Abstract
Background
The prognostic significance of immunocytochemical analysis of tumour vascular endothelial growth factor (VEGF) and its 2 receptors, VEGF-R1 and VEGF-R2, remains incompletely investigated in patients with oesophagogastric cancer.
Methods
Patients undergoing surgical resection were prospectively recruited between February 1999 and August 2000. Immunocytochemical analysis of VEGF, VEGF-R1 (Flt-1) and VEGF-R2 (Flk-1/KDR) was undertaken using validated techniques. Patients were followed up over a 10-year period using the Northern Ireland Cancer Registry.
Results
Sixty-one patients were recruited (male=45, 73.8%) with a median age of 66.0 years (range 39-83). Forty-seven (77.0%) adenocarcinomas and 14 (23.0%) squamous cell carcinomas were resected. UICC tumour staging was: stage I=14.7%, II=24.6%, III=54.1% and IV=6.6%. VEGF, VEGF-R1 and VEGF-R2 were over-expressed in tumour epithelial cells. VEGF-R2 expression was decreased in the presence of lymphovascular invasion and higher tumour grade. The 10-year survival rate was 19.7% (n=12) with a median follow-up of 808 (IQR 356-2313) days. On univariate analysis only lymphovascular invasion significantly predicted poor prognosis in this cohort (p=0.05).
Conclusion
VEGF, VEGF-R1 and VEGF-R2 were over-expressed in tumour epithelial cells. VEGF-R2 expression was decreased in the presence of more aggressive pathological variables. Larger studies are required to assess the prognostic significance of these biomarkers in oesophagogastric cancer.
Introduction
Angiogenesis plays a fundamental role in the growth of solid tumours and the subsequent development of metastases (1). Vascular endothelial growth factor (VEGF) is a potent pro-angiogenic cytokine that is important in regulating both normal and pathological angiogenesis (2). The VEGF family consists of 5 isoforms: VEGF-A (known as VEGF), VEGF-B, VEGF-C, VEGF-D, and placental growth factor. VEGF signals primarily through the receptor tyrosine kinases VEGF receptor 1 (VEGF-R1, Flt-1) and 2 (VEGF-R2, Flk-1/KDR). Experimental findings postulate that VEGF-R1 is primarily not a signalling receptor, but rather a ‘decoy’ receptor, able to regulate the activity of VEGF by rendering the ligand less available to VEGF-R2. The interaction of VEGF with VEGF-R2 is therefore felt to be the critical requirement leading to the induction of the full range of biological responses attributed to it (3).
Tumour VEGF is over-expressed in 30%-60% of patients with oesophageal carcinoma. In the subgroup of patients with squamous cell carcinoma it correlates with both lymph node metastases and outcomes (4, 5). Conversely, while over-expression of VEGF correlates with the transition from Barrett's oesophagus to high-grade dysplasia and carcinoma in situ to locally advanced disease, it does not appear to correlate with aggressive tumour characteristics or patient outcome in oesophageal adenocarcinoma (4, 5).
The role of VEGF receptor expression remains incompletely investigated in patients with oesophagogastric cancer. In oesophageal cancer no studies have been undertaken assessing the expression of VEGF-R1 or VEGF-R2 and in gastric cancer there are conflicting results about the prognostic significance (6, 7). The aim of this study was therefore to further investigate the role of expression of VEGF and its receptors in patients with cancer of the oesophagus and oesophagogastric junction.
Methods
Patients with cancer of the oesophagus or oesophagogastric junction undergoing surgical resection were prospectively recruited from 3 centres in Northern Ireland consecutively between February 1999 and August 2000. No patient had neoadjuvant chemotherapy. Pathological staging was classified according to the Union Internationale Contre le Cancer (UICC) tumour-node-metastasis (TMN) classification (8).
Tissue blocks randomly obtained from each tumour specimen were embedded in paraffin wax (Surgipath, IL, USA) before 4-μm serial sections were cut onto amino propyl tri-ethoxy silane (APES) (Sigma-Aldrich, UK) coated slides. For each tumour block the same antigen was always assessed on the same tissue section. The first 3 serial sections were assigned to antibodies reacting with VEGF, VEGF-R1 and VEGF-R2 respectively to enable correlation of VEGF expression with its 2 receptors. Serial sections were used to minimise variation in antigen expression arising from a potentially heterogeneous distribution of VEGF and its receptors within an individual tumour. Sections 4 and 5 were assigned to act as negative control sections and were stained with immunoglobulin that was non-specific for the antigen in question to ensure that no background staining took place.
