Abstract
Background:
Aerosol therapy is commonly used during treatment with high-flow nasal cannula (HFNC) in the ICU. Heated humidification inside the HFNC tubing circuit leads to unwanted condensation, which may greatly limit the efficiency of drug delivery. In this study, we aimed to investigate whether a novel humidification system, which decouples temperature and humidity control, can improve the delivered dose.
Methods:
In a bench study setup, fluorescein sodium solution was nebulized using a vibrating mesh nebulizer in an infant HFNC circuit to measure the delivered dose, with a conventional versus a decoupled humidifier. The deposition of fluorescein inside each breathing circuit component and a final collection filter at the end of the nasal cannula was collected and quantified with a UV-vis spectrometer. Droplet sizes at different sections of the breathing circuit were measured by laser diffraction. Three air flows, 5, 10, and 15 L/min; and 2 nebulizer positions, (1) at the humidifier and (2) after the inspiratory tube, were tested.
Results:
The delivered dose decreased with increasing flow for the conventional setup and was higher when the nebulizer was placed after the inspiratory tube. Turning off the conventional humidifier 10 min before and during nebulization did not improve the delivered dose. The decoupled humidifier achieved a significantly higher (P = .002) delivered dose than the conventional setup. The highest delivered dose obtained by the decoupled humidifier was 62.4% when the nebulizer was placed after the humidifier, while the highest dose obtained for the conventional humidifier was 36.3% by placing the nebulizer after the inspiratory tube.
Conclusions:
In this bench study, we found that the delivered dose for an infant HFNC nebulization setup could be improved significantly by decoupling temperature and humidity control inside the HFNC circuit, as it reduced drug deposition inside the breathing circuit.
Introduction
Aerosol therapy (eg, nebulization of mucolytics and bronchodilators) is a common treatment for critically ill patients, including children, admitted to the ICU. 1 –4 In the ICU, aerosol therapy is often performed during various respiratory support modalities. One such respiratory support commonly used for pediatric patients is high-flow nasal cannula (HFNC), where a warm and humidified mixture of air and oxygen is delivered to the patient at relatively high flows through a nasal cannula with nasal prongs. Recently, there has been an increase in the routine use of aerosol therapy during HFNC. 5 However, there are concerns about the efficacy of nebulization during HFNC 6 as many studies reported that < 20% of the nebulized dose reaches the subject’s lungs. 5,7
Many parameters have been shown to affect the delivered dose during HFNC, such as ventilator flow, nebulizer type, and nebulizer position. 7,8 It was found that a low flow is more beneficial; 7 mesh nebulizers are more efficient than jet nebulizers, 6 and placing the mesh nebulizer before the humidifier gives a higher delivered dose. 8 Similar observations have been made for pediatric HFNC in terms of flow, 9 –11 nebulizer type, 12 and nebulizer position, 9 although the effects of the various parameters for HFNC for pediatric setups are less well studied than adult setups. 13 One other factor that poses a main limitation to effective/efficient nebulization during HFNC appears to be humidification, as high relative humidity (RH) may lead to undesired condensation on the tubing walls 14 and hence increased drug deposition in the tubing. It was observed that a higher dose could be delivered under non-humidified (dry) conditions for adult HFNC, especially at high flows. 15 Furthermore, it was found that using a heated and humidified HFNC setup may result in a clinically important amount of condensation water reaching the upper airway of preterm infants. 16 Although high RH is shown to negatively affect drug nebulization efficacy, it is currently not possible to control humidity independently from temperature as conventional humidification devices in the ICU humidify the air using water vapor where the RH is always near 100%. 17,18
This study aimed to improve the delivered dose of infant HFNC nebulization setups by controlling the humidity independently. To do so, we used a novel humidification device able to decouple the temperature and humidity control inside the breathing circuit. The decoupled humidification device is expected to improve drug delivery efficacy in 2 ways: First, by minimizing the formation of condensation during humidification, and second, by turning off humidification but not gas heating during nebulization, it allows partial drug aerosol evaporation to take place, thereby minimizing deposition in the breathing circuit. The performance of an infant HFNC setup incorporating the decoupled humidification device was evaluated in terms of delivered dose and compared with the conventional setup.
QUICK LOOK
Current knowledge
Aerosol therapy is commonly used during high-flow nasal cannula (HFNC) in the ICU, but concerns about drug delivery remain. Many parameters affect the delivered dose, such as the flow and the nebulizer position. High relative humidity (RH) has a negative effect on the delivered dose, especially for pediatric HFNC, but it is currently not possible to control humidity independently with conventional devices.
What this paper contributes to our knowledge
This proof-of-principle bench study demonstrated that the delivered dose during infant HFNC nebulization increased significantly by decoupling humidity control from temperature control with a new humidification device. The device turns off active humidification during nebulization, allowing the drug aerosols to partially evaporate and humidify the gas inside the breathing circuit, reducing undesired deposition while maintaining sufficient RH inside the breathing circuit.
Methods
HFNC circuit setup
The study was performed using an experimental bench setup replicating the infant HFNC setup in the pediatric ICU at the Amsterdam University Medical Center hospital. In the lab, dry air from an air compressor was used as the air supply to the HFNC circuit. The HFNC circuit consists of several components: the humidification system, the inspiratory tube, the nasal cannula, the T-piece, and the nebulizer.
