Abstract
Purpose
The purpose of this study was to investigate the possible radioactive potential of zirconium dioxide (ZrO2) dental ceramics. Such information is necessary since they became an integral part of routine prosthetic rehabilitations and devoid of scientific information about their radioactivity creates some concern in the dental community.
Methods
Four different types of commercial ZrO2 ceramics, namely Lava, Cercon, ICE Zirkon, and Everest Bio ZS were investigated before and after the sintering process. Sintering temperatures were applied according to each manufacturer's instructions. The compositions of the presintered and sintered specimens were analyzed using x-ray fluorescence spectrometer (XRF). Concentrations of the elements and compounds were also measured by this method. Determination of radioactivity continued with gamma-spectrometry measurements and gross alpha/beta analyses.
Results
The activity of gamma and gross alpha/beta was below minimum detection limits (MDL) for presintered and sintered ZrO2. The MDLs of gross alpha/beta counting system were 0.02 Bq/g and 0.01 Bq/g for alpha and beta radioactivity, respectively. The sintering process played only a minor role in the composition of the compounds.
Conclusions
Radioactivity of the ZrO2 ceramics studied showed negligible radionuclide activity that can be considered lower than many hazardous radioactive appliances in our environment.
Introduction
Zirconium, which means golden in color, is the 18th most abounded element in the earth (1). Zirconium does not occur in nature in pure state and is refined from naturally occurring ores. It usually contains trace amounts of other elements depending upon the source of the original ore. The geologic process that forms zirconium minerals such as zirconium silicate (ZrSiO4) and zirconium dioxide (ZrO2) leads to the incorporation of some radionuclides into crystal structure (2). Zirconia is a polymorphic oxide that exists in three different crystal structures as monoclinic, tetragonal, and cubic (3-5). In particular, ZrO2 typically contains trace amounts of radionuclides such as radium, thorium, and uranium (6-8). These radionuclides generate alpha, beta and gamma radiations (9). According to safety reports about ZrO2, radioactive impurities have to be taken into account because of these radionuclides especially when they are used in industrial applications and medical areas (2).
The primary application of the ZrO2 as a biomaterial in medicine is attributed to hip replacements in the 1960s (10). The success of the biomaterial in the body depends on mechanical properties and biocompatibility. Some stabilizing oxides such as CaO, MgO, CeO2, and Y2O3 are supplemented to the structure in order to withstand the phase transformations (5,10). The positive experience and studies of ZrO2 achieved by orthopedics guides dentists for an alternative treatment strategy in odontology (1-4). ZrO2-based material in dentistry becomes a favorable technology for dentists because of their superiority in biomechanics and dentin-like structure. Nowadays, the use of ZrO2 restorations is increasing because of the developments in CAD-CAM systems(4). Partially sintered (presintered) ZrO2 ceramic blocks, that are sintered at approximately 950°C, make the material easily machined. The machined products are sintered again and excellent mechanical structure with dentin-like appearance may occur with high success (8,11). However, ZrO2 ceramics reveals a considerable higher level of radioactivity compared with other dental ceramics (9).
In the 1970s some additives were presented in dental ceramics to mimic the fluorescent characteristic of human dentin. These additives increased the radioactive potential of dental restorations because of uranium, radium, and thorium chains. The international community has taken steps to minimize the use of naturally occurring radioactive materials. According to The International Standards Organization and the American Dental Association, in conjunction with the American National Standards Institute, the porcelain formulations should not contain any radioactive substance intentionally (12). Uranium radionuclide was presented in dental ceramics until ADA/ANSI Specifications No. 69 (American National Standards Institute/American Dental Association, 1991) was accepted (13). The use of some additives, which increase radioactive potential, was subsequently restricted in dental ceramics according to ISO standards (ISO 6872 and 4824) (9). ZrO2 ceramics also led some researchers to suspect radioactivity because of the possible uranium, radium, and thorium chains (6,14). These radionuclides may be present at minimum levels or different isotopes of another element. A small dose of radiation caused by radionuclides always occurs around the environment. Moreover Vagkopoulou et al (15) have pointed out that the radioactivity of the ZrO2 ceramic used in surgical dentistry is lower than the radionuclide level of the human bone.
