Abstract
Background:
Mechanical power (MP) applied to the respiratory system (MPRS) is associated with ventilator-induced lung injury (VILI) and ARDS mortality. Absent automated ventilator MPRS measurements, the alternative is clinically unwieldy equations. However, simplified surrogate formulas are now available and accurately reflect values produced by airway pressure-volume curves. This retrospective, observational study examined whether the surrogate pressure -control equation alone could accurately assess mortality risk in subjects with ARDS managed almost exclusively with volume control (VC) ventilation.
Methods:
Nine hundred and forty-eight subjects were studied in whom invasive mechanical ventilation and implementation of ARDS Network ventilator protocols commenced ≤ 24 h after ARDS onset and who survived > 24 h. MPRS was calculated as 0.098 x breathing frequency x tidal volume x (PEEP + driving pressure). MPRS was assessed as a risk factor for hospital mortality and compared between non-survivors and survivors across Berlin definition classifications. In addition, mortality was compared across 4 MPRS thresholds associated with VILI or mortality (ie, 15, 20, 25, and 30 J/min).
Results:
MPRS was associated with increased mortality risk: odds ratio (95% CI) of 1.06 (1.04–1.07) J/min (P < .001). Median MPRS differentiated non-survivors from survivors in mild (24.7 J/min vs 18.5 J/min, respectively, P = .034), moderate (25.7 J/min vs 21.3 J/min, respectively, P < .001), and severe ARDS (28.7 J/min vs 23.5 J/min, respectively, P < .001). Across 4 MPRS thresholds, mortality increased from 23–29% when MPRS was ≤ threshold versus 38–51% when MPRS was > threshold (P < .001). In the > cohort, the odds ratio (95% CI) increased from 2.03 (1.34–3.12) to 2.51 (1.87–3.33).
Conclusion:
The pressure control surrogate formula is sufficiently accurate to assess mortality in ARDS, even when using VC ventilation. In our subjects when MPRS exceeds established cutoff values for VILI or mortality risk, we found mortality risk consistently increased by a factor of > 2.0.
Introduction
Mechanical power (MP) represents energy over time that, when applied to the respiratory system (lungs and chest wall: MPRS), is linked to both ventilator-induced lung injury (VILI) and ARDS mortality. 1 –4 In the absence of ventilators calculating MPRS directly from airway pressure (Paw)–volume curves (ie, “geometric” measurements once provided by the Bicore pulmonary mechanics monitor), mathematical formulas are needed. However, estimating MPRS by formula is complex and unwieldy, as it includes the variables minute ventilation, peak Paw, PEEP, and inspiratory flow in order to approximate the accepted standard (geometric area within Paw-volume curve).
In an attempt to address this obstacle, Chiumello et al 5 devised simplified (“surrogate”) formulas to calculate MPRS during either volume control (VC) or pressure control (PC) continuous mechanical ventilation (CMV) modes. They found both surrogate equations were highly correlated with direct (geometric) measurements of MPRS. Surrogate MPRS values averaged 0.8 J/min greater than direct, geometric measurements (8.64 ± 2.62 vs 7.84 ± 2.62, respectively, P = .01). 5
In this retrospective, observational study, we assessed whether reliance on the PC surrogate formula alone could accurately assess hospital mortality risk in subjects with ARDS managed with lung-protective ventilation (LPV) and when VC-CMV was used almost exclusively. The basis for doing so was that we recorded plateau pressure (Pplat) and driving pressure (PDR) as part of our quality assurance monitoring, not peak Paw and inspiratory flow. The justification for applying the PC surrogate formula in subjects managed with VC-CMV is described below in the Methods section (see Design Rationale).