Control samples were processed to determine if the expression of VEGF or its receptors was up-regulated in tumour cells compared with normal oesophageal cells. Immunocytochemical expression of VEGF, VEGF-R1 and VEGF-R2 was assessed in sections of morphologically normal oesophagus. Single tissue blocks were taken from macroscopically normal areas of the proximal oesophageal wall (n=14) and processed in the same way as tumour blocks.
The basic protocol for immuno-localisation of all 3 antigens was identical and was based on a standard streptavidin-biotin complex (StABC) immunoperoxidase method for paraffin wax-embedded sections (Dako, CA, USA). Endogenous peroxidases were suppressed using 3% alcoholic hydrogen peroxide (BDH, UK). A microwave protein digestion pre-treatment regime was then instigated. Slides were added to 500 mL citrate buffer (0.01 M, Sigma, UK) at pH 6.0 and heated in a standard domestic microwave oven, set on full power (850W), for 22 minutes. Following a cooling period of 20 minutes, the slides were washed with water and flooded with Trisbuffered saline (TBS). Endogenous biotin was blocked by instigating an avidin/biotin block (Vector Labs, UK). Slides were dried and avidin D solution (1:5 dilution) (Vector Labs, UK) added. At this stage non-specific immunoglobulin binding was also blocked by concurrently adding 0.25 μg/mL of non-immune goat serum (Dako, CA, USA). Tissue sections were then incubated overnight at 4°C in the presence of the appropriate antibody (rabbit polyclonal antibody to VEGF [VEGF (A-20)]: sc-152, 200 μg/mL; rabbit polyclonal antibody to VEGF-R1 [Flt-1] [Flt-1 (C-17)]: sc-316, 200 μg/mL; mouse monoclonal antibody of sub-class IgG1 to VEGF-R2 [Flk-1] [Flk-1 (A-3)]: sc-6251, 200 μg/mL, Santa Cruz Biotechnology Inc., CA, USA and biotin solution [1:5 dilution] [Vector Labs, UK]).
The negative control of VEGF (A-20) and Flt-1 (C-17) antibodies was derived from a solution of normal rabbit immunoglobulin (rabbit polyclonal IgG, 15 g/L, Dako, CA, USA). Similarly, a mouse IgG1 class-specific immunoglobulin (mouse monoclonal IgG1, 100 μg/mL, Dako, CA, USA) was used to ensure that staining on slides treated with antibody to VEGF-R2 (Flk-1) was specific.
Two observers independently assessed tissue sections microscopically. Each tumour tissue section was assessed for expression of the appropriate antigen by tumour cells, vascular endothelial cells and stromal fibroblasts. The method used to assess the degree of antigen expression in each of the 3 cellular compartments was that previously described by Douglas-Jones et al (9). Within each cellular compartment (tumour cells, vascular endothelial cells and stromal fibroblasts), the total percentage of positive cells was assessed (0%-100% in 5% intervals). Then the percentage of weakly, moderately and strongly staining cells were assessed, such that the sum of these categories equaled the overall percentage of positivity. An immunocytochemical score was then calculated as follows: score (out of 300) = (1 × percentage of weak positivity) + (2 × percentage of moderate positivity) + (3 × percentage of strong positivity) (9). Scoring was recorded independently with the mean value representing the final awarded score. If there was a discrepancy greater than 30 (10% of 300), the slide was re-examined via a double-headed light microscope and consensus was achieved.
Patients were followed up until May 2010 through analysis of the Northern Ireland Cancer Registry supplemented with additional data from the patient's general practitioner as required. The cause and date of death were recorded for each patient. Further analysis of each patient's oncology notes was also undertaken to identify those patients who received a course of adjuvant or palliative oncological therapy.
Data were expressed using median values (with interquartile range [IQR]) where appropriate. Spearman's rank testing was used to assess the correlation between the expression of VEGF and its receptors within the compartments. The Mann-Whitney U test was used to analyse the association between the study variables and standard clinicopathological features. Survival analysis was based on immunocytochemical scores from the tumour epithelial cell compartment. Univariate survival analysis was calculated using Cox's proportional hazard model. Survival analysis was calculated using the Kaplan-Meier estimation method with the log-rank test to assess the equality of survivorship function according to patient characteristics. All statistical tests were 2-sided and differences were considered significant if the p value was <0.05. Statistical analysis was performed using the SPSS statistical package (version 12, SPSS Inc., Chicago, IL, USA). Informed consent was obtained from all patients and ethical approval was obtained from the research ethics committee of the Queen's University of Belfast (application number 1/99).