The human body is exposed to radiation from the Sun, ground, electronic devices, and also naturally occurring radionuclides in our body tissues (16). If the dose exceeds the limits, it becomes harmful for health and finally, mutagenic or transforming effects may occur in some tissues (7). The patient, rehabilitated with a ceramic material containing radionuclides, is prone to radioactive contamination. In fact, a dental technician may be contaminated by this ceramic in presintered form with dust exposure during the milling process. Nevertheless, there is a paucity of information related to radioactivity risk in dental applications of ZrO2 ceramics. Therefore, the aim of this study was to investigate the radioactive potential of ZrO2 both in laboratory and clinically in presintered and sintered form since it started to be widely used as a new biomaterial in dental appliances.
MATERIAL AND METHODS
Six disc samples 10 mm in diameter and 2 mm thick were prepared from each of four different commercial ZrO2 ceramics. Half of them were sintered and the remainder were kept in original form. The codes of subgroups and sinterization process are given in Table I. Samples were cleaned in an ultrasonic acetone bath for 10 minutes.
THE CODES, BRANDS, SINTERIZATION PROCESS, AND MANUFACTURE OF THE CERAMICS USED FOR THE EXPERIMENTS. SINTERING PROCESS OF L-2, C-2, I-2, AND K-2 BRANDS WERE PERFORMED ACCORDING TO MANUFACTURER RECOMMENDATIONS AT 1500°C/11 H, 1350°C/6 H, 1500°C/5 H, AND 1450°C/10 H, RESPECTIVELY
Before performing the experiments, the composition and concentration of each ZrO2 ceramics were analyzed by PANalytical AXIOS Wavelength Dispersive x-Ray Fluorescence Spectrometer (WDXRF) with IQ+ software programme using the disc samples. The power capacity of the system is 4 kW with maximum 160 mA current. In order to check the accuracy and reliability of the results, measurements were taken in three different parts of each sample and average values were calculated and presented by the IQ+ software programme.
Many radioanalytical analyses are used to determine the radionuclides contained within the environmental samples. The most commonly preferred analyses can be assumed as gamma-spectrometry, alpha-spectrometry, gross alpha/beta (α/β) analysis, and liquid scintillation counting (17). In this study, in order to determine the radioactivity levels of ZrO2 ceramics, two samples from each subgroup were taken and grounded by mill. One of them was used for gamma spectrometry measurement and the other was used for gross alpha/beta analyses:
Gamma-spectrometry measurements
Gamma spectrometry that is a highly suitable counting technique for non-destructive analysis is used to measure the gamma radioactivity. This technique is able to measure the radionuclides directly in a very short time without any radiochemical separation procedure. In a gamma spectrometer, the activity levels of different radionuclides of a sample are estimated by a semi-conductor detector, which is aided with a computer-based multichannel analyzer. A high purity Germanium (HPGe) is frequently used as a semi-conductor detector (18).
The gamma spectrometer Canberra System 100 used in this study consists of a HPGe detector. Powder samples were placed in cylindrical containers of the HPGe detector before the measurement process commenced. Each specimen was counted for 24 h according to direct measurement. The results were transferred to the multichannel analyzer and the computer determined the activity of the radionuclides.
Gross alpha/Beta analyses
The gross alpha/beta counting system is used to measure the radioactivity levels of materials occurring α or β emitting radionuclides. This method facilitates the gain in information associated with a particular action level of a sample. Each sample for gross alpha and beta radioactivities was counted in an automatic low-background system (Protean Instrument Corporation WPC 9550). The powder samples were located in a stainless-steel planchet. Initially, the radioactivity amount was classified according to alpha or beta origin. All samples were counted automatically for gross α and gross β activities in 24 h of counting time.
There are two common units for the measurement of the radioactivity of a substance: curie (Ci) and Becquerel (Bq). Curie is the rate of radioactive decay of a substance and is equal to 3.7 × 10 disintegrations per 10 seconds. Becquerel on the other hand, represents the radioactive decay of a substance as one disintegration per second and, therefore, more essential when compared to curie. The conversion between curies and becquerels is; 1 Ci = 3.7 × 10 Bq. Although curie is the current unit for radioactivity in the USA, as the metric system becomes more common in scientific areas, becquerel is expected to be a standard for this area (19).
