Abstract
Background:
The 2014 American Academy of Pediatrics recommendation for CPAP as an alternative to mechanical ventilation for treatment of neonatal respiratory distress prompted a rapid shift to noninvasive respiratory support. Since most patients receive nasal bubble CPAP a concomitant increase in nasal pressure injuries followed. This prospective observational study aimed to develop strategies to reduce nasal mask pressure injury in neonates by (1) quantifying CPAP mask–interface pressure and (2) assessing placement variability.
Methods:
A micro-electromechanical systems pressure sensor was modified for contact pressure measurements with silicone embedding and calibrated. The CPAP generator and interface components were sized for a 24-week neonatal simulator. Thirteen neonatal ICU staff placed the simulator on CPAP at 6 cm H2O and 8 L/min of flow with no humidification. Pressure was measured at 3 locations (the forehead, nasal bridge, and philtrum) in triplicate (3 measurements/site). Descriptive statistics; a location-specific, one-way analysis of variance with a Tukey post hoc test; and a 2-sample paired t test of the means of the first and last triplicate were performed.
Results:
Pressure ranged from 12–377.3 mm Hg. The mean [SD] interface pressure at the philtrum was significantly higher than both the nasal bridge and the forehead (philtrum 173.9 [101.3], nasal bridge 67.8 [28.9], and forehead 79.0 [36.9], P < .001). CPAP fixation varied, including bonnet placement, trunk angle, mask compression, use of hook and loop extenders, and level of vigorous bubble feedback achieved.
Conclusions:
This study developed a modified pressure sensor for quantifying the pressure exerted by a nasal mask on facial skin. Maximum pressures were higher than those previously reported. Inter-individual differences were present in both quantitative and qualitative measures of pressure. Reduction of NIV-associated pressure injuries may be achieved through NIV fixation technique training and improved nasal mask stability and size increments.
Introduction
Preterm birth (< 37 weeks gestation) represents 5–18% of births globally, and the resulting complications are the leading cause of death in neonates. 1 –3 One of the most common complications of preterm birth is respiratory distress (eg, transient tachypnea of the newborn, neonatal pneumonia, respiratory distress syndrome, meconium aspiration syndrome, and pulmonary hypoplasia) with an incidence of 6.7% overall and 30.0–65.6% within the preterm population. 4,5 Many modalities of respiratory support have been utilized to treat respiratory distress in neonates, including mechanical ventilation. A meta-analysis concluded that noninvasive ventilation (NIV), specifically CPAP, compared to invasive ventilation significantly reduces bronchopulmonary dysplasia and mortality in very low birthweight neonates. 6,7 In 2014, these data prompted the American Academy of Pediatrics (AAP) to strongly recommend a diverse range of NIV modalities, including nasal bubble CPAP, as an alternative to routine intubation and ventilation. 8
The AAP’s policy statement expedited the clinical shift to NIV, especially nasal bubble CPAP. From 2010–2018, it was reported that the use of NIV respiratory support increased 8.7%/y, with most patients receiving nasal CPAP. 9 While data on the number of patients days on NIV is sparse, one level III neonatal ICU (NICU) documented that 100% of neonates born < 28 weeks gestational age (GA), 90% of neonates born between 28–31 weeks GA, and 35% of neonates born between 32–35 weeks GA required respiratory support, of which 22%, 42%, and 65% were NIV, respectively. 10 A rapid switch to NIV combined with increasing survival rates for extremely premature neonates resulted in a concomitant increase in NIV-associated pressure injuries. In the largest study of CPAP-related injury, Fischer et al 11 found that 90% of neonates < 28 weeks GA, 77% of those < 32 weeks GA, and 28% ≥ 32 weeks GA developed a nasal pressure injury, with GA and birthweight negatively correlated to the severity of the injury. One level IV NICU reported a notable increase in the frequency of stage II or greater pressure injuries after the implementation of the AAP guidelines for NIV support in preterm infants (0.01 pre guideline vs 0.34 post guideline/100 NIV days). 12
Nasal CPAP is delivered via nasal mask, binasal prongs, or a combination of both. The choice of the delivery interface depends on several factors, including GA, day of life, and facial anatomy. Differences in CPAP application and fixation may affect how an interface is selected, the frequency of change, and the amount of strap tension required to maintain a seal. Nasal prongs primarily cause nasal septum injury, while nasal masks primarily cause nasal bridge injury, though in the case of alternating use it is unclear how much each contributes to regional pressure injuries. While the precise etiology and evolution of pressure injury, especially in underdeveloped neonatal skin, are not well understood, moisture retention, friction, and sustained pressure are known to contribute to tissue breakdown. 13 Pressure (force distributed over area) at the mask-skin interface is not easily characterized in the neonatal population due to the inherent space constraints and anticipated low pressure ranges (< 25 mm Hg 14 ) that push the limits of conventional contact pressure sensors. Previous studies using varying nasal interfaces, study populations or simulators, and pressure measurement methodologies have resulted in pressures ranging from 6.5–158.3 mm Hg. 14 –19 As such, these studies vary greatly in repeatable implementation of contact pressure sensors and standardization of equipment such that the results are not generalizable across all commercially available CPAP interface systems or populations.