Results
Patient demographics
Sixty-one patients were recruited, 45 (73.8%) were men. The median age was 66.0 years (IQR 60.5-73.5).
Pathological features
The tumour was located in the middle third of the oesophagus in 3 patients (4.9%), in the lower third in 20 (32.8%) and in the oesophagogastric junction in 38 (62.3%). Forty-seven adenocarcinomas (77.0%) and 14 squamous cell carcinomas (23.0%) were resected. Histopathological assessment included T-stage: T1=6 (9.8%), T2=14 (23.0%) and T3=41 (67.2%). UICC tumour staging was stage I=9 (14.7%), stage II=15 (24.6%), stage III=33 (54.1%) and stage IV=4 (6.6%). Three tumours (4.9%) were well differentiated, 30 (49.2%) moderately differentiated and 28 (45.9%) were poorly differentiated. Forty-one (67.2%) cases were lymph node positive, 50 (82.0%) had lymphovascular invasion, and 24 (39.3%) had perineural invasion.
VEGF, VEGF-R1 and VEGF-R2
In cancers of the oesophagus and oesophagogastric junction, immunoreactivity of VEGF, VEGF-R1 and VEGF-R2 was identified in the cytoplasm of tumour cells, endothelial cells and stromal fibroblasts. All oesophagogastric tumours expressed VEGF and VEGF-R1, while 60 (out of 61) tumours expressed VEGF-R2. The median (IQR) expression of the 3 antigens in the various tissue compartments is tabulated for the 61 subjects in Table I. Tumour cell immunoreactivity of VEGF, VEGF-R1 and VEGF-R2 was significantly increased in comparison to non-keratinising squamous mucosa on control oesophageal sections (Tab. II). There was no significant difference, however, in the immunoreactivity of VEGF, VEGF-R1 and VEGF-R2 in tumour endothelial cells and stromal fibroblasts when compared to immunoreactivity in the same cellular compartment of sections from the macroscopically normal oesophagus.
IMMUNOCYTOCHEMICAL EXPRESSION OF VEGF, VEGF-R1 AND VEGF-R2 IN OESOPHAGOGASTRIC TUMOUR SECTIONS (N=61)
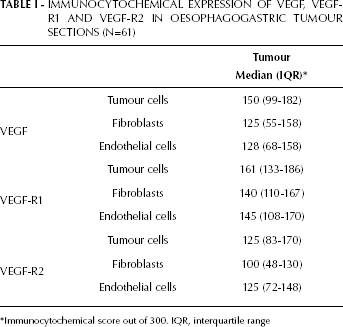
Immunocytochemical score out of 300. IQR, interquartile range
TUMOUR SECTION IMMUNOCYTOCHEMICAL SCORE VERSUS NORMAL OESOPHAGEAL IMMUNOCYTOCHEMICAL SCORE (N=14)
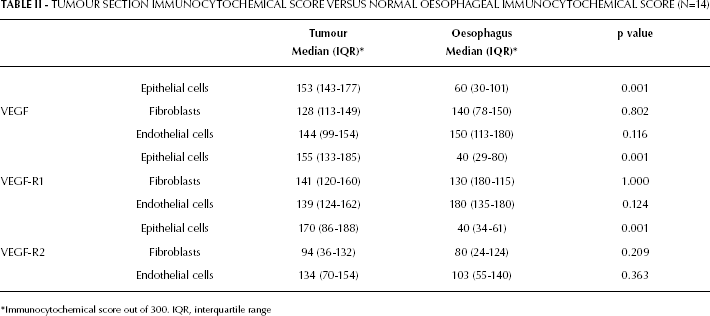
Immunocytochemical score out of 300. IQR, interquartile range
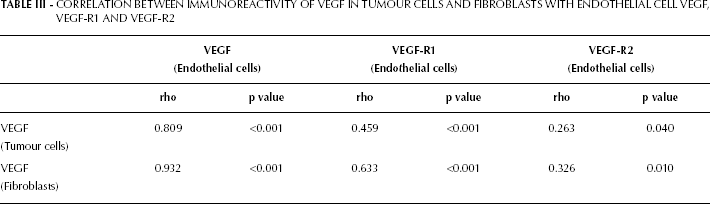
In tumour epithelial cells, immunoreactivity of VEGF correlated with the expression of VEGF-R1 (rho=0.447, p<0.001) and VEGF-R2 (rho=0.424, p=0.001). Expression of VEGF in both tumour epithelial cells and fibroblasts correlated with receptor expression in endothelial cells as summarized in Table III.