Essentially this study asked whether the fraction of MP applied by the ventilator needed to overcome flow resistance properties of the ventilator-patient system is essential to assess mortality risk in ARDS from excessive MPRS. We approached answering this question by assessing (1) the association between PC surrogate MPRS and mortality, (2) whether MPRS increased correspondingly with increasing ARDS severity (ie, Berlin classifications) and its associated mortality risk, and (3) whether mortality differed at-or-below versus above MPRS thresholds previously association either with VILI or mortality as reported in other studies. 3,4,6,7
QUICK LOOK
Current knowledge
Mechanical power (MP) applied to the respiratory system (MPRS) is associated with ventilator-induced lung injury (VILI) and ARDS mortality. Because power is energy transferred to lungs (and is a composite of all variables known to cause and perpetuate lung injury), it is widely considered the most important variable to monitor in mechanically ventilated patients. Because ventilators currently don’t calculate power from the airway pressure–volume loops, cumbersome mathematical formulas are used to estimate power. However, simplified surrogate formulas accurately estimate power and can be used to assess mortality risk at the bedside.
What this paper contributes to our knowledge
The current study demonstrates that estimating MPRS using only the pressure control surrogate equation (even in subjects managed with volume control ventilation) discriminates survivors from non-survivors across both ARDS severity classifications, as well as MP threshold levels associated with increased VILI and mortality risk. This indicates complex equations accounting for the resistive components of MP are not necessary to assess the mortality risk from MP in ARDS.
Methods
Design rationale
The PC surrogate formula (ie, MPRS = 0.098 × tidal volume [VT] × breathing frequency × (PEEP + PDR)
5
was used to assess mortality risk, without distinguishing CMV modes (ie, PDR = Pplat−PEEP vs peak Paw−PEEP, and excluding the corresponding inspiratory flow). The rationale for applying this simplified equation to both VC-CMV and PC-CMV is as follows: Pplat not peak Paw reflects the stress applied to the lungs and chest wall at end inspiration. In contrast, peak Paw and inspiratory flow largely reflect endotracheal tube resistance. Even when tube deformation and lumen occlusion (which occurs clinically) are absent, its frictional resistance approximates that of lung resistance in early ARDS (∼10 cm H2O).
8,9
Moreover, in terms of VILI risk, the relevance of accounting for frictional resistive forces is controversial.
2,10
In ARDS, the primary source of VILI is the uneven distribution of lung strain and stress during inspiration. Uneven lung strain-stress is reflected in measure of viscoelastic resistance: a phenomenon that only can be captured during a 2–5 s end-inspiratory pause when gases redistribute and lung pressures equilibrate (see Supplementary Data S1).
11
–13
Clinically, we used a 0.5-s pause time per ARDS Network (ARDSNet) ventilator protocols.
14
Therefore, Pplat measured at 0.5 s likely reflects some portion of the mechanical unevenness believed to cause VILI. MPRS also reflects the energy needed to overcome the elastic, viscoelastic, and resistive properties of the chest wall, as well as its inertial properties.
15
In the presence of morbid obesity or abdominal compartment syndrome, chest-wall elastance is markedly increased,
16
and inertial forces are no longer negligible.
17
In light of these factors, as well as the absence of esophageal manometry (to isolate the pulmonary component), all MPRS techniques are crude approximations for estimating its impact on VILI and ARDS mortality. Likewise, similar limitations apply when trying to separate artificial airway frictional resistance from that of the pulmonary airways.
Subjects
The 1,995 subjects in our ARDS LPV quality assurance database (2002–2017) have been described previously. 18,19 In this study, a subset (948 subjects) was selected based on the following criteria: (1) having met the Berlin definition of ARDS, 20 (2) both CMV and ARDSNet protocols 21,22 were initiated within 24 h of ARDS onset, and (3) subjects having survived > 24 h after protocol initiation (Fig. 1).
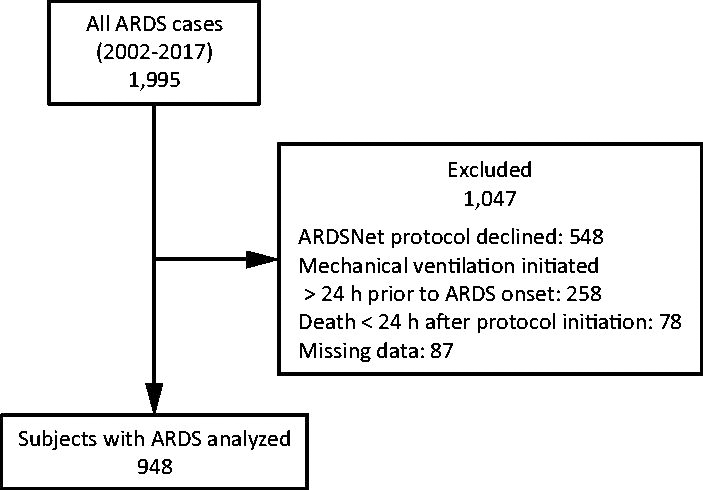
Subject selection flow chart. ARDSNet, ARDS Network.
