Abstract
Background:
Nebulized hypertonic saline (HS 7%) is commonly prescribed for cystic fibrosis (CF) patients using nebulizers and handheld devices that generate oscillatory positive end-expiratory pressure (OPEP) . There is limited evidence for the delivery of nebulized HS 7% and whether OPEP use impacts efficiency. This study examined particle size and delivered lung dose in a spontaneously breathing model between different nebulizers with and without OPEP therapy. We hypothesized that particle size and delivered lung dose would be similar among testing scenarios.
Methods:
The Pari LC Plus and AeroEclipse II provide continuous aerosol output. AeroEclipse II can also be configured as a breath-actuated nebulizer (BAN). We modeled 5 testing conditions: Pari LC Plus (continuous); AeroEclipse II (continuous and BAN), and both nebulizers (continuous) integrated with an Aerobika OPEP device. Particle droplet size (µm, Dv 50) was measured by a laser diffractometer. Nebulized HS 7% was applied to a 3D anatomic nasotracheal airway of a spontaneously breathing child lung model (frequency 15 breaths/min, VT of 350 mL, and I:E of 1:2). The nebulizers, OPEP devices (when applicable) and a capture filter were pre-conditioned in a vacuum heater and weighed (µg). The nominal HS 7% dose was quantified based on the dry salt mass. The filter was placed between the airway and lung model. The HS 7% was nebulized via a mouthpiece, until sputtering, for 3 runs for each condition (no. = 15). Following testing, each component was dried 70°C for 24 hours and reweighed. The difference in pre-post filter and nebulizer weights represented delivered lung dose and residual nebulizer dose, respectively. Values were referenced to the nominal nebulizer dose (%). The mean lung doses were compared using ANOVA.
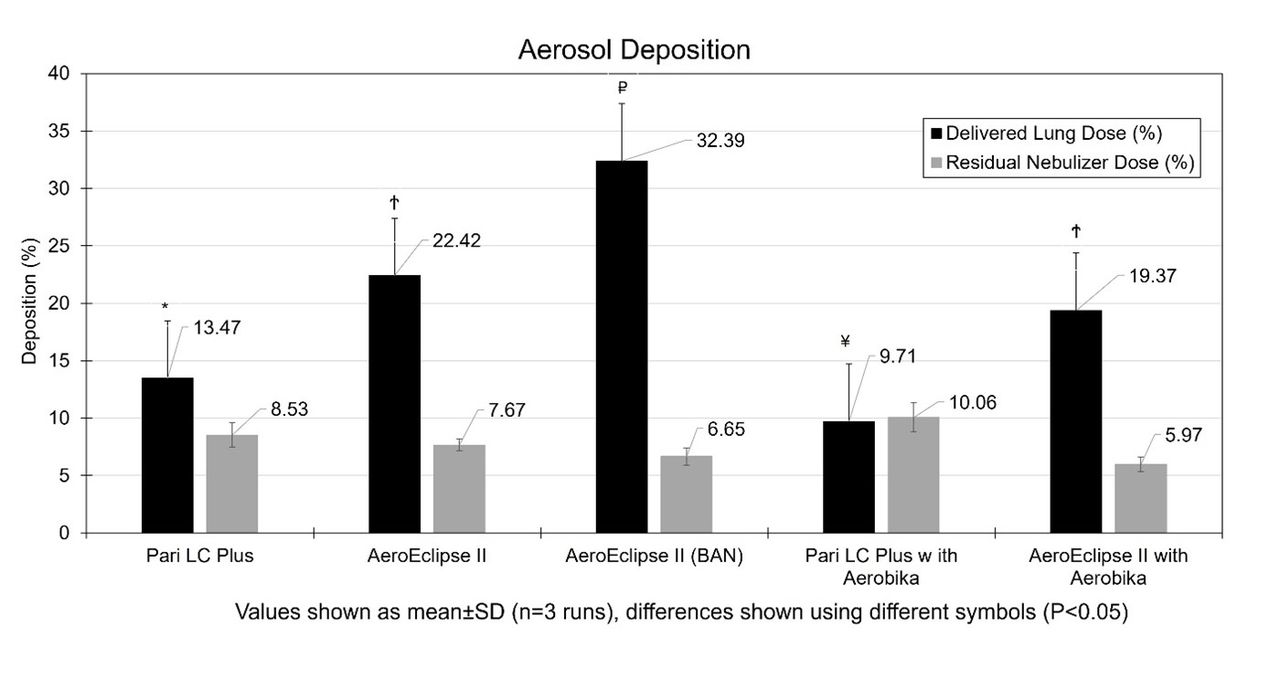
Results:
The Dv 50 was 2.2 ± 0.2, 3.2 ± 0.1, 1.94 ± 0.1, and 2.6 ± 0.1 µm for Pari LC Plus, AeroEclipse II, Pari LC Plus/Aerobika and AeroEclipse II/Aerobika, respectively. The AeroEclipse BAN-mode had the greatest lung dose (%) than the other condition (P < .05, see Figure). AeroEclipse II compared to Pari LC plus had greater lung dose with the Aerobika (P < .05). The nebulizer residual was lower with AeroEclipse II than Pari LC plus for all conditions.
Conclusions:
We showed small, inhaled HS 7% particles (<5.4µm) are generated with each nebulizer. The AeroEclipse II performed with the highest efficiency and provided similar lung deposition of HS 7% with and without OPEP. Patients may benefit from greater drug delivery using the AeroEclipse II with BAN mode.
Get full access to this article
View all access options for this article.
