Abstract
Background:
Pressure injuries (PI) occur often in healthcare and cost billions of dollars annually in the U.S. Device-related PIs (DRPI) occur in up to 41% of hospital admissions. Up to 35% of DRPIs are caused by respiratory equipment. In our pediatric institution, we saw numerous respiratory related DRPIs that were not reported but required intervention. We aimed to increase early reporting of respiratory DRPIs.
Methods:
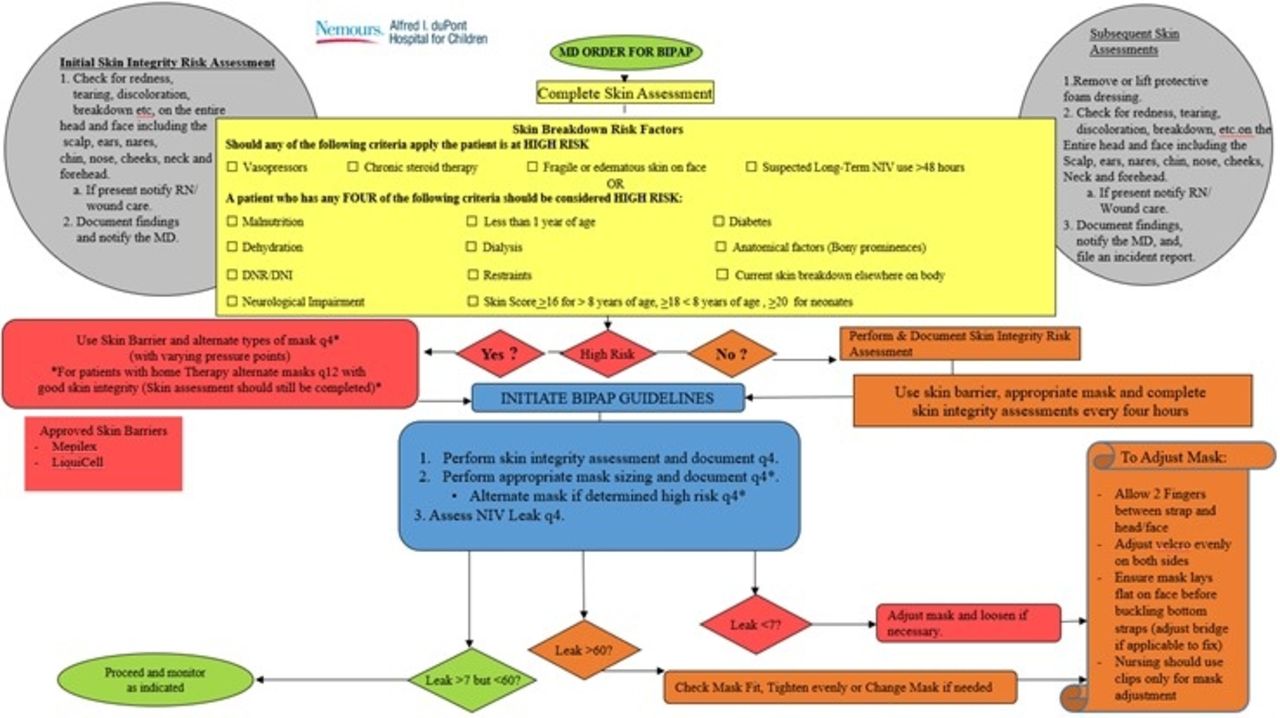
A Pressure Injury Prevention Task Force (PIPTF) was formed in Q3 of 2019. The PIPTF included multi-disciplinary team members including RNs, RTs, MDs, and wound care. The task force utilized existing tools to provide re-education related to pressure injury reporting, prevention, and notification. These tools included our NIV Skin Assessment Algorithm (Figure 1) and electronic health record notification for wound care referral and documentation. The PIPTF also reviewed pressure injuries as they were reported in real-time in order to offer expert insight and evaluate our processes from a continuous improvement perspective. In an IRB approved, retrospective analysis, from 1/1/2018 to 4/30/2021, we reviewed the incidence and prevalence of respiratory related DRPIs, and initial PI stage to evaluate the effectiveness of our processes and re-education efforts.
Results:
In 2018, we had 1 reported respiratory-related DRPI that was stage 2. In 2019, there were 13 reported respiratory-related DRPIs, 61.5% of these reported injuries occurred in Q3 and Q4 of 2019 which coincides with the PIPTF re-education efforts. In 2019, the number of reported respiratory-related DRPIs in 2020 increased to 26 (66% increase when compared to 2019). In 2021 (1/1-4/30), there has been 1 reported respiratory-related DRPI. A reduction in unstageable pressure injuries was also noted when comparing 2019 to 2020/2021. Contributing device and PI stage information for reported events is shown in Table 1.
Conclusions:
A multidisciplinary PIPTF can influence pressure injury prevention. While an increase in reportable DRPIs is not ideal on the surface, many of these injuries would have been unreported previously. The reduction in unstageable DRPIs could align with earlier detection of PIs and will be explored. Further studies must be done to evaluate the effectiveness of this model in other patient populations.
Table 1 shows the breakdown of pressure injury by year, type, and contributing respiratory related equipment.
Figure 1 shows the skin algortithm used for patients with non-invasive orders in our facility. View all access options for this article.Contributing Respiratory Device and Pressure Injury Stage of Reported Injuries
Year
Stage I
Stage II
Stage III
Unstagable
Mucosal
Skin Injury
Deep Tissue Pressure Injury
2018 (2)
2 Non-Invasive Ventilation
2019 (12)
3 Non-Invasive Ventilation
2 Non-Invasive Ventilation2 Tracheostomy1 Endotracheal Tube
3 Non-Invasive Ventilation
1 Tracheostomy
2026 (26)
1 Non-Invasive Ventilation
6 Non-Invasive Ventilation6 Tracheostomy1 Endotracheal Tube
1 Tracheostomy Tube1 Endotracheal Tube
2 Nasal Cannulas3 Endotracheal Tubes
1 Nasal Cannula1 Ventilator
2 Non-Invasive Ventilation2 Tracheostomy
2021 (1)
1 Endotracheal Tube
Get full access to this article
