Abstract
Background:
In vitro assessment of trans-nasal aerosol delivery via HFNC has been conducted with anatomical models and intubation manikins that are facsimiles and may not be anatomically correct. As a precursor to planned bench studies, we have created and validated an open-source anatomical nasal model that can be 3D-printed using readily available materials and printers.
Methods:
A nasal airway cast (solid) model, derived from a CT scan of the skull, was downloaded from NIH 3D Print Exchange as a STL (Standard Tessellation Language) computer file, widely used in 3D-printing. In Computer Aided Design (CAD) software (TinkerCAD, Autodesk), the solid nasal cast was converted to a negative space surrounded by a solid head and face structure. The nares of the nasal airway were aligned with the nares of the face/head and its hypopharynx was aligned to create an opening on the inferior posterior surface of the head. The new CAD was converted to an STL file, prepared for 3D-printing using slicer software, and printed with ultraviolet-curable polyurethane resin at a resolution of 0.05 mm. A 3D-printed 22-mm diameter tubing adaptor was glued over the orifice at the hypopharynx to allow a respiration simulator and aerosol collection filter for inhaled mass (IM) determination to be attached. The model was initially tested to ascertain trans-nasal aerosol transit by fitting a medium Fisher & Paykel Optiflow HFNC to it (Fig. 1) and administering a 3 mL bolus of radiolabeled saline via a nebulizer attached to the dry side of the heated humidifier at a HFNC gas flow of 30 L/min. The IM filter was placed after the hypopharynx and attached to a sinusoidal piston pump set to simulate a distressed breathing pattern (30 breaths/min, tidal volume 750 mL, I:E 1:1). After nebulization was complete, all components including the head model were imaged by gamma scintigraphy to assess deposition of radioactive aerosol throughout the system and determine the mass balance.
Results:
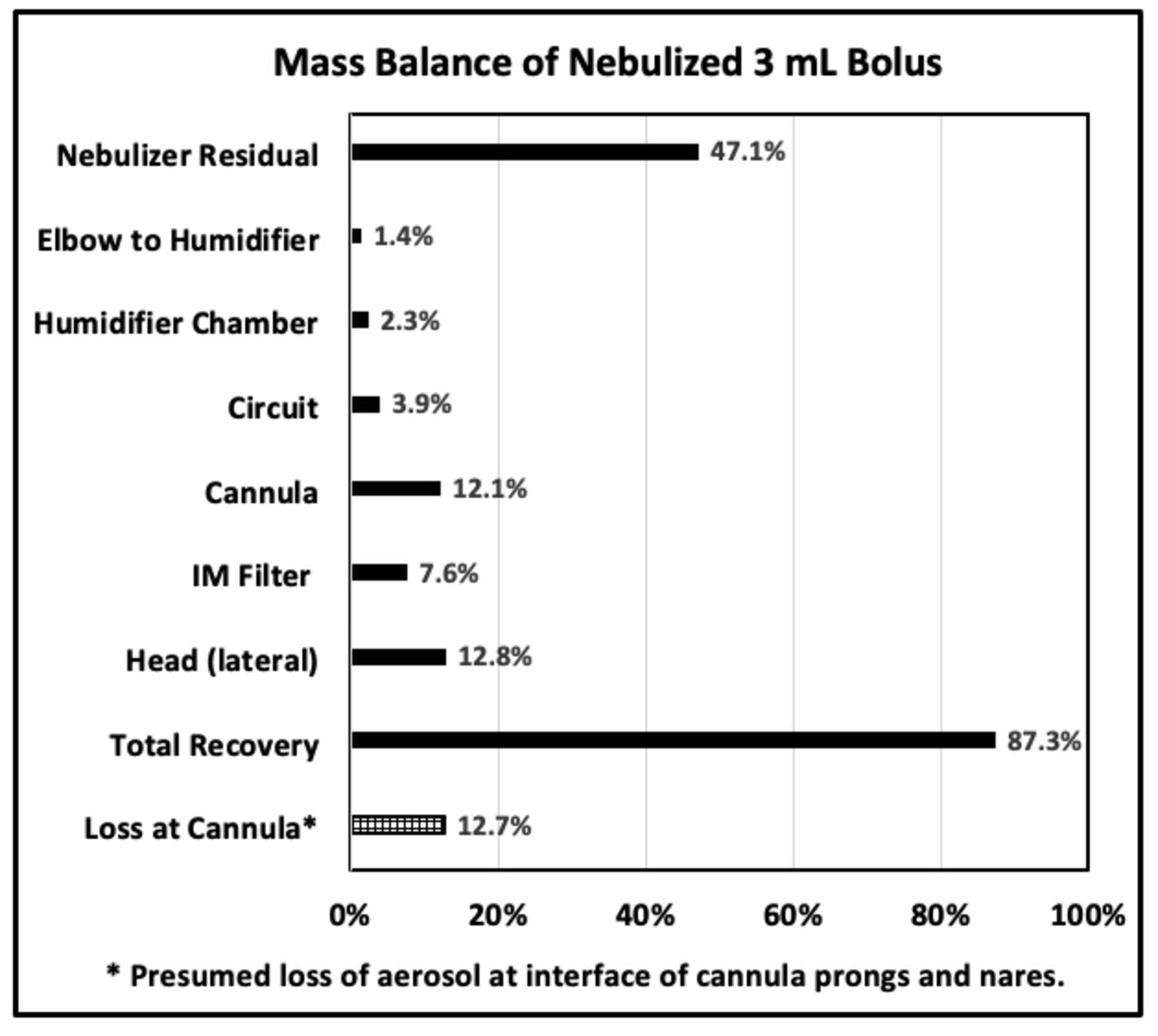
Figure 2 shows mass balance and distribution of radioaerosol deposition including 7.6% inhaled mass plus system losses of aerosol and loss (12.7%), by deduction (crosshatch), at the interface of cannula prongs and nares.