CORRELATION BETWEEN IMMUNOREACTIVITY OF VEGF IN TUMOUR CELLS AND FIBROBLASTS WITH ENDOTHELIAL CELL VEGF, VEGF-R1 AND VEGF-R2
The age and sex of the patient did not influence immunocytochemical expression of either VEGF or its 2 receptors. The relationship between clinicopathological features and tumour cell expression of VEGF, VEGF-R1 and VEGF-R2 is expressed in Table IV. Although there was no significant difference in expression of VEGF, VEGF-R1 and VEGF-R2 between adenocarcinoma and squamous cell histological sub-types, there was a tendency to higher immunoreactivity of VEGF in adenocarcinoma tumour cells compared to squamous tumour cells (155 [102-187] vs 123 [73-152], p=0.06). Higher T stage and the presence of lymph node metastases were not associated with an increased expression of VEGF or its receptors. VEGF immunoreactivity was also unaffected by tumour grade. In contrast, immunocytochemical expression of VEGF-R2 was significantly lower in the presence of lymphovascular invasion while expression of both receptors generally decreased as tumour grade increased from low to high grade (i.e. well to less differentiated tumours).
RELATIONSHIP BETWEEN CLINICOPATHOLOGICAL FACTORS AND VEGF, VEGF-R1 AND VEGF-R2
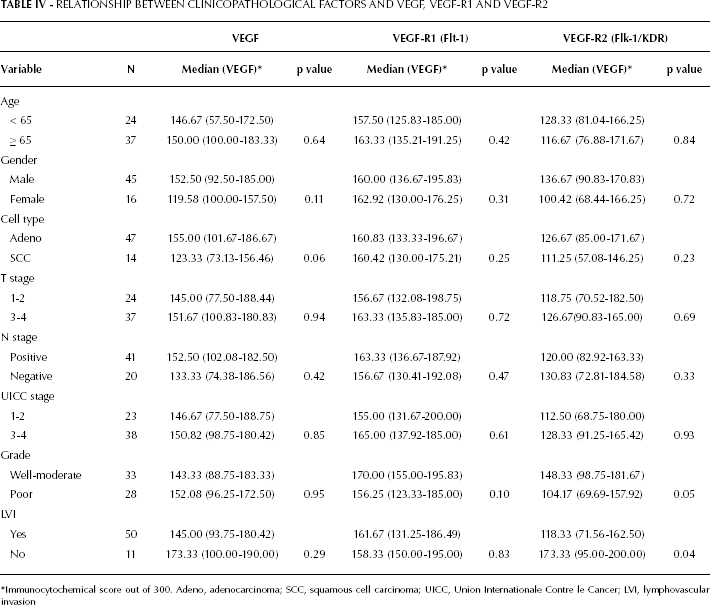
Immunocytochemical score out of 300. Adeno, adenocarcinoma; SCC, squamous cell carcinoma; UICC, Union Internationale Contre le Cancer; LVI, lymphovascular invasion
Oncological management
Four patients (6.6%) received adjuvant chemotherapy and 2 patients (3.3%) required adjuvant radiotherapy. During follow-up, 3 patients (4.9%) were administered palliative chemotherapy, 3 (4.9%) palliative radiotherapy and 3 (4.9%) were administered both modalities for disease recurrence.
Survival
Overall 5-year and 10-year survival rates were 29.5% (n=18) and 19.7% (n=12) respectively with a median follow-up of 808 (IQR 356-2313) days. Using Kaplan-Meier survival analysis there was no difference in long-term outcome comparing values greater or less than the median for VEGF (521 vs 947 days, p=0.93), VEGF-R1 (535 vs 808 days, p=0.99) and VEGF-R2 (973 vs 543 days, p=0.48). Univariate survival analysis of clinicopathological variables and the described immunocytochemical markers is summarised in Table V. Although a trend towards decreased survival was observed for patients with positive lymph nodes (p=0.08) and advanced UICC stage (0.09), only lymphovascular invasion significantly predicted poor prognosis in this cohort (p=0.05).