Poorly fitting masks have driven researchers to pursue custom masks, other studies have determined that improperly or non-ideally positioned CPAP interfaces contribute highly to nasal injury and can be improved by quality improvement efforts and regular NIV training. 12,20 –23 In this preliminary study, the interface pressure between a commercially available nasal CPAP interface and a 24-week GA neonatal simulator was characterized at the philtrum, nasal bridge, and forehead along the nasal mask interface. The objective of this study was to quantify the pressure exerted on the face of a neonatal simulator by a CPAP nasal mask with bubble CPAP use and to determine potential variability in the implementation of CPAP components and fixation techniques via image documentation. Taken together, the results will be utilized to develop strategies for reducing interface pressure while maintaining a robust seal.
QUICK LOOK
Current knowledge
Nasal masks are common interfaces to deliver nasal CPAP as they are perceived to be effective and less injurious. However, nasal masks have been demonstrated through in vitro testing to exert substantial pressures ranging from 6.5–158.3 mm Hg on the contacting skin, leading to pressure injury.
What this paper contributes to our knowledge
This paper describes the use of a pressure sensor that was modified and calibrated for use in contact pressure measurements of CPAP nasal masks and the underlying skin. The pressure exerted by a mask interface on a high-fidelity neonatal simulator at a CPAP of 6 cm H2O and 8 L/min of flow was quantified. Pressures ranged from 12–377 mm Hg, with wide variability in the placement and securement of the CPAP interface across clinical staff. Small movements in the nasal mask are associated with large pressure changes on the skin. Noninvasive ventilation (NIV) application and fixation technique training will reduce NIV-associated pressure injury.
Methods
Sensor manufacturing and calibration
A clinical research–grade Millar 1F (0.33-mm catheter) micro-electromechanical system (MEMS) Connect (Millar, Houston, Texas) sensor (5 μV/V/mm Hg sensitivity) was modified by embedding it in a thin (1.3 mm) layer of Dragon Skin 10 MEDIUM silicone (Smooth-On, Macungie, Pennsylvania) using a methodology derived from Motie et al 24 and Mehta et al. 25 Briefly, the MEMS sensor is composed of a sensing diaphragm recessed along the length of a 1F catheter (Fig. 1A). The recession necessitates silicone embedding such that the pressure is translated continuously to the sensing diaphragm (Fig. 1B). The sensor was oriented with sensing diaphragm facing downward on a transparent polystyrene plate and secured with tape. Positioning was confirmed with digital stereoscope imaging (AmScope SM-4TZ-144A Stereo Microscope, AmScope 18MP USB 3.0 Digital Camera [MU1803-HS-CK]; AmScope, Irvine, California). The silicone was prepared according to the manufacturer’s specifications, and a small amount (∼2 g) was deposited around the sensor such that the sensor was covered on all sides. The silicone was allowed to cure at room temperature for 5 h. The process was repeated with the sensor diaphragm positioned upward. The sensor was removed from the plate, and the residual silicone was trimmed to approximately a diameter of 5 mm (Supplementary Figure S1). This process ensured that the sensor pad was flat on both sides in the direction of the sensing diaphragm.
