Abstract
Background:
At our institution, we have received multiple reports of failure of a vibrating mesh nebulizer during inhaled epoprostenol delivery. There have also been recent published and anecdotal reports of nebulization stoppages using this nebulizer (Aerogen Solo, Aerogen, Galway Ireland). We conducted this experiment to determine if we could repeat the intermittent failures previously reported.
Methods:
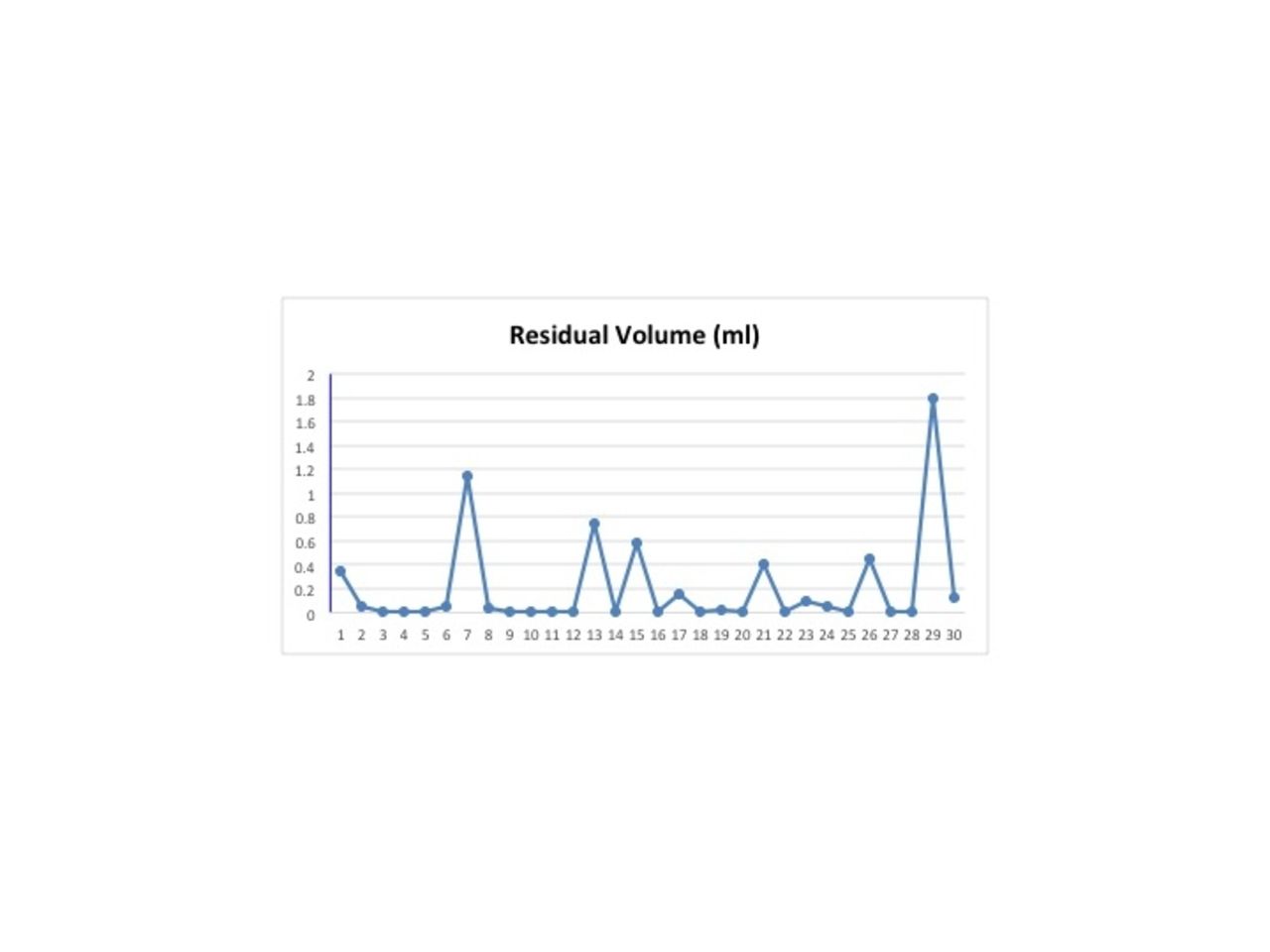
In the first phase of the study, 10 different nebulizers of the same lot were run 3 times each alternating with 2 new, previously unused electronic controllers. A total of 30 runs were completed while varying the order in which the nebulizers were tested. Each nebulizer was filled with 3 mL of normal saline and run for a total of 10 minutes. The time was recorded when the nebulizer was visualized to stop producing an aerosol. At the end of the 10-minute run time, residual volume was measured using a slip tip syringe. Failure was defined as a residual volume greater than 10% of the filling volume or 0.3 mL. The manufacturer reports the residual volume should be <0.1 mL for a 3 mL dose. The second phase of the study used the same controllers and nebulizers to mimic continuous nebulization. A syringe pump was used to infuse normal saline into the nebulizer at two rates: 4.2 and 7.0 mL/h. Each nebulizer had a total of 4 runs receiving both infusion rates with a different controller. The syringe pump was turned off at 1 hour and residual volume was again measured.
Results:
In the first phase of the study there were 7/30 runs which had a residual volume greater than 0.3ml or a failure rate of 23.3%. During the second phase of the study, one of the 40 continuous runs failed with a total residual volume of 5.75 mL. As expected intermittent nebulization was noted throughout phase 2 due to the nebulizer output of 0.3 mL/min exceeding the syringe pump flow of 0.07 and 0.12 mL/min respectively. There appeared to be no correlation between the controller used and the nebulizer failures.
Conclusions:
In phase one of this trial the Aerogen Solo nebulizer failed at a rate of 23.3% using a failure threshold 3 times greater than the manufacturer’s specifications. Further study is needed to determine the impact of this finding on therapeutic outcomes during medication nebulization. Disclosures: None
Get full access to this article
View all access options for this article.