The justification for these criteria are, first, those mechanically ventilated prior to ARDS onset may have been exposed to VILI. Second, our focus was the impact of MPRS on mortality in the exudative phase of ARDS. After ∼48 h, lung injury begins to transition to a proliferative or fibrotic stage, 23 therefore possibly producing changes in lung mechanics and MPRS. Thus, excluding subjects in whom ARDS onset preceded ARDSNet protocol initiation would introduce interpretive ambiguity by mixing subjects at different stages of ARDS evolution. Third, evaluating subjects who survived beyond the day of ARDS onset allowed time for potential optimization of LPV. Fourth, excluding those who died on the day of ARDS onset eliminated additional ambiguity because whatever MP moribund subjects were exposed to over a matter of hours prior to death cannot reasonably be attributed to mortality risk.
Our sample consisted of 108 subjects with mild (11%), 440 with moderate (46%), and 400 with severe ARDS (42%). During the study period, subjects were managed almost exclusively with VC-CMV, as stipulated by the ARDSNet protocols. 21,22 Approval to use our quality assurance data was granted by the University of California, San Francisco Institutional Review Board (approval reference number: 268589).
Measurements and calculations
Lung and chest wall subcomponents of MPRS (MPL and MPCW, respectively) were estimated using respiratory system elastance (ERS) data and its lung (EL) and chest wall (ECW) subcomponents in ARDS subjects from our previous study: 24 EL/ERS (0.74), ECW/ERS (0.26). These values were consistent with other studies (Supplementary Data S2). Other calculations included comparing MPRS to normal resting power (MPRS ratio) 25 using a value of 4 J/min, 26 MPRS index adjusted power to predicted body weight, 27 and Δ MPRS as the change in power ∼24 h after protocol initiation (ie, a signifier for LPV optimization).
Assessments
MPRS and its subcomponents were assessed from several perspectives: (1) the overall mortality risk for sample population; (2) mortality differences between non-survivors and survivors across Berlin definition classifications; (3) changes in MPRS from initiation of LPV to the first full day of ARDSNet protocol management between non-survivors and survivors; and (4) mortality risk between subjects falling at-or-below versus above MPRS thresholds of 15, 20, 25, 30 J/min. These corresponded with estimated MPL thresholds of 11, 14, 18, and 22 J/min, respectively.
We tested threshold values that approximated those of preclinical studies of VILI induced in normal lungs and a clinical study of ARDS. One preclinical study found an MPRS threshold of 25 J/min (MPL of 13 J/min) was associated with greater lung damage. 28 In their subsequent study, MPRS thresholds of 15 J/min and 30 J/min produced morphologic and histologic injury. 6 In another preclinical study, an MPL 15 J/min (MPRS of 18 J/min) produced whole lung edema. 7 The clinical study found that an MPRS threshold of 19 J/min was associated with increased mortality risk. 4
Statistical analysis
Data are expressed as median and (25-75%) interquartile range, as all variables failed a normality test (D’Agostoni and Pearson method). Direct comparisons between non-survivors and survivors were analyzed by Mann-Whitney test. Cross-group comparisons were done by Kruskal-Wallis and Dunn post test. The Fisher exact test was used to assess mortality risk above and below the aforementioned thresholds. Univariate logistic regression was used to assess the mortality risk of each MP-associated variable (eg, MPRS, MPL, MPCW). Alpha was set at 0.05.
Results
Achieving LPV goals
Regarding traditional pulmonary mechanics measured ∼24 h after ARDSNet protocol initiation, the most salient findings (for both non-survivors and survivors) were 75% of Pplat and VT values met protocol goals (Supplementary Table S1). Respiratory system compliance was significantly lower, and PDR was significantly higher among non-survivors.