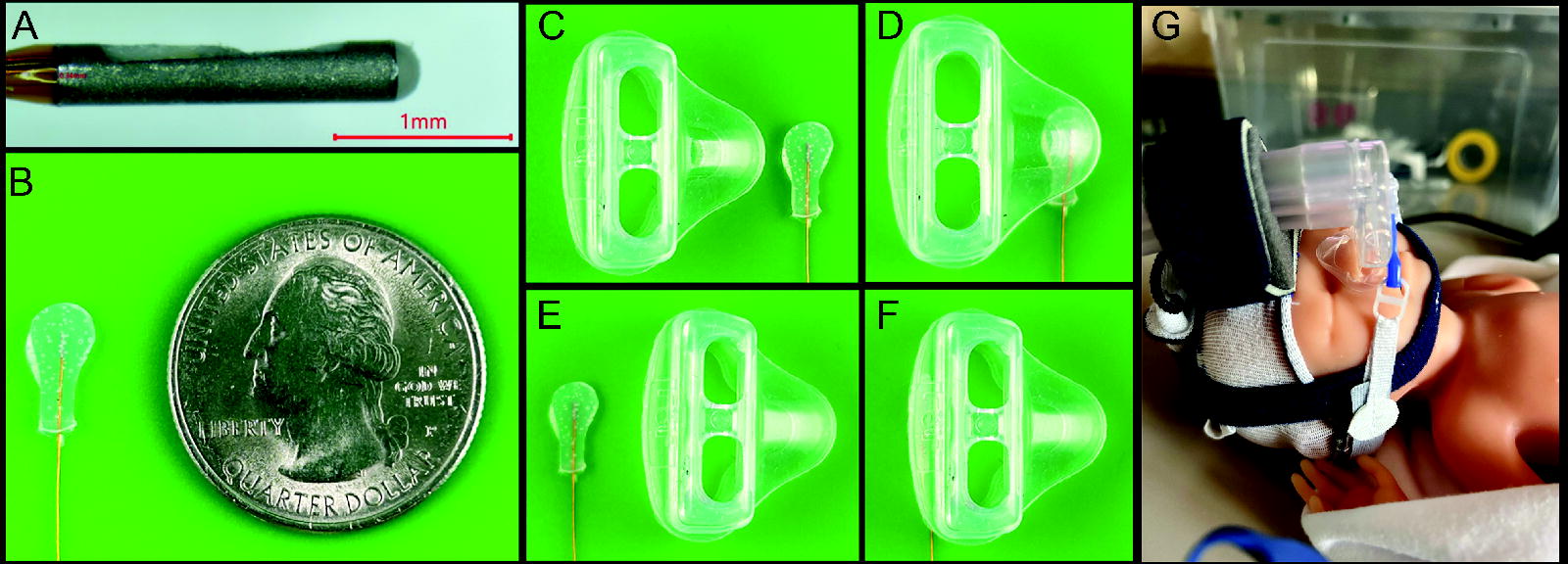
Millar 1F micro-electromechanical system Connect sensor
A National Instruments data acquisition unit (myDAQ, National Instruments, Austin, Texas), with a voltage sensing range of ± 10 V, and accompanying LabVIEW software (National Instruments) composed the data acquisition system. The sensor was connected to analog input zero (ai0+, ai0−), and a + 5V excitation voltage (resulting in a 25 μV/mm Hg sensor sensitivity) was supplied via the myDAQ unit (Supplementary Figure S2). Data were collected continuously at a rate of 1 kHz and filtered with a rectangular moving average filter (no. = 1,000).
