Abstract
Background:
The standard for administering aerosolized medication (AM) to infants and toddlers has been via jet nebulizer with face mask (FM). Continuous nebulization (CN) with a bronchodilator has been used to treat asthma symptoms. High flow nasal cannula (HNFC) is the treatment of choice for kids in respiratory distress. Globally, trans-nasal (TN) pulmonary AM delivery via HFNC has been receiving increasing clinical attention for comfort treatment, but, presents a challenge when used in conjunction with CN via FM. Several published in vitro studies demonstrated that AM deposition in infant lung models were vastly reduced when interfaced with a FM. Particularly, if applied on top of the HFNC appliance or if a pacifier is present or the baby is stressed. Furthermore, comfort levels for infant and parent may decrease and impact AM delivery.
Methods:
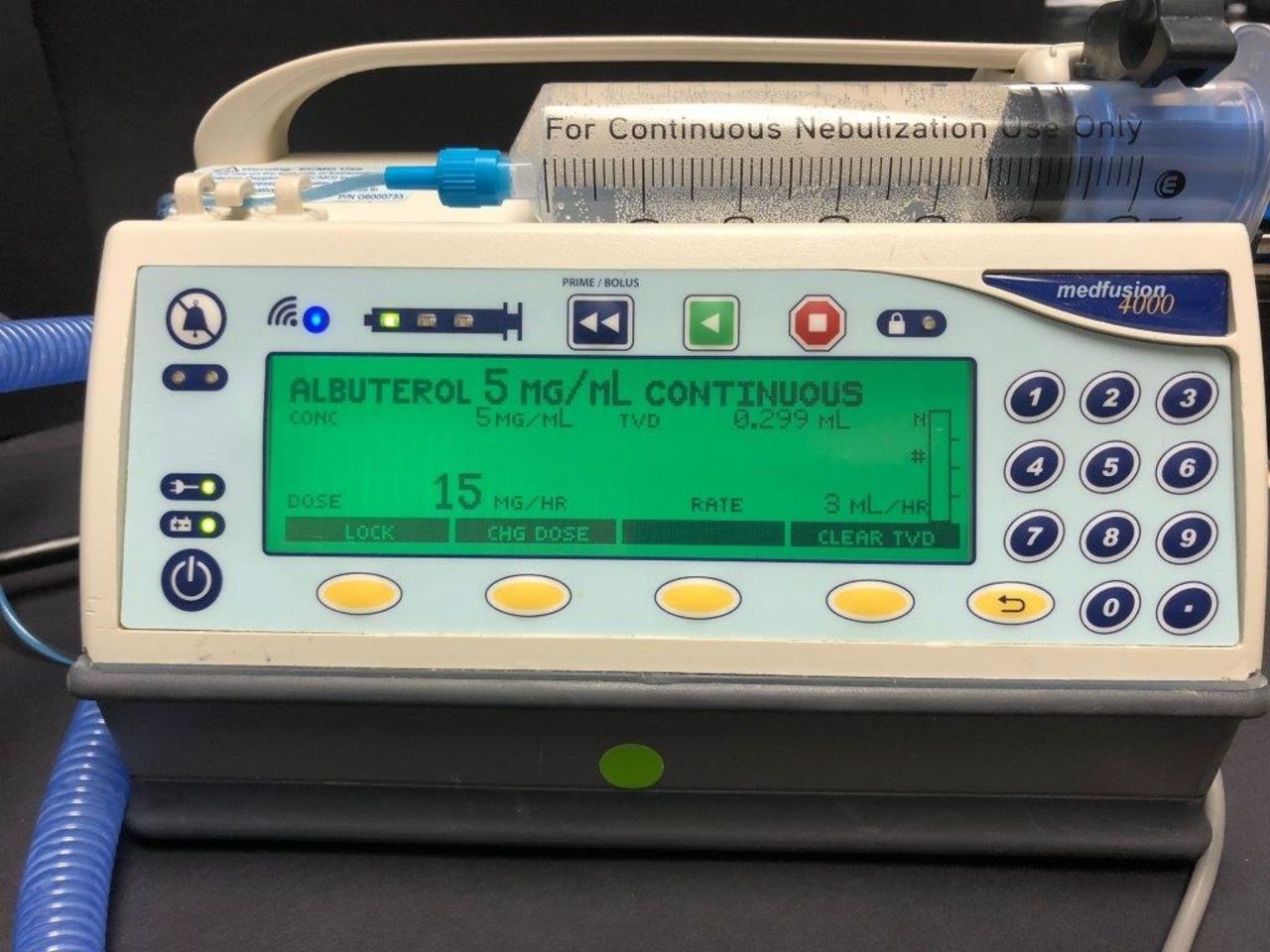
This study examined the performance of a vibrating mesh nebulizer (VMN) (Aerogen nebulizer), HFNC in conjunction with an MedFusion 4000 syringe pump (SP) to measure AM output at varied mg/h. The HFNC equipment consisted of: (MR850 humidifier chamber (HC) with an RT329 infant heated wire circuit), products by Fisher & Paykel Healthcare. Our Pharmacy Department added 0.5% (albuterol - 5 mg/mL continuous) into the SP library program. NaCL solution was substituted for albuterol, 20 mL was drawn up into a 60 cc syringe was connected to the SP. The VMN was installed on the dry side of the HC. An RT329 circuit was used during the evaluation with gas flows set at 2 to 6 L/min. The SP was programmed in mg/h. The liquid solution was dropped onto the VMN for the creation of aerosol mist.
Results:
Infusion rate equal aerosol output rate mL/h; 5 mg/h = 1 mL/h, 7.5 mg/h = 1.5 mL/h, 10 mg/h = 2 mL/h, 15 mg/h = 3 mL/h, and 20 mg/h = 4 mL/h.
Conclusions:
The SP performed up to manufactures specifications comparing set mL/h to total volume delivered in mL/h at different mg/h. The advantages of TN-AM via SP; precise drug concentration delivered in mg/h, no (NaCL) dilution required with less AM loss into the surrounding environment with more available for inhalation. The FM is not required and may improve comfort score and overall AM deposition. Liberation is achieved by modifying mg/h or frequency/h. More In vitro and in vivo investigations are needed to determine objective dose effectiveness of TN - AM delivery, or if different mg/h concentrations have an effect on clinical outcomes.
Get full access to this article
View all access options for this article.