Two different humidification systems as shown in Figure 1 were tested and compared. The first is the conventional coupled heater/humidifier device currently used for HFNC in the ICU (Figure 1A. This humidifier (F&P 950, Fisher & Paykel, Auckland, New Zealand) consists of a pot containing water heated by a heating plate underneath. This humidifier also controls the active heating of its specialized pediatric inspiratory tube (Neonatal Optiflow Junior heated circuit kit 950N40, 11-mm inner diameter, Fisher & Paykel). For this humidifier, the target temperature at the subject was set at 37°C, and the humidity level cannot be controlled independently. To study the effect of turning off this humidifier on the delivered dose for this setup, we first ran the system at the chosen flow (15 L/min) with the humidifier turned on until its display indicated it had reached the target 37°C. Subsequently, the humidifier (along with the tubing heating) was turned off, and we waited 10 min before starting nebulization. The humidifier was kept off during nebulization for this test. For all other experiments with this humidifier, the humidifier was kept on before and during nebulization.
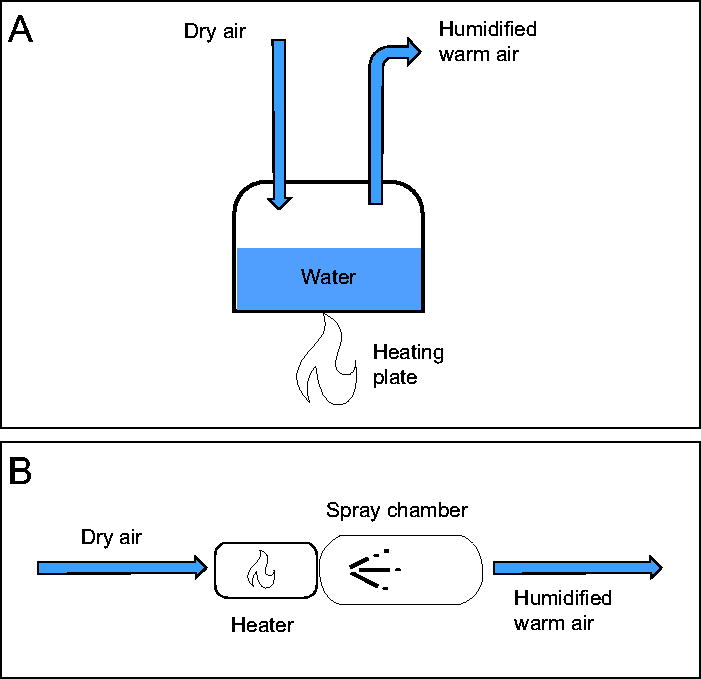
Illustrations of the 2 different humidification systems.
The second type of humidifier tested is a decoupled humidifier, shown in Figure 1B, where the temperature and humidity can be controlled separately through a heater and a spray chamber. A different inspiratory tube (VentStar pediatric tubing, 11-mm inner diameter, Dräger, Lübeck, Germany) than the one used for the conventional humidifier was used; it was not possible to use the same tubing for the 2 humidifiers because the conventional humidifier tubing is only compatible with the conventional humidifier. The humidifier and the inspiratory tube can be viewed as a package that together controls the temperature and humidity inside the breathing circuit. Therefore, an inspiratory tube with a similar heating mechanism (embedded heated wire and smooth bore) and tubing inner diameter was used for the decoupled humidifier. The decoupled humidifier was also set up to achieve 37°C at the patient both with and without active humidification. The target absolute humidity during HFNC when there is no nebulization is 40 mg/L. The following procedure was performed during experimental measurements with the decoupled humidifier: Before nebulization, the system was turned on, where a pump added moisture inside the circuit by spraying a fine mist of water. To provide the required energy to turn the mist into water vapor, the air was heated up just upstream of the spray injection point. Once the temperature and humidity measured at the end of the nasal cannula stabilized (no significant fluctuations) after around 10 min, the liquid pump was turned off and the heater was turned to a lower setting to maintain the gas temperature. The temperature and humidity probe was removed, and the collection filter was attached. Nebulization started 5 min after the liquid pump was turned off.
After leaving the humidifier, the warm humidified air is further actively heated in the inspiratory tube until it reaches the nasal cannula that no longer has active heating. For this study, experiments were conducted at 3 different infant HFNC air flows (5, 10 and 15 L/min). A single infant nasal cannula (OPT316 Optiflow Junior Nasal Cannula for Infant, Fisher & Paykel) suitable for this range of flows tested was used for all experiments.
A vibrating mesh nebulizer (Aerogen Solo, Aerogen, Galway, Ireland) was chosen because it can achieve higher drug deposition in the patient than a jet nebulizer, 8 attributed to a higher respirable fraction of aerosols and lower residual dose. 19,20 Figure 2 shows the nebulization breathing circuit used in the experiments, including the 2 feasible nebulizer positions to be tested for each humidifier. For the conventional humidifier setup (Figure 2A), the nebulizer positions are before the humidifier and after the inspiratory tube. For the decoupled humidifier setup (Figure 2B), the 2 nebulizer positions are after the humidifier and after the inspiratory tube.
