Abstract
Background:
Medical device-related pressure injuries (MDRPIs) result from the use of devices designed and applied for diagnostic or therapeutic purposes and the resultant pressure injury generally conforms to the pattern or shape of the device. MDRPIs account for more than 30% of all hospital-acquired pressure injuries. Assessing rates within individual institutions and developing targeted, evidence-based interventions to reduce their occurrence, is necessary to reduce and maintain lower rates. Our institution is a large academic medical center located in the Northeast. In 2018, our Hospital-Acquired Condition (HAC) rounds and quarterly hospital pressure injury prevalence data of MDRPIs revealed that endotracheal tubes and their securement devices represented the highest percentage of MDRPIs in our institution. Inconsistent practices and documentation, as well as a lack of staff knowledge across nursing and respiratory disciplines was also noted during HAC rounds. Historically at our institution, endotracheal tube management (eg, assessment, documentation, management of the equipment, skin assessment) has been performed by both nurses and respiratory therapists (RTs), although their workflow was separate. Our wound ostomy and continence (WOC) nursing team and lead RTs saw an opportunity to collaborate to reduce MDRPIs associated with ETTs and their securement devices.
Methods:
Analyzed electronic survey and skin prevalence data to identify gaps in knowledge, practice and documentation. Developed and delivered education to 80% of ICU RN/RT staff. Re-educated on ETT securement device products with company reps. Created speedy rounds concept: a routine RT/RN collaborative quick look assessment of respiratory devices performed every shift in the ICU.
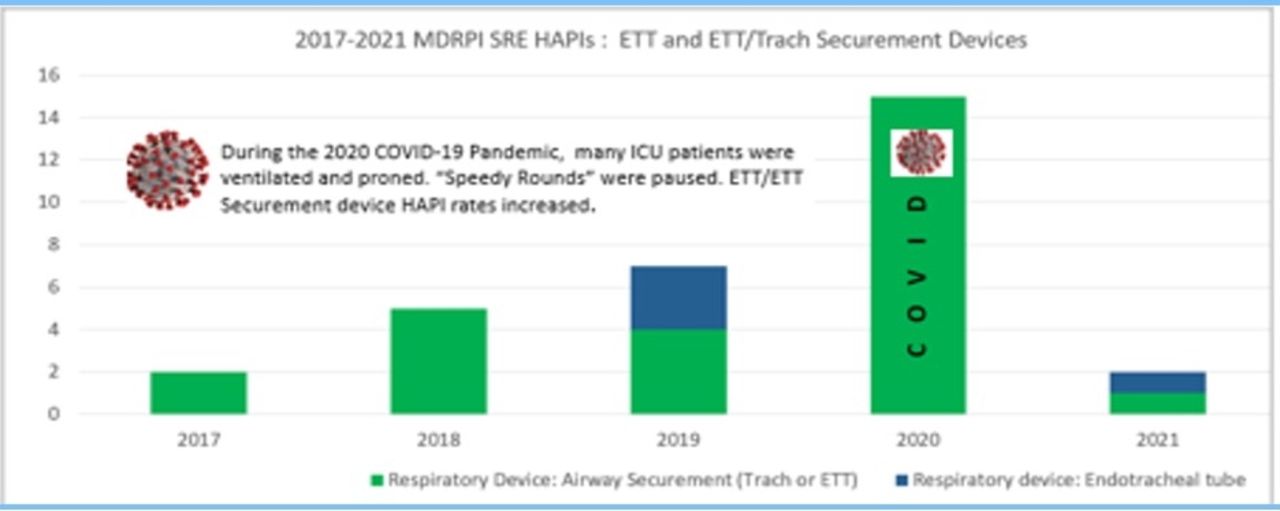
Results:
Speedy rounds increased RN/RT engagement in reducing MDRPIs associated with ETTs and ETT securement devices. Subsequent quarterly skin prevalence data and serious reportable events data have demonstrated a sustained reduction in MDRPI rates related to ETTs and their securement devices.
Conclusions:
Improved interdisciplinary collaboration has demonstrated decreased MDRPIs. Future work will continue, knowledge and documentation practices will be reassessed, and the concept/process will be translated to other medical devices in an effort to decrease MDRPIs.
Get full access to this article
View all access options for this article.
