Abstract
Background:
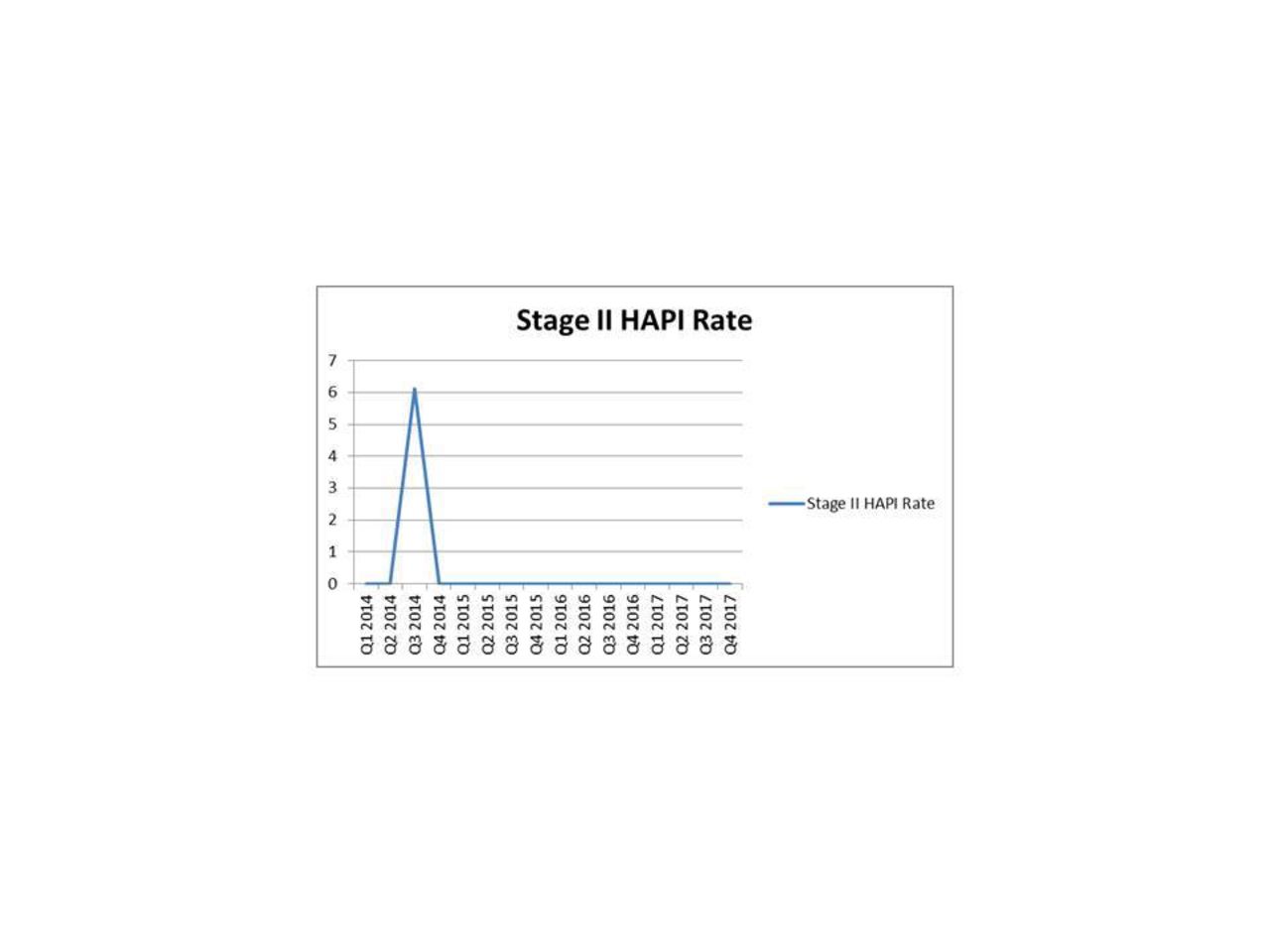
CPAP is used as a method to support ventilation and oxygenation in pre-term infants. In addition to having an immature respiratory system, pre-term infants also have very immature and fragile skin. The interfaces most commonly used with CPAP devices are known to cause pressure injuries in this patient population. The aim of this quality improvement project was to develop a multidisciplinary team to reduce the incidence of device related hospital-acquired pressure injuries (HAPI) in the NICU due to Q3 of 2014 presenting with an increased Stage II rate.
Methods:
A multidisciplinary team formed and evaluated risk factors for pressure injuries. A key driver diagram (see fig. 1) was created with the following primary drivers: 1. Skin integrity by providing education to identify pressure injury, applying skin protectant, and introducing an RT/RN coordinated skin assessments. 2. Evaluation of interface by alternating the interfaces of mask and prongs, adopting Bubble CPAP (BCPAP), encouraged a decreased usage of mask (due to fixation limitations), changing interfaces only as needed due to skin protectant. 3. Developed the team by creating a core RT/RN group, encouraged collaboration with wound service, and provided re-education on the usage of BCPAP and skin protectant. 4. Event review, communication, and awareness by implementing reporting process for skin concerns, reviewed events at multidisciplinary meetings, and displayed 'I'm bubbling' sign outside of patient's room. The outcome measure is the rate of HAPI per NICU patient census. Plan-Do-Study-Act (PDSA) cycles to test and learn from change were utilized. After the first PDSA cycle was complete HAPIs were continued to be noted. Upon completion on the second PDSA cycle HAPIs were eliminated; therefore, the team maintained the current assessment and patient care routine.
Results:
Quarter 3 of 2014 had an increase in device related HAPI in the NICU with a rate of 6.12. All subsequent quarters have maintained a HAPI rate of zero. It was noted that with over 3,100 BCPAP days per year, there was no increase in HAPI.
Conclusions:
Based on the above mentioned interventions our team has eliminated device related HAPI. We continue to monitor skin integrity with RT and RN simultaneously; assessing includes in and around nares, behind and in ears, and entire head. We continue to review literature and develop a best practice guideline for the utilization of BCPAP in premature infants.
Get full access to this article
View all access options for this article.
