Abstract
Background:
Inhaled epoprostenol (iEPO) has been utilized to improve oxygenation in mechanically ventilated subjects with severe hypoxemia. However, there is no data reported on iEPO via high-flow nasal cannula (HFNC). Thus, we conducted this retrospective study to evaluate the clinical effects of iEPO via HFNC on oxygenation.
Methods:
IRB approval was obtained to review subjects in the adult intensive care units (ICUs) that had received iEPO via HFNC between July 2015 and April 2018. These subjects received iEPO via HFNC for more than 30 min to treat severe hypoxemia comorbidity with pulmonary hypertension or right heart dysfunction. Subjects' data was extracted from the electronic medical record. A patient was considered a responder to iEPO if their SpO2/FIO2 increased by 20% or more. Other outcome data included incidence of intubation, occurrence of systemic hypotension, ICU length of stay, and hospital survival.
Results:
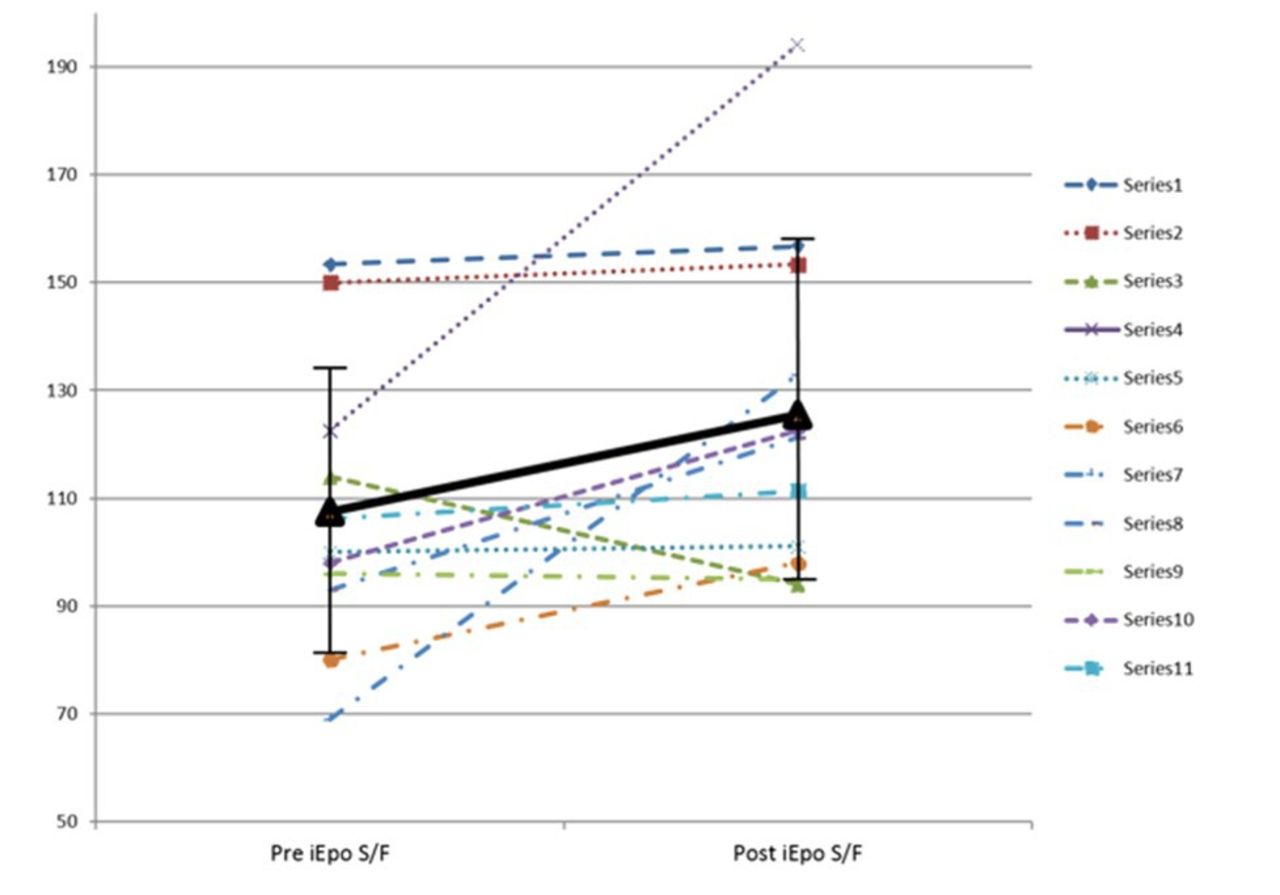
A total of 11 subjects were enrolled in the study, of whom 4 were male (36. 4 %), age 57.5 ± 22.1 years, and APACHE II score at ICU admission was 18.5 ± 5.7. Ten subjects had more than three chronic heart or lung comorbidities; seven of them used home oxygen. After inhaling epoprostenol, subjects' SpO2/FIO2 ratio improved from 107.5 ± 26. 3 to 125.5 ± 31.6 (P=0.026) within 30-60 min. Five (45.5%) subjects had SpO2/FIO2 improvement > 20%. Heart rate, blood pressure and respiratory rate were not significantly different. Intubation was avoided in seven subjects, and seven subjects were discharged home.
Conclusions:
This retrospective study demonstrated the feasibility and safety of iEPO via HFNC in improving oxygenation. Carefully titrating flow need and evaluating subjects' response may help identify responders and avoid delaying other interventions. This study supports the need for a larger prospective randomized control trial to further evaluate the efficacy of iEPO via HFNC in improving outcomes.
Get full access to this article
View all access options for this article.
