Abstract
Background:
Preterm infants are at risk to develop respiratory distress syndrome (RDS) due to insufficient production of surfactant. These preterm infants may require surfactant replacement therapy (SRT). Current modes to deliver SRT include the intubation, surfactant, rapid extubation technique (InSurE) and the less invasive surfactant administration (LISA) technique. LISA requires a small catheter for tracheal catheterization to deliver SRT while the patient remains on continuous positive airway pressure (CPAP) via a cannula, avoiding the need of positive pressure. We hypothesized no significant difference in number of surfactant doses would be required for LISA and InSurE techniques.
Methods:
A retrospective cohort analysis of aggregate patient data was conducted in a level III NICU, with IRB approval, from January 2018 to December 2018. Patient population included preterm infants at risk for RDS, <32 weeks, <1,300 g, any patient with respiratory failure requiring intubation and mechanical ventilation, or infants with meconium aspiration syndrome (MAS). Demographic information included: respiratory condition, method of surfactant delivery, doses required, respiratory support, gestational age (GA), and gender. A t-test was conducted using statistical software, SPSS 24 (IBM, Armonk, New York) with an alpha of .05 considered statistically significant.
Results:
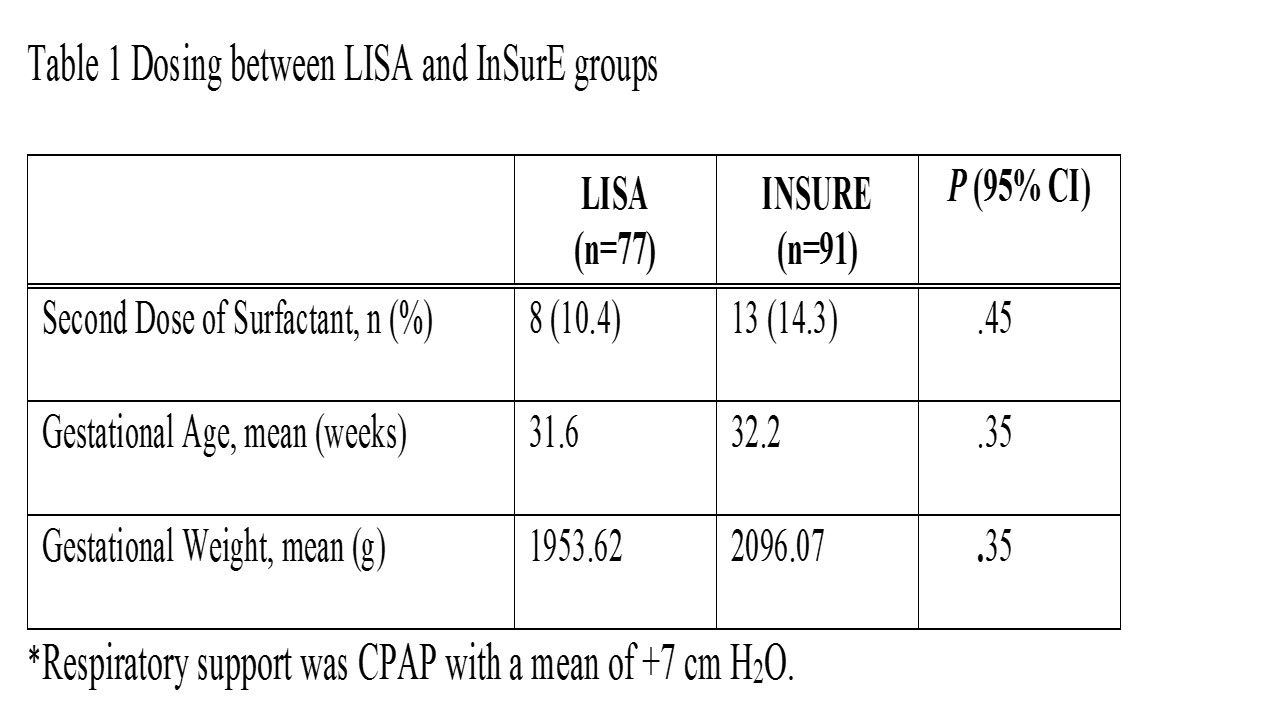
Dosing was tracked as initial and second dose. Number of doses for LISA mean (SD) 1.0 (0.30) and InSurE mean (SD) 1.4 (0.35) were not statistically different (P = .45). Requirement of second dosing was similar for LISA and InSurE, P = .45 (Table 1).
Conclusions:
Number of surfactant doses was similar for LISA and InSurE administration methods. Adverse events and long-term effects related to LISA and InSurE surfactant administration, such as the impact of positive pressure, should be studied.
Get full access to this article
View all access options for this article.