UNIVARIATE SURVIVAL ANALYSIS OF CLINICOPATHOLOGICAL AND STUDY VARIABLES INCLUDING HAZARD RATIOS AND 95% CONFIDENCE INTERVALS FOR 10-YEAR SURVIVAL
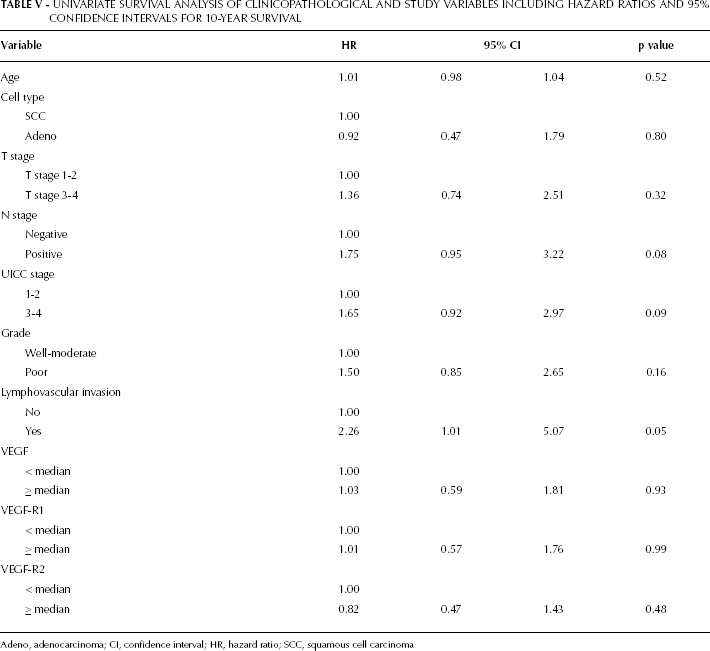
Adeno, adenocarcinoma; CI, confidence interval; HR, hazard ratio; SCC, squamous cell carcinoma
Discussion
VEGF and its 2 receptors were detected by immunocytochemistry in tumour cells, stromal fibroblasts and vascular endothelial cells of patients with oesophagogastric cancer. In comparison to normal oesophageal epithelium, VEGF, VEGF-R1 and VEGF-R2 were up-regulated in oesophagogastric cancer cells. No significant difference was observed between VEGF, VEGF-R1 and VEGF-R2 expression in tumour stromal fibroblasts and endothelial cells in comparison to control sections taken from macroscopically normal oesophagus.
Expression of VEGF receptors on tumour cells has been described previously and VEGF may behave as an autocrine growth factor for tumour cell lines that also express VEGF receptors in tandem with the growth factor itself (10-12). Demonstration of autocrine growth pathways in certain tumours suggests that VEGF may not have its biological mode of action solely on the tumour vasculature, but also on the tumour cell population itself via VEGF-R1 and VEGF-R2 (12, 13). In this study, immunoreactivity of VEGF in oesophagogastric tumour cells correlated with immunocytochemical expression of both receptors, suggesting a possible autocrine function for VEGF via either VEGF-R1 or VEGF-R2 or both. The interaction between VEGF and its receptors with regard to a potential autocrine loop directly affecting tumour cell proliferation remains poorly understood and may rely on interaction with both receptors. Although this data might support an autocrine role for VEGF, potentially mediating tumour cell mitogenesis, it cannot determine which receptor, if any, is of paramount importance.
Previous studies have demonstrated that, although VEGF protein is detected by immunocytochemistry in endothelial cells, mRNA transcripts of VEGF have not been detected (14). Observed immunocytochemical staining for VEGF on endothelial cells is therefore thought to represent binding of VEGF secreted by adjacent tumour cells (14) and immunocytochemical expression of VEGF by endothelial cells is considered to be exogenous in origin. This study demonstrates a strong correlation between tumour cell immunoreactivity of VEGF with endothelial cell immunoreactivity of VEGF, VEGF-R1 and VEGF-R2 (Tab. III). Within the limitations of this immunocytochemical study, it is interesting to speculate on possible paracrine loops involving tumour-secreted VEGF and endothelial cell expression of VEGF receptors in mediating tumour angiogenesis. At sites of VEGF over-expression by tumour cells, Dvorak et al (14) have shown that both VEGF-R1 and VEGF-R2 are over-expressed in the endothelium of adjacent microvessels.