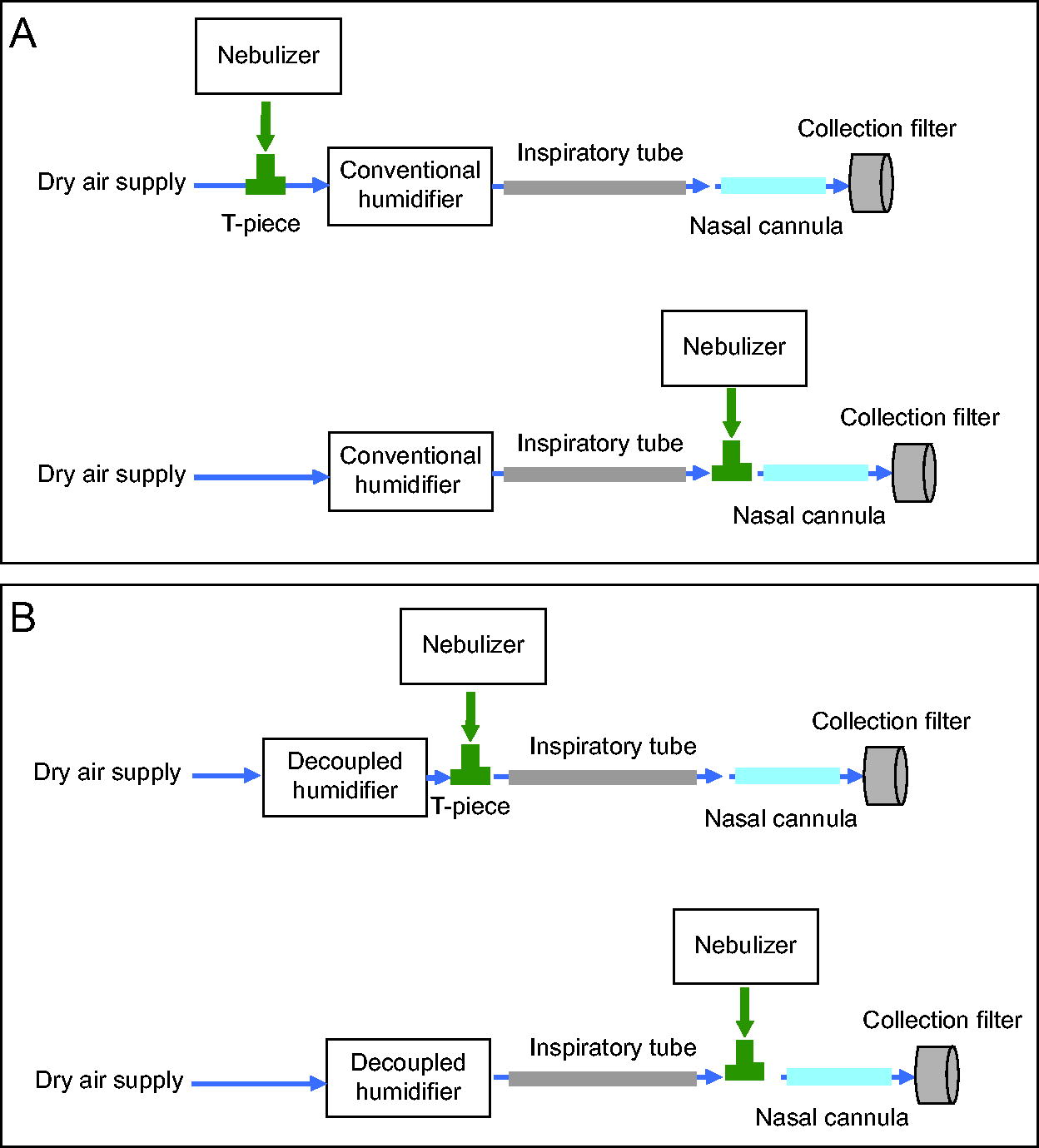
Experimental high-flow nasal cannula setup showing the various compartments and device positions for the 2 humidifiers.
Temperature and humidity measurements
To observe the gas conditions inside the HFNC circuit, the temperature and humidity at the end of the inspiratory tube were measured with a temperature and humidity sensor (SHT4x, Sensirion, Stäfa, Switzerland) while the temperature and humidity at the end of the nasal cannula were measured with a different temperature and humidity sensor (EE33, E + E, Engerwitzdorf, Austria). Each measurement was repeated 3 times. These measurements were performed separately from the delivered dose measurements so that the sensors would not influence the depositions.
Quantification of delivered dose
The primary outcome of this study was the dose captured in an absolute filter (Respirgard II 303, Vyaire Medical, Mettawa, Illinois) at the end of the nasal cannula, representing the delivered dose through the nasal prongs that is also the exposure dose to the patient. A secondary outcome is the deposited dose in each component of the HFNC circuit. A fluorescein sodium solution was used to model aerosol drug formulations in order to quantify the delivered dose and visualize depositions inside the various HFNC setup compartments. The solution is made from dissolving fluorescein sodium powder (C.I.45350 Acid Yellow 73, Sigma-Aldrich, Burlington, Massachusetts) in demineralized water and filtered before usage to prevent nebulizer clogging due to impurities in the solution. Three mL of 0.2% (w/w) fluorescein sodium solution was placed in the nebulizer drug compartment and nebulized to completion. The fluorescein sodium aerosols then deposited in various locations in the tubing, connectors as well as in the collection filter.
At the end of the experiment, all components of the circuit were disconnected, and each component was thoroughly rinsed with demineralized water, and the obtained solution was collected as the sample. The solution was then further diluted and quantified by measuring the absorbance at 460 nm using a UV-Vis spectrometer (Evolution 201, Thermo Fisher Scientific, Waltham, Massachusetts). Knowing the dilution procedure of the breathing circuit samples, the dose in each component was calculated as the ratio between the mass of fluorescein sodium in that component to the mass of the total amount of fluorescein sodium nebulized in the circuit.
Droplet size measurements
To investigate differences in deposition for the various setups, the droplet size distributions at the different sections of the circuit were obtained using a laser diffractor (Spraytec, Malvern Panalytical, Worcestershire, United Kingdom) as an additional secondary outcome. During droplet size measurements, isotonic saline solution (0.9% sodium chloride injection solution) was used for nebulization. The isotonic saline solution was used instead of water for injection to prevent the self-charging phenomenon during nebulization with the mesh nebulizer. Since water for injection is a non-conductive liquid, it leads to electrostatic charging of the droplets during nebulization that causes the droplets to fly toward the inner walls of the T-piece and reduces the nebulizer output to nearly zero. Using isotonic saline prevents this phenomenon and enables meaningful droplet size measurements. It is also more comparable to the (often electrically conductive) inhalation drug solutions used in the ICU. The measurements were performed < 2 cm from the end of the breathing circuit to minimize evaporation in the free air. The result was averaged over a fixed time interval with outliers removed. The median droplet size of each distribution was computed by the Spraytec software.