Results
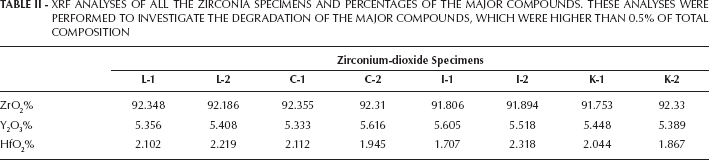
Elemental analyses assessed by XRF spectrometer are presented in Table II. The analyses revealed that the difference between the presintered and sintered forms were not significant. Minimal changes occurred in all samples at the fractional data of the total percentage. However, a differentiation in compounds was not expressed. Although XRF analyses showed compositions of the specimens with high sensitivity, an element or compound present below the 0.5% of total composition cannot be detected. In Figure 1 and 2, Everest Bio ZS samples were randomly represented as a sample graph since XRF measurement of ZrO2 samples in all groups showed similar trends before and after the sintering process.
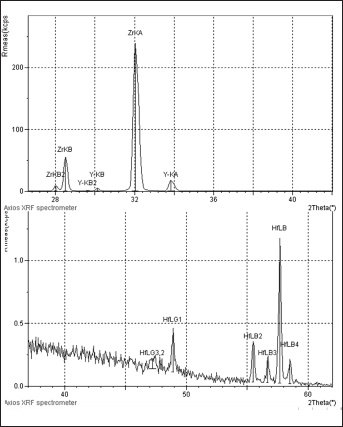
Representative x-ray fluorescence spectrometer (XRF) analysis for the Zr and Hf elements of Everest Bio ZS ZrO2 ceramic specimen in presintered form. The peaks are the characteristic x-rays of the elements in the specimens. The x-axis shows the energy levels.
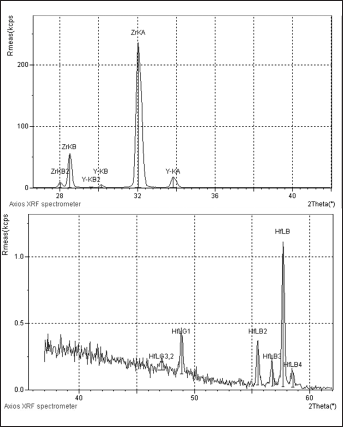
Representative x-Ray Fluorescence Spectrometer (XRF) analysis for the Zr and Hf elements of Everest Bio ZS ZrO2 ceramic specimen in sintered form. According to the graphs there is no significant difference between the two forms. In all the groups and forms no difference was detected where K-1 and K-2 specimens were randomly selected to assess their energy levels.
XRF ANALYSES OF ALL THE ZIRCONIA SPECIMENS AND PERCENTAGES OF THE MAJOR COMPOUNDS. THESE ANALYSES WERE PERFORMED TO INVESTIGATE THE DEGRADATION OF THE MAJOR COMPOUNDS, WHICH WERE HIGHER THAN 0.5% OF TOTAL COMPOSITION
For a 24 hour counting time, no measurable level of gamma emitting radionuclide activity was found. The activity of gamma and gross alpha/beta did not exceed the minimum detection limits (MDL). The MDLs of gross alpha/beta counting system were 0.02 Bq/g and 0.01 Bq/g for alpha and beta radioactivity, respectively.