Mortality and MP
Hospital mortality was 35.6%, and all MP variables were significant predictors of mortality risk (Table 1). For every 1 J/min increase in MPRS, mortality risk increased by 6%. The MPRS ratio of 5.8 (4.4–7.8) was ∼6-fold greater than normal (ie, ∼4 J/min). MPCW was associated with a higher mortality risk than MPL. MPRS index appeared to carry a more pronounced mortality risk.
Mortality risk for various aspects of mechanical power in ARDS
All tested variables P < .001.
MPRS, power applied to the respiratory system; MPL, power applied to the lungs; MPCW, power applied to the chest wall; MPRS ratio, applied power relative to power transfer under normal physiologic conditions at rest (ie, 4 J/min); MPRS index, power normalized to predicted body weight; Δ MPRS, change in applied power 1 d following initiation of lung-protective ventilation.
Mechanical power characteristics across Berlin definition ARDS classifications
All MP measures increased significantly with increasing ARDS intensity: moderate versus mild (P = .02), severe versus moderate (P < .001), and severe versus mild (P < .001) (Fig. 2). The median increase in MPRS was 113% between mild to moderate ARDS, 116% between moderate and severe ARDS, and 131% between mild and severe ARDS. Likewise, MPRS index increased with ARDS intensity: 0.30 J/min/kg predicted body weight, 0.36 J/min/kg predicted body weight, and 0.42 J/min/kg predicted body weight for mild, moderate, and severe ARDS, respectively, with the same proportional increases and associated P values described above.
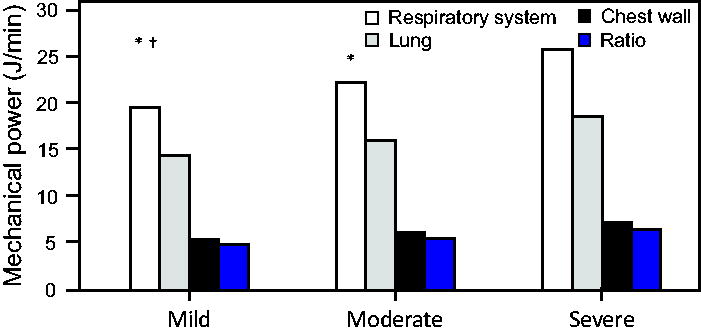
Respiratory system mechanical power and its lung and chest-wall subcomponents across Berlin definition classifications. * P < .001 compared to severe ARDS. † P = .02 compared to moderate ARDS.
Mortality and changes in MP from ARDS onset to the following day
Particularly noteworthy was that from ARDSNet protocol initiation to the following day MPRS among all subjects decreased by −0.9 (−4.2 to 2.4) J/min from 24.7 (18.5–32.7) J/min to 23.3 (17.6–31.3) J/min (P = .02). However, non-survivors had an increase in MPRS of 0.9 (–5.1 to 6.8) J/min, whereas in survivors MPRS decreased by –1.5 (–5.9 to 2.5) J/min (P < .001). The corresponding change in MPL among non-survivors and survivors was 0.7 (–3.7 to 4.9) J/min versus –1.1 (–4.3 to 1.8) J/min (P < .001). In other words, by ∼24 h into ARDS a difference in MPRS of 2.4 J/min (MPL of 1.8 J/min) had already separated non-survivors from survivors.
Differences in MP and mortality between Berlin classifications
When differences in MPRS and its subcomponents were compared between non-survivors and survivors across Berlin classifications, median MPRS among non-survivors always exceeded 24 J/min in contrast to survivors (< 24 J/min) irrespective of ARDS severity (Table 2). Extrapolating these findings to corresponding estimates of MPL, the median values among non-survivors always exceeded 17 J/min regardless of ARDS severity. Likewise, MPRS ratio was > 6 times normal values in non-survivors and < 6 in survivors. Mortality rates observed with increasing ARDS severity were consistent with previous findings. 20
Power distribution across Berlin definition categories between non-survivors and survivors
P = .034.
P < .001.
P = .033.
MPRS, power applied to the respiratory system; MPL, power applied to the lungs; MPCW, power applied to the chest wall; MPRS ratio, applied power relative to that at rest under normal physiologic conditions (ie, 4 J/min); MPRS index, power normalized to predicted body weight.
