Abstract
Background:
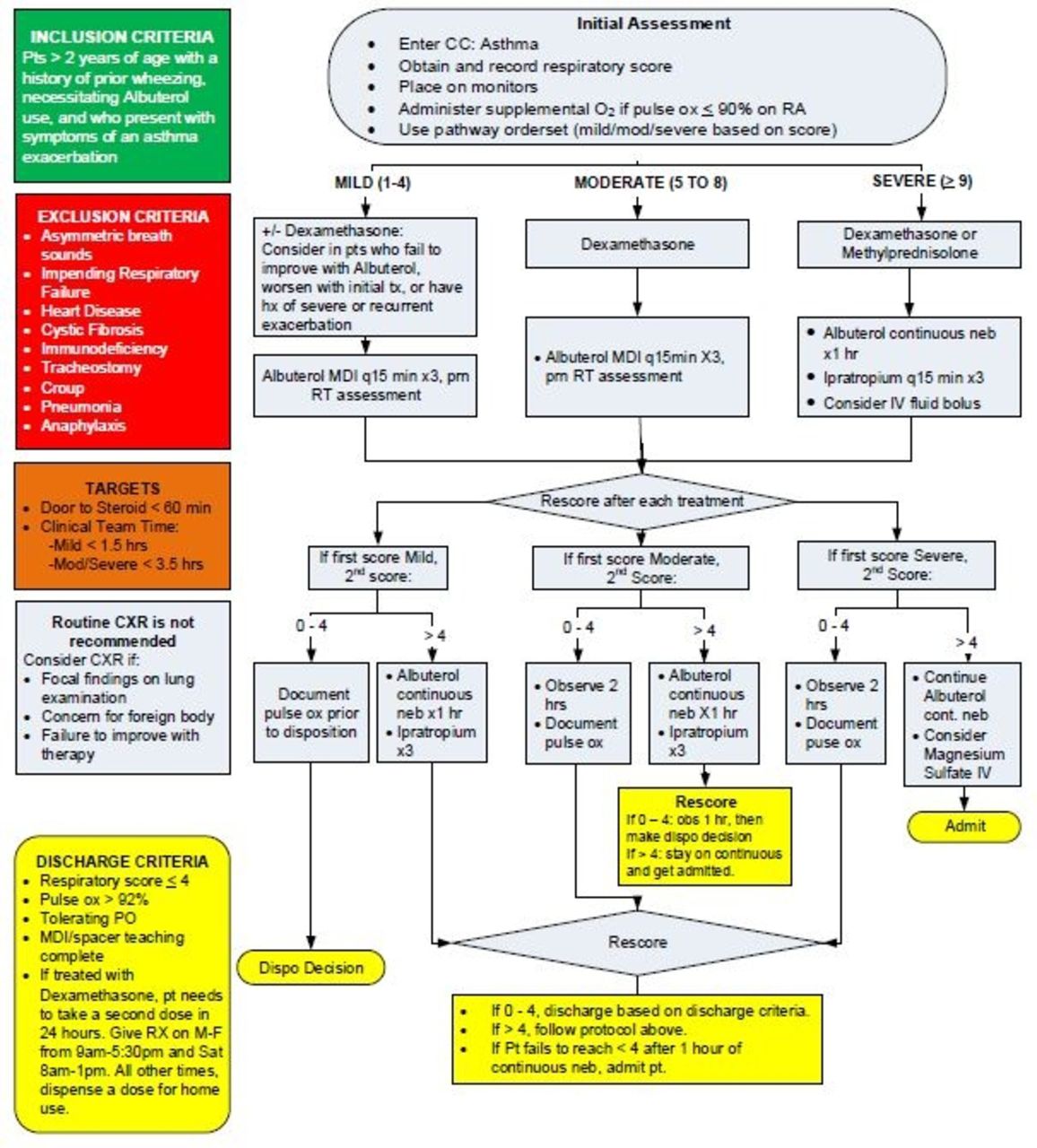
In our pediatric emergency department (ED), severe asthma (SA) is one of the leading diagnoses associated with hospital admission. To provide optimum care and decrease admission rate (AR) for SA, we have initiated protocols to ensure patients receive immediate care, even before physician evaluation. For patients with SA, continuous albuterol is the initial treatment utilized within our protocols. We aimed to compare the effectiveness of 3 back-to-back treatments when delivered via Aerogen Ultra for SA patients in place of our current protocol in our pediatric ED on AR (Figure 1).