The custom sensor was calibrated using a water column that was fabricated out of 9.525-mm (3/8 inch) inner diameter clear vinyl tubing connected to a corresponding shut-off valve. The tubing was marked with 2.54-cm (1 inch) increments, and the tube was level mounted vertically to a wall. A 2-g sinker mass was taped to the sensor wire, and the sensor was submerged into the water column at 15.24-cm (6 inch) increments with continuous data collection. Calibrations pre and post embedding in silicone confirmed the need to calibrate each sensor post–silicone embedding (Supplementary Figure S3). The calibration was repeated six (no. = 6) times per sensor after embedding in silicone and prior to each new data collection activity. A best-fit line was determined resulting in a unique pressure calibration for each sensor.
Sensor repeatability was within 7% across the entire range of the water column, with repeatability improving to ∼2% with increasing pressure. Calibrations were limited to a water column depth of 106.7 cm (42 inches) (equivalent to 78 mm Hg of pressure) due to the non-modifiable sensor wire length. Calibrations within the 78 mm Hg range were linear. As such, a linear calibration was assumed throughout the entire measurement range. This is a reasonable assumption as the unembedded sensor is rated for a linear 5μV/V/mm Hg sensitivity for a pressure range of −50 to +300 mm Hg. However, even pressures higher than 300 mm Hg are well within the sensing voltage range (±10 V) of the measurement system. At an excitation voltage of +5 V, the sensor sensitivity becomes approximately 25 μV/mm Hg (depending on the silicone embedding) such that for every 1 mm Hg change in input pressure the output voltage will change by 25 μV. For pressures modestly higher than the manufacturer’s rated limit of 300 mm Hg, the sensor output voltage would be well within the voltage sensing range (±10 V) of the measurement system.
Test setup, pressure measurements, and analysis
This prospective observational study was completed over a five-week period from January 2024–February 2024 within the University of Cincinnati Medical Center (UCMC) NICU. A high-fidelity, Premie HAL S108.100 24-Week neonatal simulator (manikin) (Gaumard Scientific, Country Walk, Florida) was utilized for all experiments. The pressure measurements were made on a manikin, not on human infants. The personnel who applied the mask were performing their normal job. The investigator did not use, study, analyze, or generate identifiable private information or identifiable biospecimens from these personnel. Institutional review board approval was not required under 45 Code of Federal Regulations 46. The CPAP equipment investigated was a Bubble CPAP generator and a FlexiTrunk interface (Fisher & Paykel Healthcare, Aukland, New Zealand) with the accompanying components, including the bonnet, nasal mask, and chin strap. A small nasal mask, small hat (17–22 cm), and small chin strap (20–26 cm) were selected as appropriately sized components for the neonatal simulator by a lead respiratory therapist prior to data collection. The FlexiTrunk, chin strap, and hook and loop extenders were reused. A new bonnet and nasal mask were used for each trial.
Thirteen data sets were collected after CPAP placement by UCMC NICU staff composed of registered nurses (n = 6) and respiratory therapists (n = 7). The neonatal simulator was positioned on a NICU counter surface conveniently accessible to the NICU staff, and the CPAP generator was set to 6 cm H2O at 8 L/min of flow without humidification. The components were placed near the neonatal simulator before each trial. The NICU staff were asked to place CPAP on the neonatal simulator in the same exact way that they would a patient, such that an adequate seal was achieved. The process was timed from start to finish of CPAP application. Once the staff determined that the fit was acceptable by using bubble feedback, visual inspection, and experience, interface pressure measurements of the philtrum, nasal bridge, and the forehead (under the foam block of the FlexiTrunk interface) were taken in triplicate (3 measurements at each location except in the case of sensor malfunction) by sliding the sensor between the neonatal simulator and interface component at each location. Triplicate measurements at each location ensured that the custom-built sensor captured data for each trial. Images of the neonatal simulator were taken pre– and post–pressure measurement using a 12 MP iPhone camera (iPhone 14, Apple, Cupertino, California). All interface components were removed and disassembled between trials.
The sensor-specific calibration curves were used to convert voltage measurements to corresponding pressure values. All statistical analyses were completed in Minitab (Minitab, State College, Pennsylvania). Descriptive statistics were generated both within trial triplicates and across all trials. A 2-sample paired t test of the mean was utilized at each location to determine if the first pressure measurement was different than the last of each triplicate. The location-specific difference in pressures was analyzed with a one-way, 2-tailed analysis of variance (ANOVA) and a Tukey post hoc test at a significance level of P < .05.