MP thresholds and mortality risk
Across the 4 MPRS and corresponding MPL thresholds, mortality steadily increased for both at-or-below and above cohorts at each threshold level. However, mortality in the above cohort was consistently and substantially greater: its associated risk factor always exceeded 2.0 (Table 3). The absolute cumulative mortality increased by 6% (23−29%) in the at-or-below cohort compared to 14% (38–51%) in the above cohort. This translated into total mortality rate increases of 126% versus 134%, respectively. The highest separation in mortality between cohorts (Δ22%) occurred at the MPRS (MPL) threshold of 30 (22) J/min, with a mortality of 51% (Fig. 3).
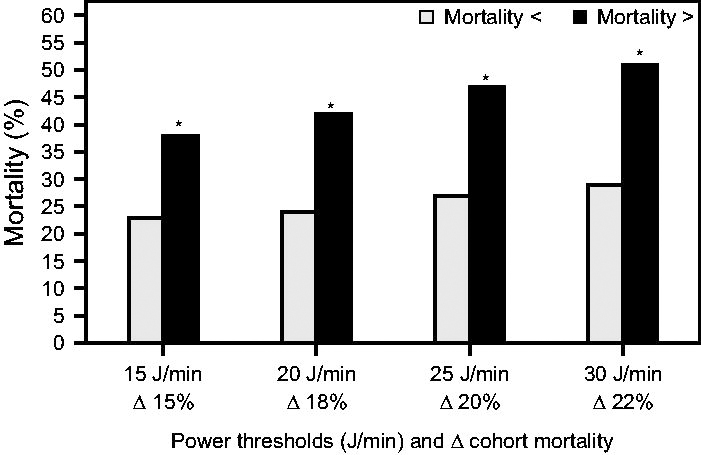
Mortality across 4 respiratory system mechanical power thresholds. Percentages listed above the bars reflect the difference in mortality between the above-threshold cohort versus the at-or-below threshold cohort. * P < .001 for mortality comparisons between cohorts measured at each threshold.
Mortality rates and risk for subjects with ARDS above and below mechanical power thresholds
P < .001 for all mortality differences between each power threshold.
MPRS, mechanical power applied to the respiratory system; MPL, mechanical power applied to the lung.
Discussion
Our main finding was the PC surrogate MPRS equation 5 was strongly associated with mortality across all ARDS severity classifications. This despite subjects managed almost exclusively with VC-CMV. Eighty-five percent of our subjects had MPRS levels > 15 J/min (associated with VILI in animals with normal lungs), 6 and 65% had MPRS levels above 20 J/min that exceeded a mortality threshold found in subjects with various etiologies of acute respiratory failure (Table 3). 4
Interestingly, 34% of subjects who met ARDSNet study mortality risk–exclusion criteria with corresponding MPRS ≤ 25 J/min had similar mortality to 3 ARDSNet trials that occurred during our data collection period (24–27% vs 25–28%, respectively). 22,29,30 Yet, a particularly worrisome finding was mortality exceeding 50% in those whose MPRS exceeded 30 J/min (MPL > 22 J/min): a mortality rate associated with late 20th century ventilator management prior to the adoption of LPV. 31,32
Elevated levels of MPRS associated with VILI also were observed in our subjects. This has practical implications for achieving safer threshold levels (ie, MPRS < 15 J/min, MPL < 12 J/min) proposed by others. 33 Our results suggest the likelihood of achieving these stringent threshold (without liberalizing sedation usage or dosing) is improbable. Moreover, in severe ARDS, such thresholds might require extracorporeal membrane oxygenation in cases that otherwise wouldn’t meet salvage criteria. The vast majority of survivors across ARDS severity classes were managed with MPRS > 15 J/min: 74% (mild), 81% (moderate), and 87% (severe). Overall, 59% of our survivors required MPRS > 20 J/min (MPL > 14 J/min).
