Abstract
Background:
The American Thoracic Society and European Respiratory Society 2005 guidelines recommend weekly spirometry linearity checks for flow sensing devices and weekly syringe DLCO checks using a calibrated 3-L syringe. The expected failure rate for these checks is unknown. One abstract from a multi-center study suggests < 10% failure rate. This study identified the current frequency and source of failures with syringe linearity and syringe DLCO checks. Quality control (QC) failure reasons were also categorized.
Methods:
A mechanical QC program for an inhaled medication study included biweekly syringe linearity and syringe DLCO checks to ensure reliable equipment function for subject testing. Results were sent to a central repository for expert review. Onsite training was provided to 114 pulmonary function labs (PFLs) from North America, Europe, and Israel. Over 42 months ending in 2018, 108 PFLs continued with the study. The syringe linearity checks used peak flows < 2 L/s, 3 to 7 L/s, and > 8 L/s. Acceptable linearity data were within the expected flow range and had FVC variance ≤ .09 L. Syringe DLCO checks were accepted when 2 trials fell within pre-established ranges for DLCO, IVC and VA. Percentage failure rates for onsite training checks and subsequent study period were calculated. The reasons for failure were categorized.
Results:
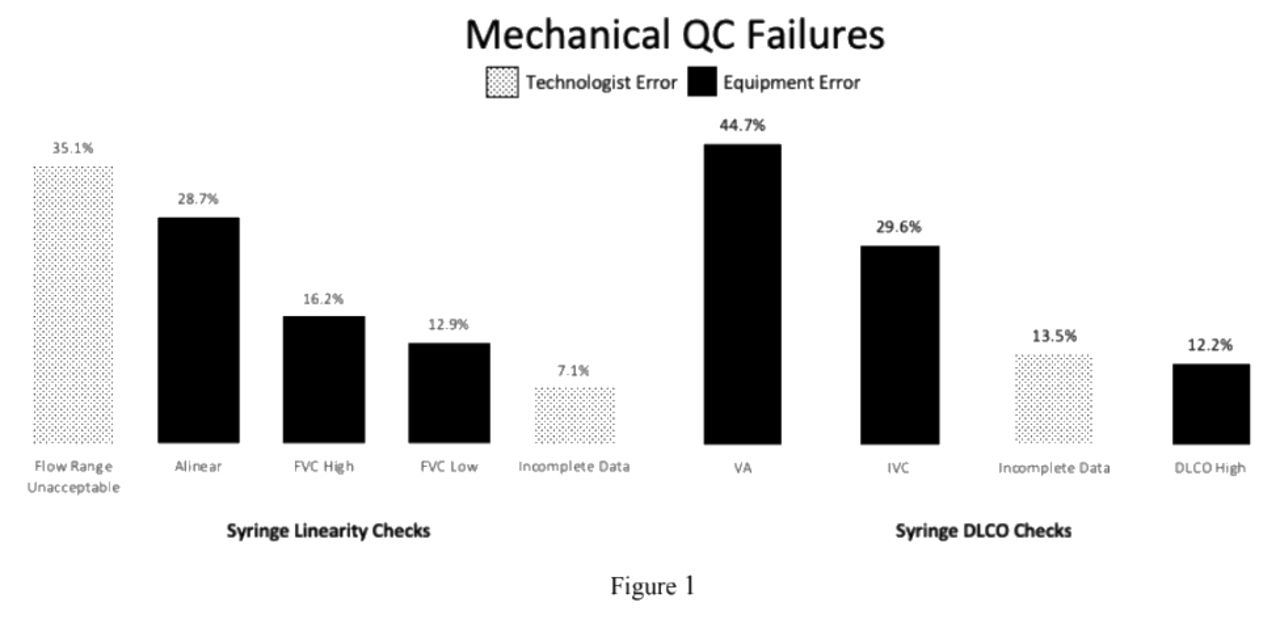
Of 114 PFLs, onsite training syringe linearity failure occurred in 2 (1.8%) sites, syringe DLCO check failures occurred at 6 (5.3%) sites. Syringe linearity failures during training were related to inadequate 3-L calibration syringe equipment. Of the 6 syringe DLCO failures, 3 were due to inadequate calibration equipment and 3 required service calls for hardware repairs. During the study period, 108 sites completed 5083 syringe linearity and 4992 syringe DLCO checks. Of the 5083 syringe linearity checks, 496 (9.76%) failed and of the 4992 syringe DLCO checks 311 (6.23%) failed. Reasons for QC failure are shown in Figure 1.
Conclusions:
The failure rates for both syringe linearity and syringe DLCO checks were < 10%. Over 40% of syringe linearity checks failed due to technologist errors. In contrast, equipment errors accounted for 86% of syringe DLCO failures. The QC failure reasons define and target areas for corrective action and education for PFL staff. Assuring that equipment is within established QC limits is essential for accurate subject test results.
Get full access to this article
View all access options for this article.
