Abstract
Background:
It is estimated that 16.5 million patients worldwide per year undergo mechanical ventilation (MV) as a life-saving intervention in the ICU [1]. Nevertheless, MV leads to complications such as ventilator-induced diaphragm dysfunction (VIDD) [2-3]. Techniques preventing or attenuating VIDD through diaphragm stimulation and activation should be further investigated. We hypothesized that time from start of stimulation to onset of diaphragmatic contraction (latency) is independent from stimulation intensity using bilateral noninvasive phrenic nerve electromagnetic stimulation. Primary endpoint was time from start of electromagnetic stimulation to effective stimulated inspiration (defined as time point of negative peak pressure) in anesthetized patient.
Methods:
To gather the data necessary, we performed bilateral noninvasive electromagnetic phrenic nerve stimulation on 5 ASA I/II obese patients scheduled for elective surgery with general anesthesia and RSI induction. The pulsed electromagnetic field (inter-pulse frequency 25 Hz) was applied at different intensities (30%, 40%, 50%) with 2 seconds duration without ramp. Ventilator setting was CPAP with PEEP according to transpulmonary pressure measurement during the stimulation period.
Results:
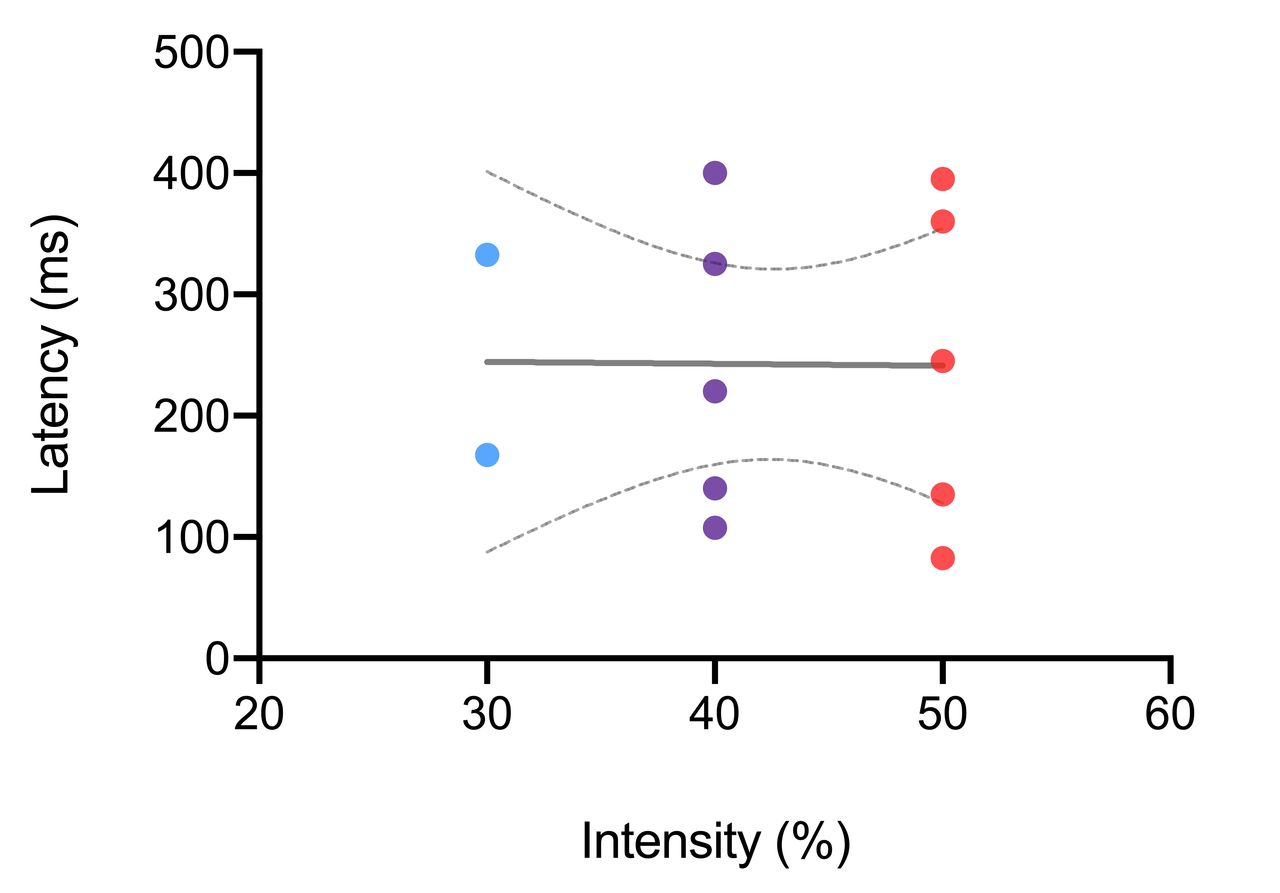
Based on all patients the mean time from stimulation to inspiration (negative peak pressure) was 250.0 ± 116.7 ms, 238.5 ± 123.2 ms and 243.5 ± 136.2 ms at 30%, 40% and 50% intensity, respectively (Figure 1). There was no linear relationship between stimulation intensity and contraction latency; the diaphragm contraction lag was independent of the stimulation intensity (P = 0.9745, r2 = 0.0001075; Figure 2). The overall contraction lag (all intensities combined) was 242.5 ± 116.3 ms.