Our results support our hypothesis accounting for resistive work is unnecessary for clinical purposes. It also supports our reasoning that the major sources of VILI are elastic and viscoelastic properties of a heterogeneously injured lung: a substantial portion of which might be captured with a brief pause time (0.5 s) that we adopted by adhering to the ARDSNet protocols. 34
In reviewing the literature, a nettlesome problem became apparent: How should MPRS be targeted since studies have used different equations with varying complexity? Among surrogate formulas relevant to our study, some excluded PEEP (ie, static MP) but included 50% of peak Paw multiplied by PDR. 2 Others included PEEP and used 50% of PDR. Although not explained, a 50% correction factor is an attempt to estimate the absorption of pressure (energy) across the lung parenchyma. 2,3
Of particular interest to us were studies done by Xie et al 10 and Costa et al 2 who used a 50% correction factor as a means of estimating “elastic power” (EP) (Supplementary Data S3): Xie and colleagues 10 found EP was highly accurate in identifying severe ARDS (threshold of 14.6 J/min). Costa and colleagues 2 parsed EP into its static and dynamic subcomponents (as well as a resistive subcomponent) to estimate total MP. Their relevance to our study was that only the dynamic EP (and not the resistive subcomponent) was associated with mortality risk: odds ratio (95% CI) of 1.31 (1.19–1.45). 2
These 2 studies are directly related to our estimates of MPL (0.74 correction factor) compared with their estimates of EP (0.5 correction factor). 10,2 In our post hoc analysis, we found that our MPL and EP using their equations produced the same results (Supplementary Data S3). However, because the equations differ, the conversion factors are not interchangeable. Ours requires a conversion factor more reflective of the EL/ERS relationship reported in ARDS.
We also compared our mortality to that found by Wu et al 35 who used the same surrogate formula as we used. They also observed elevated levels of mean MPRS at baseline ∼28 J/min (VT 8.4 mL/kg predicted body weight) compared to our baseline measurements (when expressed as mean) of ∼27 J/min (VT 7.1 mL/kg predicted body weight). A similar finding was that after 2 d of LPV they reported average MPRS had decreased in survivors and increased in non-survivors (mean total difference of Δ 5.1 J/min). In our subjects, the total difference between non-survivors and survivors after ∼1 d was Δ 2.5 J/min.
We feel compelled to comment upon two of our other findings. First, that MPCW carried a greater mortality risk than MPL was baffling (being a derived variable not directly relevant to VILI). The only cogent explanation we can offer is that MPCW was a latent signifier for subjects with intra-abdominal hypertension and, therefore, associated with heightened mortality risk. 36 Unfortunately, our database lacked supporting information. Second, the higher mortality risk associated with MPRS index is concerning. We speculate that ECW was lower in smaller stature, leaner subjects so that a higher fraction of MPRS was applied to the lungs, thereby increasing VILI risk.
The limitations of our study were its retrospective nature and the limitations of our quality assurance data. Most vexing was the potential contribution of subject effort to MPRS during assisted CMV. Our sedation practice (Particularly during the first days after ARDS onset was to promote synchrony while maintaining VT and Pplat targets). Passive ventilation was pharmacologically induced when asynchrony interfered with LPV goals and gas exchange stabilization. This practice was followed with particular fidelity during the initial days after implementing ARDSNet protocols.
Conclusions
In summary, for bedside management in the absence of automated ventilator measurements of MPRS, the PC-CMV surrogate formula is a reasonable method to estimate MPRS even when using VC-CMV. Despite the ambiguities and limitations described above, surrogate estimates of MPRS are strongly associated with mortality risk across threshold values and degrees of ARDS severity. Our data suggest that exceeding an MPRS threshold of 30 J/min (MPL of 22 J/min) greatly increases mortality risk when it occurs early in the exudative phase of ARDS. Prospective trials are needed to examine varying MPRS safety thresholds across both different severity classifications and pathological stages ARDS. Of equal importance, it would behoove such studies to assess the impact of MPRS thresholds on competing needs such as its impact on sedation requirements and the related issues of duration of mechanical ventilation and ventilator-associated events.
Footnotes
Author Disclosure Statement
Mr. Kallet discloses a relationship with ContinuED. Dr. Lipnick has disclosed no conflicts of interest.
Elements of this paper were published as abstracts in R
Supplementary Material
Supplementary Data S1
Supplementary Data S2
Supplementary Data S3
Supplementary Table S1
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