Although tumour cells represent the major source of VEGF, it must be noted that tumour-associated stroma may also be an important source of VEGF, particularly in tumours with foci of relative hypoxia (14-17). VEGF may therefore be produced in either tumour cells or stromal fibroblasts before being transported to endothelial cells where interaction with VEGF-R2 and/or VEGF-R1 leads to proliferation and angiogenesis. Although this is an attractive hypothesis, the presence of a protein is not absolute proof of an antigen's functional role in tumour growth. An in situ study by immunocytochemistry can help to focus on those possible autocrine and paracrine loops that merit validation by further functional studies (18). It must be also noted that, unlike Decaussin et al (18), we were not able to demonstrate over-expression of VEGF, VEGF-R1 and VEGF-R2 in stromal fibroblasts.
In this study, expression of VEGF-R2 decreased as tumour grade progressed from low to high grade (i.e. from well to less differentiated tumours) (Tab. IV). This is analogous to expression of oestrogen and progesterone receptors in breast cancer. Tumours of the breast that are receptor positive tend to be of lower grade than those that are receptor negative (19). Similarly, VEGF-R2 expression was inversely related to the detection of lymphovascular invasion, a pathological feature of more aggressive tumours. However, other important pathological variables such as advanced T stage and the presence of lymph node metastases were not found to influence the expression of VEGF or its receptors.
Immunocytochemical expression of VEGF has been shown to have prognostic significance for various cancers including oesophagogastric cancer (20-23). In non-small cell lung cancer, despite strong expression of VEGF, VEGF-R1 and VEGF-R2 in tumour cells, tumour endothelium and stromal fibroblasts, neither VEGF, receptor 1 or receptor 2 correlated with patient survival (18). In oesophageal cancer, no previous studies have been undertaken that look at the expression of VEGF-R1 or VEGF-R2. In gastric cancer, immunocytochemical expression of VEGF-R2 has been demonstrated in tumour endothelium and in occasional tumour cells (6). VEGF-R2 expression, however, did not correlate with VEGF expression or patient survival (6). Conversely, in a trial assessing the impact of VEGF receptor expression on outcome in 86 patients with gastric cancer, Hirashima et al (7) demonstrated that stromal vessel VEGF-R1 and VEGF-R2 expression correlated with shorter survival while VEGF-R1 expression correlated with lower response to adjuvant chemotherapy.
In this study, while neither VEGF nor VEGF receptor expression was a significant predictor of long-term outcome, the sample size was too small to reliably analyse prognostic significance. VEGF-R2 expression did correlate inversely with lymphovascular invasion, which was associated with decreased survival, and although the result was not statistically significant, patients with VEGF-2 values greater than the median survived longer. On the basis of these results, further series with an increased study population and particular reference to VEGF-R2 may detect a significant difference in survival, should one truly exist.
Significant methodological differences may account for discordant results when assessing biological markers using immunocytochemistry (variable antibodies and staining procedures), flow cytometry and molecular techniques. The utilisation of multiple analytical methods to determine the expression of VEGF and its receptors may improve the reliability of the study. However, it should be noted that the authors did not identify any difference in clinicopathological parameters or long-term outcome when VEGF was measured in both tumour epithelial cells and the circulation in the same cohort of patients using an ELISA technique (24).
In this study VEGF, VEGF-R1 and VEGF-R2 were measured at a fixed time point, the time of surgical excision. Changes in the expression of these molecules during progression of the disease or, more importantly, neoadjuvant therapy may provide a better understanding of their role and allow for clinical application, respectively. Dynamic changes in the expression of VEGF and its receptors during neoadjuvant therapy for oesophagogastric cancer could potentially highlight a subgroup of patients who would benefit from additional adjuvant therapy. This latter point may be of particular relevance if the ongoing Medical Research Council ST03 phase III trial comparing peri-operative chemotherapy, with or without bevacizumab (an anti-VEGF monoclonal antibody) (25), demonstrates a survival advantage in the bevacizumab group.
CONCLUSION
VEGF, VEGF-R1 and VEGF-R2 are over-expressed in tumour epithelial cells. In these cells VEGF expression correlates with VEGF receptor expression (VEGF-R1 and VEGF-R2) and may represent the existence of an autocrine relationship. VEGF-R2 expression is decreased in the presence of the adverse pathological variables lymphovascular invasion and higher tumour grade. This study however is too small to reliably analyse the prognostic significance of these biological markers and further larger studies should be undertaken using multiple methodological techniques assessing dynamic tumour expression during neoadjuvant treatment.