Statistical analysis
Statistical analysis was performed for the primary outcome. Each delivered dose quantification test case was repeated 3 times. The mean and SD of each independent test case performed in triplicates were calculated. A 2-sample Welch t test was used to check for statistical significance when comparing 2 independent values (statistical significance defined as P < .05). Differences in means between the delivered dose for the 3 flows, 2 nebulizer positions, and 2 types of humidifiers were evaluated using 2-way factorial analysis of variance (ANOVA). Statistical analyses were performed in R (R Foundation for Statistical Computing, Vienna, Austria).
Results
Effect of flow, nebulizer position, and humidification on the conventional humidifier setup
Figure 3 shows the effect of air flow and nebulizer position on the deposition in various components of the HFNC circuit. The delivered doses for various flows and nebulizer positions for the conventional humidifier setup are summarized in Supplementary Table S1. When the nebulizer was placed before the humidifier, as is common practice in the ICU, the delivered dose decreased with increasing flow. Drug losses occurred mainly in the nasal cannula and the inspiratory tube. In the inspiratory tube, the tubing section near the integrated sensor turned visibly yellow from accumulated fluorescein sodium during the experiments Supplementary Figure S1, while condensation water was observed to accumulate inside the small, bifurcated tubing of the nasal cannula (Supplementary Figure S2). Deposition in the inspiratory tube increased with the flow. It shows that the delivered dose of the conventional humidifier setup was generally low, with a highest delivered dose (collection filter dose) of 25.3 ± 5.5% at 5 L/min and a lowest delivered dose of 12.0 ± 0.7% at 15 L/min. The deposition in the collection filter was higher when the nebulizer was placed after the inspiratory tube, and the dependence on the flow became less. The increase in delivered dose due to the change in nebulizer position was significant at 10 L/min (P = .049) and 15 L/min (P = .039) but not at 5 L/min (P = .76). A maximum delivered dose of 36.3 ± 8.6% was achieved at 10 L/min. As the nebulizer was placed after the inspiratory tube, there was no deposition in the inspiratory tube. However, there was more deposition in the nasal cannula and the T-piece than when the nebulizer was placed before the humidifier. A 2-way factorial ANOVA suggests that the nebulizer position had a significant effect on the delivered dose (P = .001) for all flows, while the flow did not have a significant effect for both nebulizer positions (P = .26). The interactive effect of nebulizer position and flow on the delivered dose was not significant (P = .053).
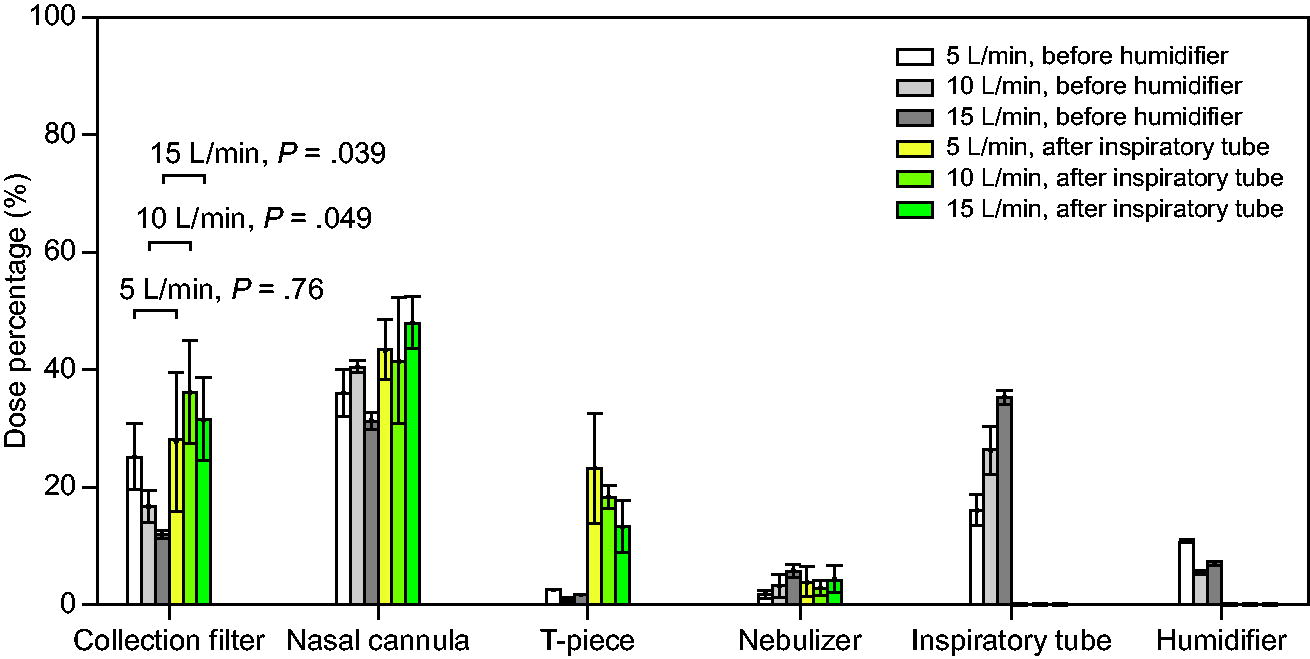
Deposition (mean ± SD) in various breathing circuit components for 2 different nebulizer positions when using the conventional humidifier. Collection filter (delivered dose), P values are indicated for the delivered dose.
