status and postoperative radiotherapy. When TLI was considered according to PgR status, a graded increase in axillary relapse was observed, ranging from 2% when both markers were favorable to 14% when they were both unfavorable (p=0.002). A similar graded increase in risk with the 2 markers was also evident in the pT1 and pT2-4b categories separately, so that the axillary relapse incidence reached 20% in the most unfavorable category (pT2-4b, TLI >3%, PgR-negative).
We also found that TLI and PgR status were significantly related to breast cancer mortality. Specifically, although the multivariable model found that none of the variables investigated was on its own significantly associated with mortality, a model comparing TLI >3% plus PgR-negative cases with all other 3 TLI and PgR categories combined showed that mortality was significantly higher (p=0.028) in the former, most unfavorable group. This finding seems to support the hypothesis that antiestrogen treatment is likely to be most effective against ER-positive tumors that are also PgR-positive and have a low proliferation rate (34).
The ability of tumor proliferation rate and PgR status to predict ipsilateral axillary relapse in the absence of axillary dissection has, to our knowledge, not been previously investigated. This contrasts with the numerous studies that have investigated characteristics associated with the presence of axillary metastasis. Such characteristics include tumor size, tumor grade, multifocality, vascular and lymphatic invasion (35), hormone receptor status and tumor proliferation activity (36, 37), and more recently, molecular markers (38). No molecular marker has been shown to be sufficiently accurate to obviate the need for formal axillary staging using sentinel node biopsy or axillary clearance (38).
An added value of our study consists in its prospective planning of biomarker determination on tumor specimens, which may ascribe a level of evidence to our results. In fact, on the basis of criteria recently updated by Simon et al (39), the determination of tumor proliferative activity was included in a prospective, nonrandomized trial not specifically designed to address it. In addition, the cell proliferation assay was prospectively carried out, as an active nucleotide incorporation requires viable tissue, using standard operating procedures and quality control assessment for the in situ evaluation (23). Tumor proliferation rate is one of the most extensively investigated biological properties of breast cancers because of its established association with disease progression and metastatic spread (20-22). Several approaches have been used in the past to assess tumor proliferation activity in breast cancer, including mitotic index (40), flow cytometry determination of DNA content in different phases of the cell cycle, particularly the S-phase, and the fraction of cells incorporating tritiated thymidine (TLI) or the thymidine analog bromodeoxyuridine (41). As regards the measure of mitotic activity (volume/corrected mitotic index, mitotic activity index, and mitotic index), its prognostic role has been thoroughly investigated (42, 43) and it is currently included in all pathological grading systems since it represents the driving prognostic force of grade systems in breast cancer. The Ki67 nuclear antigen is currently the most widely determined indicator of breast cancer cell proliferation. Ki67 is present in all active phases of the cell cycle but is absent in G0 cells (44). However, the immunohistochemical techniques used to determine the marker vary widely, as do the methods of interpreting findings, which limits Ki67's value in disease prognosis and in predicting response to treatment, notwithstanding the guidelines recently published to overcome some of these limitations (44). The other methods also have inherent advantages and disadvantages and do not always correlate well with one another. In fact, moderate or poor concordance has been observed not only between indices detecting cells in different phases of the cell cycle (S-phase fraction and Ki-67), but also between those that mark the same phase of the cycle (S-phase fraction and TLI) (41). Other approaches (such as cyclin E (45), minichromosome maintenance protein 2, geminin and Aurora kinese A) have been recently proposed, with promising results that need further validation (46).
In the present study, dealing with long-term results of a series of consecutive patients prospectively recruited to a nonrandomized study, we decided to investigate the prognostic role of cell proliferation and PgR for several reasons, including their a priori consideration in the trial protocol and thus their prospective evaluation at the time of surgery, and their association with clinical outcome following endocrine treatment, since all the women who entered our study received adjuvant tamoxifen. Moreover, we used TLI to assess tumor cell proliferation because it was a well-established method at the end of the 1980s and is the one with which we have ample experience (23). The interest in TLI was to find and use a quantitative variable, whose interlaboratory reproducibility was assured by a national quality control program, instead of biological features such as tumor grade, which classifies tumors into 3 subgroups (grade 1, 2, and 3) – with grade 2 including about half of the tumors – with variable clinical behavior and is characterized by some lack of reproducibility.
However, certain limitations should be noted in our study. Lymphatic and vascular invasion were not routinely assessed at the time of patient accrual and, in addition, we could not evaluate the expression of HER2 in archival material, since at the time of patient recruitment (from 1987 to 1992) the fixative procedure used in the pathology department was Bouin's solution, which preserves tissue histology very well but provides unreliable results for determining the expression of the HER2 protein.
Despite these reasons of concern, our finding that high TLI is significantly associated with a high rate of ipsilateral axillary relapse is consistent with previous findings that TLI predicts distant metastases and long-term clinical outcomes (11, 20-22). We conclude that further studies, particularly on younger, and possibly also ER-negative patients, are warranted to investigate whether tumor proliferation rate may be able to predict axillary disease with sufficient accuracy to indicate whether axillary surgery should be performed electively.
Several issues, including those reported in the present study, support the importance of evaluating tumor cell proliferation, whose powerful prognostic relevance clearly emerged even within molecular signatures derived from high-throughput analyses (47), mainly for luminal (ER+) breast cancers. In order to provide tissue-based read-out of tumor cell proliferation, which can be feasible in all the cases and robust in terms of prognostic power, much efforts are nevertheless still required to identify biomarkers whose analytical procedure can be easily standardized, and whose results can be a priori challenged in a head-to-head comparison with classical methods to evaluate proliferative activity.