Methods:
We completed an IRB-exempt QI project in our ED that included patients with a history of asthma and/or wheezing with prior albuterol use, > 2 years of age, on the Asthma Pathway, and a Respiratory Score (RS) ≥ 9. 47 patients received 3 back-to-back albuterol/ipratropium treatments via Aerogen Ultra with standard dosing based on weight in place of continuous albuterol. RTs documented RS pre/post each treatment and nebulizer interface (mask or mouthpiece). A retrospective chart review was completed from 2/15/2019-4/15/2019 compared to the study period (2/15/2022-4/15/2022) to determine impact of the trial on AR. Additional variables included escalation to continuous albuterol and PICU AR.
Results:
SA AR for 2019 was 0.71 and 0.73 for 2022, which was not statistically significant (P = .78) (Figure 2). However, our PICU AR for SA decreased significantly from 0.19 in 2019 to 0.04 in 2022 (P = .01). 57.4% of SA still required continuous albuterol post receiving Aerogen Ultra treatments during the trial.
Conclusions:
During this Aerogen Ultra trial, we experienced no change in AR, however, our PICU AR for SA decreased significantly. Completing this trial increased RT workload as 57.4% of patients still required continuous albuterol after Aerogen Ultra treatments. Further studies should be completed to evaluate RT workload units associated with utilizing the Aerogen Ultra and its impact on a wider SA population.
Figure 2 shows results from the before and during the trial period. Figure 1 shoes the Asthma Pathway in our pediatric ED prior to this study implementation.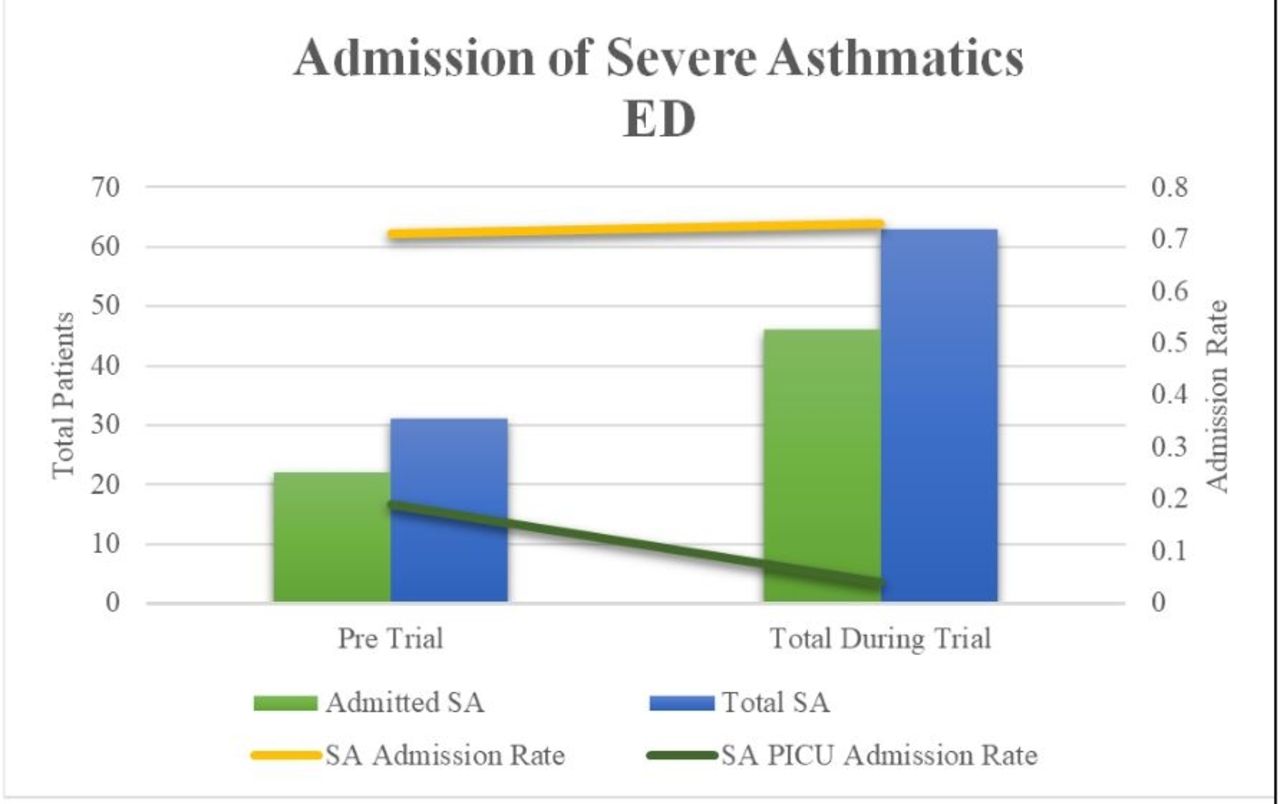
Get full access to this article
View all access options for this article.