To investigate the effect of humidification for the conventional humidifier setup, we obtained the delivered dose of the humidifier when it was turned off for 10 min to compare that with the delivered dose when the humidifier was left on. The nebulizer was placed before the humidifier, and a flow of 15 L/min was used for this comparison since this flow gave the lowest delivered dose (according to Fig. 3) for this nebulizer position. Contrary to what is expected, turning the humidifier off resulted in a significantly (P = .01) lower delivered dose (7.5 ± 1.3%) than when the humidifier was on (12.0 ± 0.7%). (Supplementary Figure S3) shows the temperature, absolute humidity, and RH at the end of the inspiratory tube during the 10 min after turning off the humidifier. The RH increased by 1.8% (from 78.9% to 80.7%), while the absolute humidity in the system was reduced by 52% (from 38.9 g/m3 to 18.6 g/m3) in 10 min. In fact, the RH in the tubing first increased to up to 91.6% before dropping again.
The droplet size distribution of aerosols exiting the T-piece, the inspiratory tube, and the nasal cannula were obtained by the laser diffraction setup. The median droplet size for different cases with the conventional humidifier are shown in Supplementary Table S2. The droplet size immediately after the T-piece was similar for all flows. The droplet size was smaller at the end of the inspiratory tube than before the inspiratory tube, and this reduction in droplet size was slightly more pronounced for higher flows. The droplet sizes were further reduced at the end of the nasal cannula.
When the nebulizer was placed after the inspiratory tube, at 5 L/min, the droplet size after the nasal cannula was similar to when the nebulizer was placed before the humidifier. The droplet size after the nasal cannula decreased more with increasing flows when compared to placing the nebulizer before the humidifier. At 15 L/min, the droplets could no longer be measured by the laser diffractor, possibly due to the low droplet concentration.
When the humidifier was turned off for 10 min before nebulization, the droplet size at the end of the inspiratory tube did not change by more than 0.1 μm when compared to the case with the humidifier on. The droplet size at the end of the nasal cannula became slightly (0.2 μm) larger.
Effect of independent humidity control with the decoupled humidifier
Figure 4 shows the deposition in various components of the breathing circuit for different flows and nebulizer positions for the decoupled humidifier setup. For this humidifier, placing the nebulizer after the humidifier led to a higher delivered dose than placing it after the inspiratory tube. Placing the nebulizer after the inspiratory tube led to significant deposition in the nasal cannula, more than twice the deposition in the collection filter at high flows. Placing the nebulizer after the humidifier led to 45.2–62.4% delivered dose for the range of flows tested. The highest delivered dose of 62.4 ± 6.4% was obtained at 10 L/min. For this nebulizer position, the highest losses in the circuit occurred in the nasal cannula and the T-piece, while the deposition inside the inspiratory tube was relatively low (< 10%).
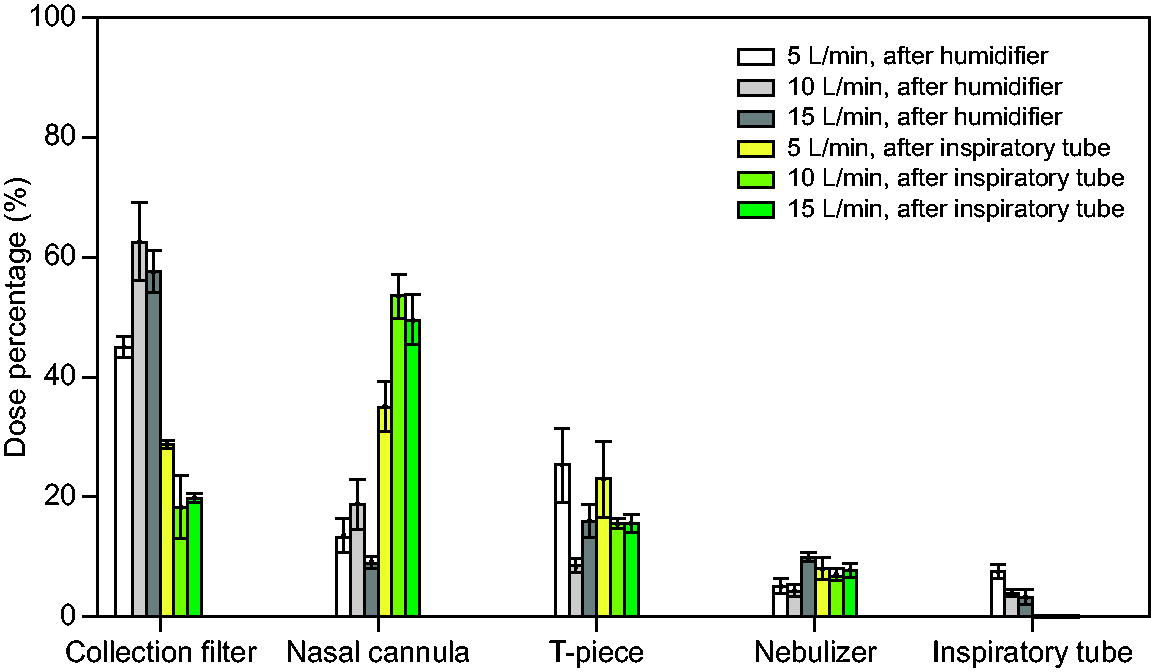
Deposition (mean ± SD) in various breathing circuit components for 2 different nebulizer positions when using the decoupled humidifier.