Discussion
Both the medical and dental professions are concerned about the safety of dental treatment and any potential health risks that might be associated with the materials used to restore teeth (20). If a material is accepted as biocompatible, this product should not be responsible for inflammatory reactions and not provoke allergic, immune, toxic, mutagen or carcinogenic reactions (1,6). Nonetheless, Veronese et al. (13) generalized that if a patient is rehabilitated with any prosthetic restoration, the risk of radionuclide activity may be 10 times greater than other healthy members of the population. Although Akagawa et al. (21) presented ZrO2 ceramic as an inert material, a few studies assessed the ZrO2 ceramics for their radioactive potential. Some reviews about ZrO2 ceramics did not consider the risk of radioactive potential (4,10). However, Schmalz et al. (9) stated that radioactivity concentration of modern ZrO2 ceramics which may contain some radioactive contaminants was below the normal threshold level. The radioactivity level of the ZrO2 ceramics has to be evaluated according to the sintering process; the alteration in the temperature may modify the activity of radionuclides. The effect of the sinterization of ZrO2 on radioactivity levels was not assessed until this study. Although the measurable level of some radionuclides could not be determined, the activity of gamma and gross/alpha were below the MDL for presintered and sintered ZrO2 in this study. The Gross alpha/beta counting system's MDLs were 0.02 Bq/g and 0.01 Bq/g for alpha and beta radioactivity, respectively. According to manufacturers' recommendation the radioactivity of Z-Blank is measured at a value of 0.005 Bq/g, much lower than the maximum value of 1 Bq/g permitted by ISO 6872.
When the ZrO2 ceramic is used in a restoration, alpha particles may only play a minor role for oral tissues because alpha particles have a maximum range of 30 μm in tissues and they may already be absorbed by the saliva and plaque covering the restoration before reaching the radiosensitive cells in the basal layer of the oral mucosa(9). The beta and gamma particles may have a higher range of absorption in the tissues. Hence, a greater volume of the body's tissues are exposed to beta and gamma as compared to alpha particles (14). Gamma particles induce dose to all the tissues of the oral mucosa depending on the energy of the gamma ray photons (22). On the other hand, these particles are at an extremely low level for contaminations and very hard to measure. Covacci et al. (7) investigated 10T1/2 cells because of the possible mutagenic and transforming effects of purified and unpurified ZrO2 ceramics. Cells were used between the 11th and 18th passage and subcultivation of stock cultures was performed before they reached confluence. Samples did not show mutagenic and transforming effects on 10T1/2 cells.
Dental materials are grounded and polished in the patients' mouth, sometimes with unintentional damage to the oral mucosa; thus resulting in more exposure to the dental material used in the restorations. Furthermore, the possibility of the patient swallowing radioactive particles because of teeth grinding or dental treatment is important and should be tested. Possible inhalation of radioactive compounds by patients, technicians, and dentists has to be taken into account when examining the biocompatibility of these dental materials in future studies.
Willman (23) pointed out that the manufacturing process of ZrO2 ceramics is an important step for human health because of possible radioactive impurities not effectively eliminated from the compound. This study assessed the specimens not only at the end of the process but also prior to sintering. Developments in recent years prove that ZrO2 ceramics are suitable for CAD/CAM processes in all three conditions; unsintered, partly sintered (presintered), and sintered stages. The presintered ZrO2 blanks have a chalk consistency and they are easily workable by the dental laboratories with any kind of CAD/CAM devices to produce crowns and FDPs. Because of the easier workability and faster machining, the use of presintered blanks is predominant today. After a final sintering process, some alterations in density, grain size, amount of cubic phase in the ZrO2 body occur but at the same time ultimate strength is achieved (24). This alteration to the sintering process is not only a physical but a chemical reaction. Some ions in ZrO2 ceramic appear to concentrate within the large grains after sintering (25). If only sintered or presintered specimens were analyzed in this study, we would have agreed that they were the same materials before the experiments. The XRF results suggested that chemical composition of the samples did not vary by sintering process. Ratios of the compounds of specimens in presintered and sintered forms did not show significant alteration according to similar studies about the chemical composition of the ZrO2 ceramics. Vagkopoulou et al. (26) designated the amount of ZrO2+ Y2O3+ HfO2 more than 99% of the material. The distribution of the compounds in the ZrO2 ceramics was found above 90% and Y2O3 encompassed 5% according to the findings of Papanagiotou et al. (27). Deny and Kelly (28) indicated the amount of HfO2 as approximately 2% in zirconia samples. In light of these studies, chemical compounds of both presintered and sintered ZrO2 ceramic samples were detected within the normal limits of the compositions in this study.
To sum up, both presintered and sintered samples presented radioactivity below the threshold levels in this study. The activity of gamma and Gross alpha/beta was also found to be below the MDL for the ZrO2 ceramics studies. Radioactivity levels and XRF measurements did not reveal any difference between presintered and sintered ZrO2 ceramics.