Conclusions:
Time to inspiration after bilateral noninvasive stimulation of the phrenic nerve was independent of the used stimulation intensities (30%, 40%, 50%). Stimulation intensity does not seem to need to be adjusted to ensure adequate conduction time but can be adjusted according to other necessary parameters (e.g., diaphragm contraction and tidal volume). </br> References 1. Adhikari et al. Critical care and the global burden of critical illness in adults. Lancet 2010. 2. Goligher et al. Mechanical ventilation–induced diaphragm atrophy strongly impacts clinical outcomes. Am J Respir Crit Care Med 2018. 3. Dres et al. Critical illness-associated diaphragm weakness. Intensive Care Med 2017.
Figure 1. Latency between start of stimulation and start of diaphragm contraction (negative peak pressure) Figure 2. Linear regression of latency for 30%, 40% and 50% intensity. 95% CI is depicted.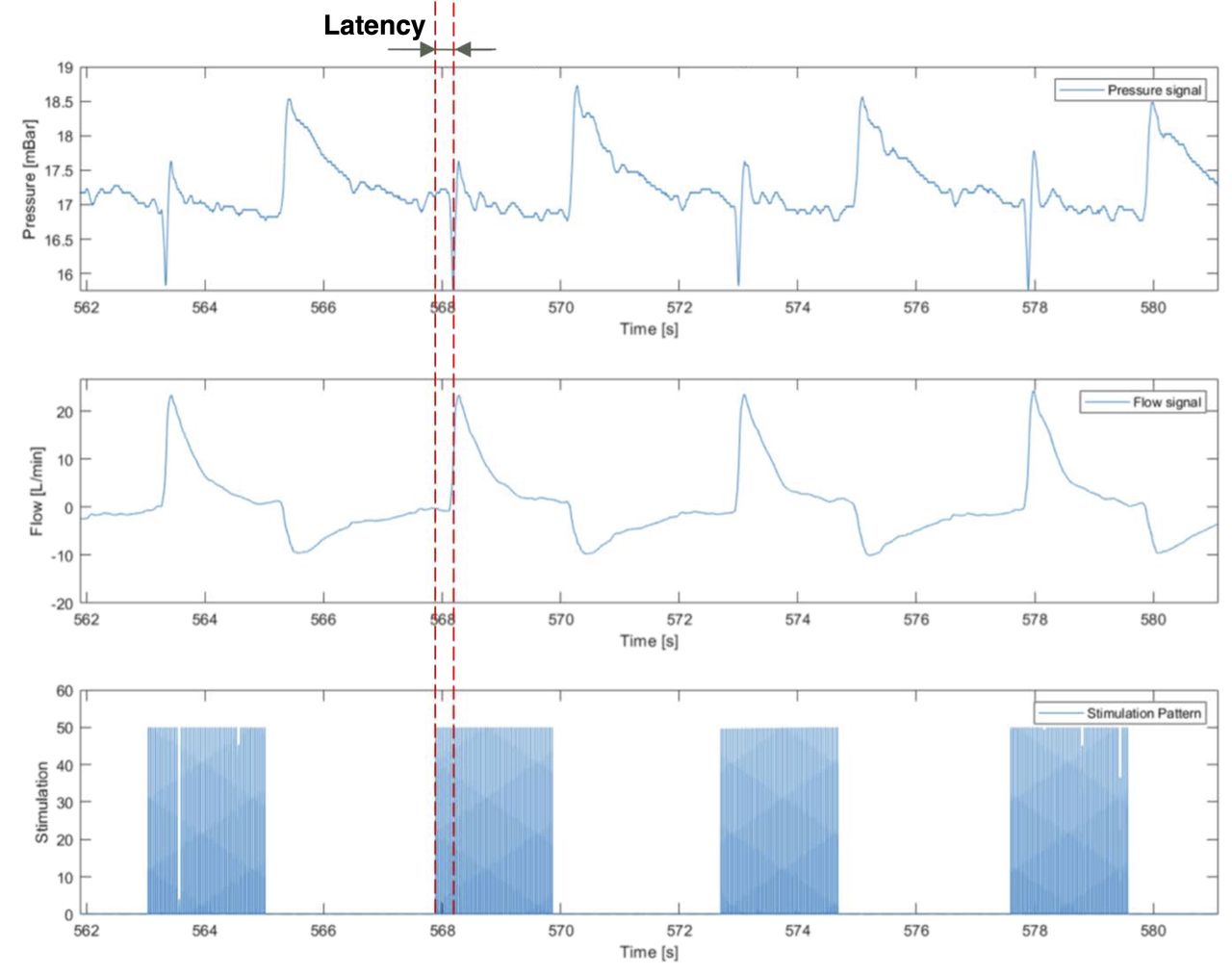
Get full access to this article
View all access options for this article.