Figure 5 shows the comparison between the conventional setup and the decoupled humidifier setup in terms of deposition in all circuit components when the nebulizer was placed near the humidifier. For the conventional setup, the nebulizer was placed before the humidifier as in the ICU, while for the decoupled setup the nebulizer was placed after the humidifier. The delivered doses for the conventional and decoupled humidifier setup at various flows are summarized in Supplementary Table S3. At a flow of 15 L/min, the decoupled humidifier setup (57.5 ± 3.5%) achieved a delivered dose around 5 times the dose of the conventional humidifier setup (12.0 ± 0.7%) currently used in the ICU. The decoupled humidifier setup is considered to have had a significantly higher (P = .002) delivered dose than the conventional setup. A 2-way factorial ANOVA suggests that the humidifier type had a significant effect on the delivered dose (P < .001) while the flow did not (P = .87). The interaction effect of humidifier type and flow on the delivered dose was also significant (P = .002).
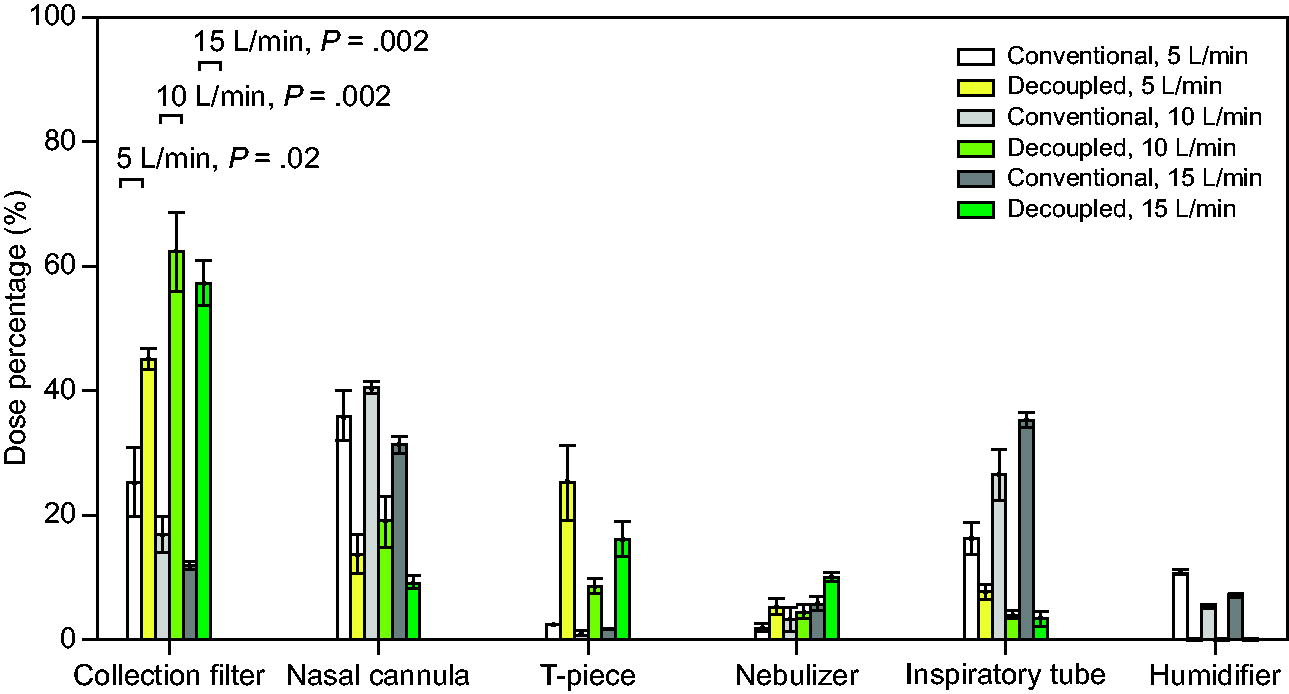
Deposition (mean ± SD) in various breathing circuit components for the decoupled humidifier setup versus the conventional humidifier setup. The nebulizer was placed close to the humidifier for both setups: before the humidifier for the conventional setup and after the humidifier for the decoupled setup. P values are indicated for the delivered dose.
During HFNC with active humidification and no nebulization, the absolute humidity at the end of the inspiratory tube was 38.1 ± 1.8 mg/L for a flow of 15 L/min, 41.0 ± 1.4 mg/L for a flow of 10 L/min, and 34.3 ± 5.1 mg/L for a flow of 5 L/min. Table 1 summarizes the RH inside the inspiratory tube at the beginning (immediately before the nebulizer was turned on and 5 min after the active humidification was turned off) and at the end of nebulization for different flows for the decoupled humidifier setup. The decoupled humidifier setup was not actively humidifying the air immediately before nebulization and had a much lower RH than the coupled humidifier that had near 100% RH. No condensation was observed before nebulization for the decoupled humidifier. Table 1 also shows that the aerosols generated by the nebulizer increased the humidity inside the tubing.
Relative humidity at the end of the inspiratory tube, at the start of nebulization (immediately before turning on the nebulizer), and at the end of nebulization for the decoupled humidifier setup
Data are presented as mean ± SD.
RH, relative humidity.
Supplementary Table S4 shows the median droplet size after the T-piece and after the inspiratory tube for various flows when the nebulizer was placed after the humidifier for the decoupled humidifier setup. It can be observed that the droplet size reduced more in the inspiratory tube for higher flows. At 15 L/min, the droplets exiting the inspiratory tube could not be measured anymore, probably due to small diameters and low droplet density. Furthermore, there is more reduction of droplet size in the inspiratory tube at 10 L/min and 15 L/min for the decoupled humidifier setup when compared to the conventional coupled humidifier setup.
