Abstract
Background:
Mechanical ventilation with positive pressure ventilation, causes damage to surfactant deficient lungs. Administration of surfactant via aryngeal mask airway (LMA) offers a less invasive method of delivery without the need for intubation and mechanical ventilation. Early studies have shown that placement of the LMA is less difficult and less traumatic for the patient. The goal of surfactant administration via LMA is to remain off of mechanical ventilation and avoid the harmful effects of positive pressure ventilation, which can lead to long-term morbidity. Literature review supports the existing knowledge that the use of less invasive methods for the delivery of surfactant in the neonate is comparable to endotracheal administration, with a decrease in FIO2, mechanical ventilator days, and overall length of stay. This study was designed to implement the existing evidence into our clinical practice to help improve the overall outcome of our patients.
Methods:
To compare retrospective rates from 2015-2016 of surfactant therapy failure following in and out surfactant via intubation with a prospective Quality Study using alternative surfactant administration. The objective of this Quality Study is to avoid mechanical ventilation in neonates’ ≥ 25 weeks/≤1500 grams with mild to moderate RDS.(Institutional review board (IRB) approval was obtained for this project.) Inclusion Criteria: Gestational age (GA) 25 weeks and greater and ≤ 1500 grams Age < 48 hours RDS as defined by an FIO2 requirement of 0.40 and greater on CPAP.
Results:
Retrospective Results: 2015- Average ventilator days (AVD)-14; 2016-AVD-15.5. Length of stay (LOS) 2015-65.2; 2016-59.3. CLD 2015-24%; 2016-14.3%. Prospective Results: AVD-2017-12.2; 2018-11.8. LOS-2017-55.8; 2018-54.0. CLD 2017-10.6%; 2018-9.8%.
Conclusions:
Our results support the use of a less invasive method to deliver surfactant efficiently to decrease intubation and mechanical ventilation days. Data showed a decrease in the use of oxygen, LOS, AVD, and CLD rates. This is a 5-year study, initial results support the use of less invasive surfactant delivery and data will continue to be collected and reviewed.
Carilion Children's LMA Procedures FY 17 -FY 19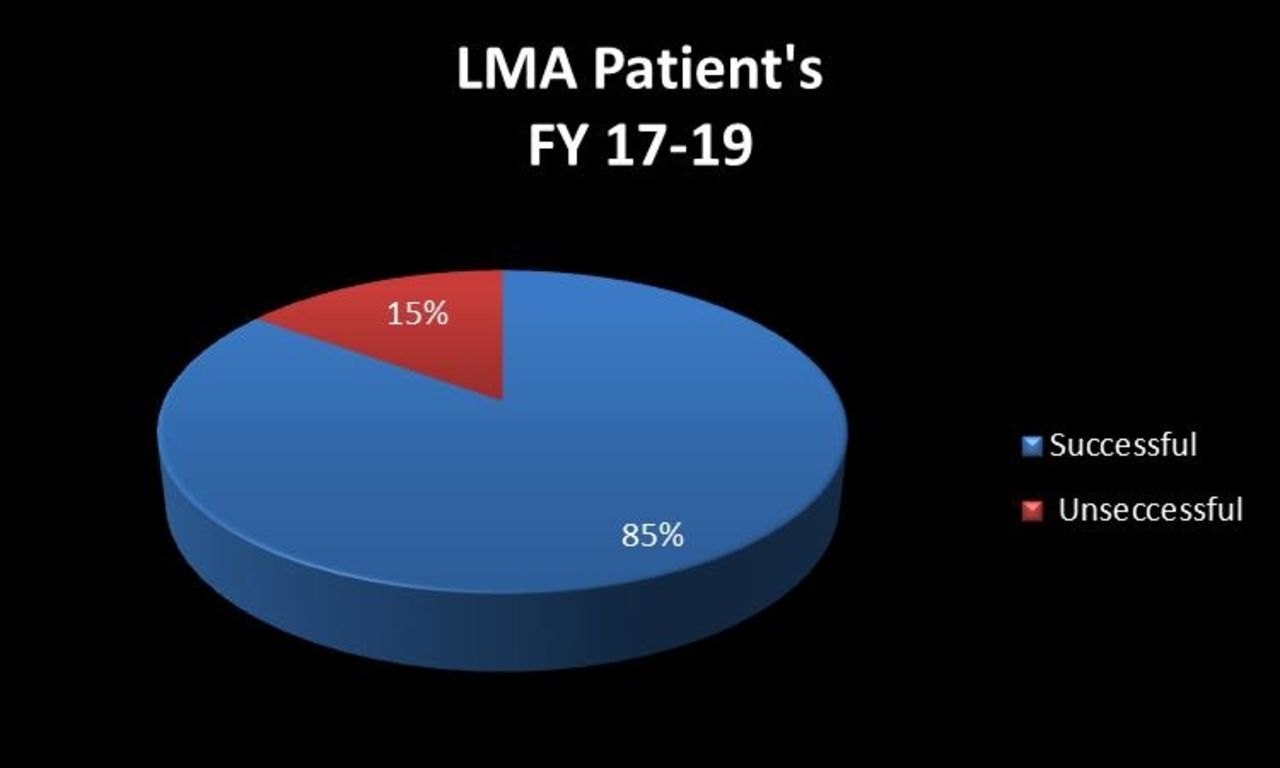
Get full access to this article
View all access options for this article.