Conclusions:
The initial experiment on the new open-source anatomically correct nasal airway model shows that aerosol delivered via HFNC can transit the model from the nares to the hypopharynx and that the model will be acceptable for conducting more comprehensive studies of HFNC aerosol delivery.
Fig. 1. 3D-printed head with complete nasal airway. Fig. 2. Mass balance.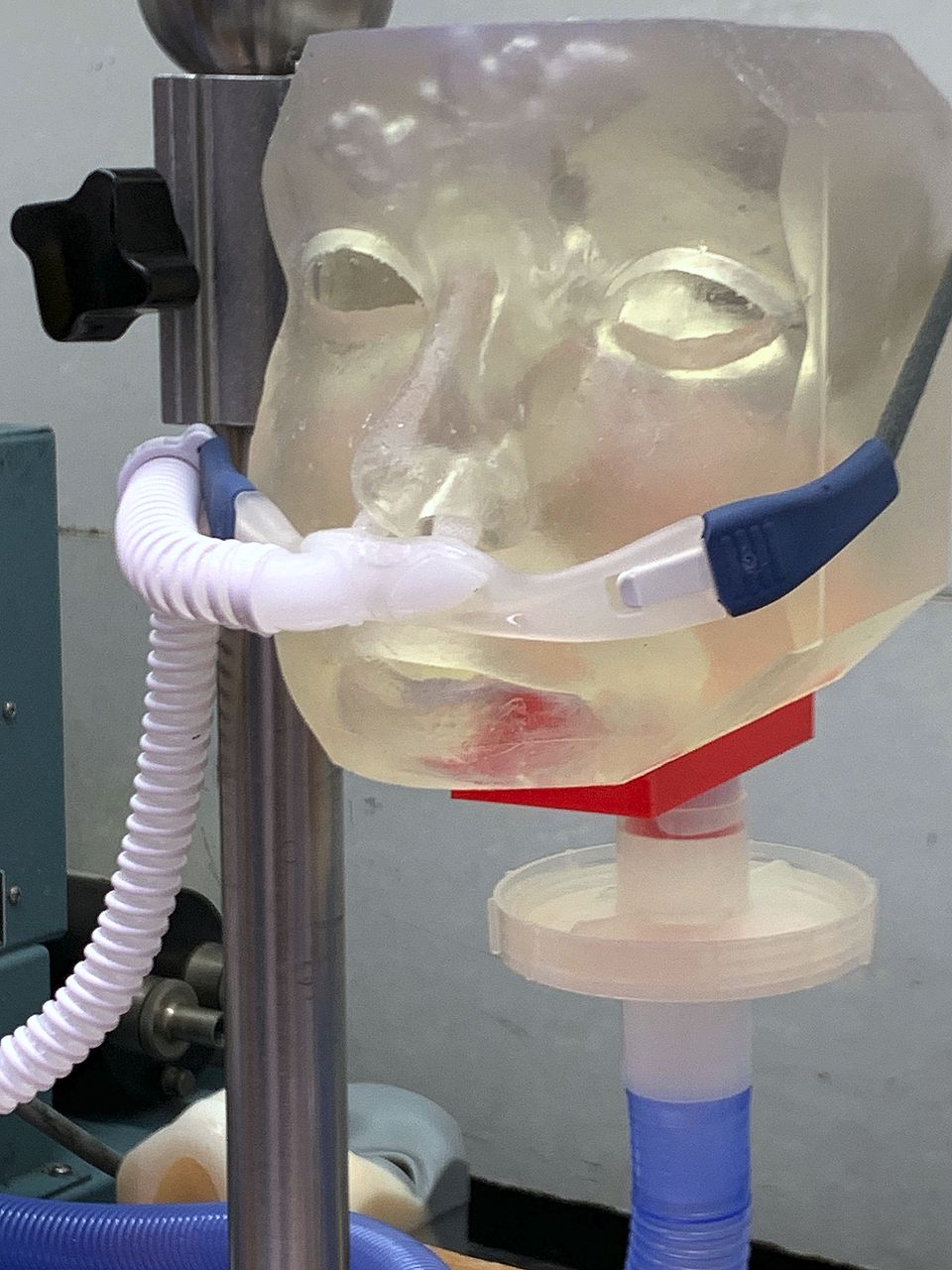
Get full access to this article
View all access options for this article.