Discussion
The main goal of this study was to investigate whether decoupling humidity control from temperature control could improve the delivered dose (primary outcome) during aerosol therapy for an infant HFNC setup. Using a decoupled humidifier, we found that independent control of humidity and gas temperature during nebulization led to a significantly higher delivered dose than the conventional coupled humidifier setup for all flows tested, as partial evaporation of the nebulized aerosols led to less deposition inside the breathing circuit while maintaining sufficient RH.
In the context of aerosol therapy during HFNC, this is the first study to address the negative effect of high RH levels in the breathing circuit on the delivered dose. We showed that the delivered dose could not be improved by turning off the conventional humidifier shortly before nebulization as it did not lead to a sufficient decrease in RH. To address the limitations of the conventional humidifier, we replaced it with a decoupled humidifier and demonstrated the new setup’s ability to improve the delivered dose by reducing deposition in the breathing circuit. Although tested on an infant HFNC setup, the decoupled humidifier is likely to improve the delivered dose for other patient groups as well, which could be a subject of further study. The increase in the delivered dose potentially enables aerosol therapy to become more efficient and effective during HFNC treatment of patients in the ICU.
Conventional humidifier setup
For validation, we first studied the conventional HFNC setup at various flows and nebulizer positions. Our results showed that a higher air flow led to a lower delivered dose when the nebulizer was placed before the heated humidifier. This negative influence of higher flows is consistent with findings in the literature. 7,9 –11 To understand why this is the case, we measured the droplet size distribution in the breathing circuit and deduced that undesired deposition in the circuit is likely due to 2 different contributions, both of which could result in the observed reduction of droplet size: (1) the collision of (larger) droplets into tubing structure due to inertial impact and turbulence and (2) absorption of droplets into the condensation of water vapor on the inner walls of the circuit. At high flows, the larger droplets are more likely to hit the wall or the integrated sensors in the tubing as they are less able to follow or drift in the air flow. There is also more turbulence in the tubing at higher flows, leading to more local deposition. Condensation mainly occurred inside the nasal cannula, likely due to the temperature drop from the heated inspiratory tubing to the unheated nasal cannula.
In addition, our results showed that the delivered dose of the conventional setup at higher flows could be improved by placing the nebulizer after the inspiratory tube. This is contradictory to previous adult 8 and pediatric results 9 in the literature, for which it was found that placing the nebulizer before the humidifier led to a higher delivered dose. This can be likely attributed to the different humidifiers and inspiratory tubes used. To our knowledge our study is the first on the topic using the F&P 950 humidifier system. The relevant adult 8 and pediatric 9 studies in the literature used different humidifier systems such as the F&P 850 that have different inspiratory tubes with very different internal structures and tube heating that would lead to very different depositions. Hence, it is not possible to make a fair comparison between our results and the literature due to the difference in the equipment used without looking into the effect of these structural and functional differences on deposition.
Furthermore, our study showed that turning off the humidifier briefly before and during nebulization did not improve the delivered dose because of the high RH that remains in the circuit tubing. The RH in fact increased due to the drop in temperature and the presence of condensation in the breathing circuit, despite a significant drop in the absolute humidity. This implies that evaporation in the tubing was not enhanced as hoped, as corroborated by the droplet size measurements. Overall, this shows that turning off the humidifier briefly before nebulization is not a feasible solution for improving the delivered dose of the HFNC nebulization system. This result adds weight to the recommendation against turning off the humidifier during HFNC in addition to safety concerns. 8,21 More importantly, this demonstrated the need for a decoupled humidification system, as it’s currently not possible to lower the air humidity independently from temperature.
Decoupled humidifier setup
As the conventional humidifier was not able to address the relevant concerns, we replaced it with a decoupled humidifier that enables independent control of heating and humidification in the HFNC circuit. During nebulization, the conventional humidifier humidifies the air by heating a pot of water while the decoupled humidifier humidifies the air by (partially) evaporating the nebulized drug aerosols. For the decoupled humidifier, placing the nebulizer after the humidifier was more advantageous than placing it after the inspiratory tube. This is mainly due to differences in deposition in the nasal cannula. When placing the nebulizer at the humidifier, as the droplets travel through the inspiratory tube where the RH is low, they experience partial evaporation before flowing into the nasal cannula. These droplets entering the nasal cannula are then smaller and thus less likely to hit the tubing wall.
When the nebulizer was placed near the humidifier, the decoupled humidifier setup gave a much higher (around 5 times) delivered dose than the conventional humidifier setup, especially at high air flows. One obvious contribution to the improvement is that the aerosol no longer passed through the heated water pot, where around 10% of the drug was lost. The more notable contribution was due to decreased undesired deposition in the inspiratory tube and nasal cannula. By turning off any active humidification during nebulization, the warm and low-humidity environment inside the tubing caused the nebulized aerosols to evaporate partially, which increased the RH inside the circuit. The smaller particles are also less likely to collide with the internal structures, thereby reducing deposition in the inspiratory tube and nasal cannula. As a result, the delivered dose for the decoupled humidifier setup was no longer limited at higher flows.
Clinical outlook
This study functions as a proof of principle and focuses on an infant HFNC setup. This is because the problem with condensation due to heated humidification is most pronounced for infant HFNC circuits due to the small tubing diameters, and the consequences of this condensation liquid potentially entering the airway are more severe for infants than larger patients. 16 In its current state of development, the decoupled humidifier cannot yet be safely used on patients, awaiting further optimization and research for formal medical device applications. Theoretically, this type of humidifier could be used by clinicians in all situations where active humidifiers are currently used, eg, HFNC, invasive mechanical ventilation, CPAP, and under certain (pre-)anesthesia settings, for patients of all ages. However, more extensive testing on a wider range of flows is required for application in other patient groups. The advantage of the decoupled humidifier is that clinicians could have the freedom to individually set the desired level of absolute humidity and temperature for their patients through the decoupled heater and humidifier. To optimize intermittent aerosol therapy during HFNC, a function could be built where the clinician could temporarily reduce the absolute humidity delivered by the system for a limited duration (eg, 10 min), after which the system will automatically revert to the original absolute humidity setting. In this time window, the clinician could apply aerosol therapy with much higher efficiency. Although active humidification was turned off completely during nebulization in this study, it is possible to set a minimum humidity level (eg, 10–20 mg/L) to increase the RH at the end of the nasal cannula based on patient requirements and air flows. In case the nebulizer stops unexpectedly (eg, due to clogging, running out of medicine), the decoupled humidifier should detect and compensate for the drop in RH by immediately increasing active humidification to maintain the minimum humidity level set by the user, limiting the risk of dry air entering the airway with the decoupled setup. This function needs to be demonstrated in future testing. A possible follow-up study would be to investigate the interactive effect between active humidification and the evaporation of the aerosol droplets and to find an optimal balance of the 2 processes to achieve the optimal combination of humidification and medication delivery at a certain air flow.
Limitations
This study has several key limitations. First, this proof-of-principle study currently only evaluated the delivered dose, meaning the dose delivered through the nasal prongs, and did not quantify the inhaled dose/lung dose which would be lower than the delivered dose due to losses in the nasal cavity and exhalation. Although breathing parameters are not expected to affect flow conditions inside the HFNC circuit and the delivered dose through the nasal cannula, they would affect the lung dose. No breathing simulator or mannequins were used in this study, which may help quantify lung dose and losses in the nasal cavity. Further testing in spontaneous breathing simulations and/or research subject settings is warranted.
Second, the nasal cannula was positioned horizontally to prevent deposition fluids from flowing between the various compartments due to gravity. This is not the clinical practice as the nasal cannula is positioned upward in the ICU, which leads to condensation liquid flowing to the lowest point in the circuit. This choice of placement might have led to large variability in the delivered dose when the nebulizer was placed after the inspiratory tube for the conventional setup. This is because there was a significant amount of condensation fluid inside the tubing for this test case and some of this fluid containing drug particles could have moved downstream to the next component during the experiment, going from the T-piece to the nasal cannula and from the nasal cannula to the collection filter. This could have led to an overestimation of the collection filter dose. However, placing the nasal cannula upward would not have avoided this issue completely, as we observed some fluid droplets shooting out of the nasal cannula when the nasal cannula was positioned both horizontally and vertically at high flows because of the small inner diameter of the nasal cannula tubes. The placement of the nasal cannula is not expected to affect the results for the test cases with the other nebulizer position or the decoupled humidifier, as they had much less condensation. Moreover, gravity does not affect the deposition of the small droplets that would reach the patient as they are dominated by diffusion.
Furthermore, the nebulizer position near the humidifier was different for the 2 humidifiers. For the conventional humidifier, the nebulizer was placed before the humidifier, while for the decoupled humidifier the nebulizer was placed after the humidifier. This was because placing the nebulizer immediately after the humidifier is not possible for this conventional humidifier due to direct electrical connections between the humidifier and the inspiratory tube required for heating control at this location. Even if it were possible, placing the nebulizer immediately after the conventional humidifier would likely lead to increased condensation and deposition in the nebulizer-humidifier connector, as corroborated by the literature with a similar humidifier setup, 22 making it likely less efficient than placing the nebulizer before the conventional humidifier, which is also the common clinical practice. On the other hand, it was also not possible to place the nebulizer before the decoupled humidifier as the aerosolized drugs would likely lose stability and medical properties due to the heater and contaminate the heater stage. Therefore, although it would be beneficial to have direct comparisons of the 2 humidifiers for the same nebulizer positions, this was, not possible due to equipment constraints. However, this inconsistency in nebulizer positions between the 2 humidifier setups does not affect the main result, which is that the decoupled humidifier setup achieved a much higher delivered dose than what is possible by the conventional setup for the flows tested.
Lastly, it is known that the parameters such as density, viscosity, and surface tension of the nebulized solution can affect droplet size distributions and depositions. The fluorescein solution we used to visualize and quantify depositions is similar to low-concentration drugs in the ICU such as albuterol in terms of density and viscosity, and it is also a salt solution. It is less representative of more viscous drugs such as antibiotics. Future testing with various drugs is needed to see how the deposition and droplet sizes might differ.
Conclusions
This work evaluated the delivered dose in aerosol therapy during HFNC treatment in a simulated infant setting for various flows and nebulizer positions when using a conventional humidifier versus a decoupled humidification system. We demonstrated that decoupling temperature and humidity control increased the delivered dose by enabling partial evaporation of the drug aerosol. Future research and development are needed to test, validate, and incorporate the decoupled humidifier in a clinical setting to provide more effective aerosol therapy during HFNC for ICU patients.
Footnotes
Authors' Contributions
Mr. Huijgen is an employee at Medspray BV. Dr. van Rijn is a cofounder of Medspray BV. Medspray BV developed the decoupled humidifier technology.
Disclaimer
The study was performed at the University of Amsterdam, Amsterdam, the Netherlands, and Medspray BV, Enschede, the Netherlands.
Funding Information
This study was funded by Dutch Research Council NWO, IPP Grant “Innovative Nanotech Sprays,” ENPPS.IPP.019.001.
Supplementary Materials
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Table S1
Supplementary Table S2
Supplementary Table S3
Supplementary Table S4