Results
The experiments resulted in 102 pressure measurements in total after removing those affected by sensor malfunction. The pressures across all sites (philtrum, nasal bridge, and forehead) ranged from 12.3–377.3 mm Hg. The mean (SD) pressure (mm Hg) at the philtrum, nasal bridge, and forehead was 173.9 (101.3) mm Hg, 67.8 (28.9) mm Hg, and 79.0 (36.9) mm Hg, respectively. A one-way ANOVA determined that there was a statistically significant difference in pressure between at least 2 groups (F2,99 = 27.8, P < .001). The Tukey post hoc test found that the mean pressure (mm Hg) was significantly different between the philtrum and the nasal bridge (P < .001, [CI 68.9–143.4]) and between the philtrum and the forehead (P < .001, [CI 57.7–132.2]). There was no statistically significant difference between the forehead and the nasal bridge (P = .75) (Fig. 2). The time (min:s) required to place CPAP ranged from 1:54–6:45 (mean 4:39, SD 01:15).
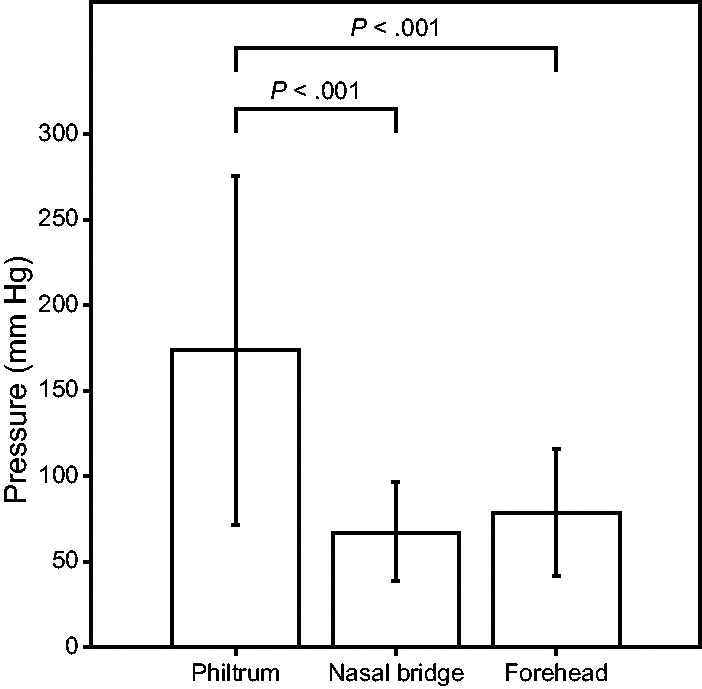
Mean and SD of the measured interface pressure (mm Hg) pooled across each site. Mean (SD, 95% CI): philtrum 173.9 (101.3 [152.0–195.9]), nasal bridge 67.8 (28.9 [45.9–89.7]), and forehead 79.0 (36.9 [57.1–101.0]). Location-specific differences were analyzed with a one-way, 2-tailed analysis of variance and a Tukey post hoc test.
Photographs of the resulting CPAP placements demonstrated a markedly wide variation in the fixation technique and implementation of CPAP interface components, including bonnet placement (to the nape and eyebrow line or intermediate placement), trunk angle (level or angled), mask compression (mask shape maintained or compressed), and use of hook and loop extenders (Fig. 3) (Supplementary Figures S4, S5, S6, S7, and S8). A bubble-verified seal was achieved in each case, but the level of vigorous bubble varied. The mean interface pressure recorded at the philtrum, nasal bridge, and forehead decreased from the first to the third measurement of the triplicate but was not statistically significant at a significance level of .05 (philtrum t 22 = 0.02, P = .98; nasal bridge t 22 = 0.57, P = .58; and forehead t 22 = 0.35, P = .73) (Fig. 4).
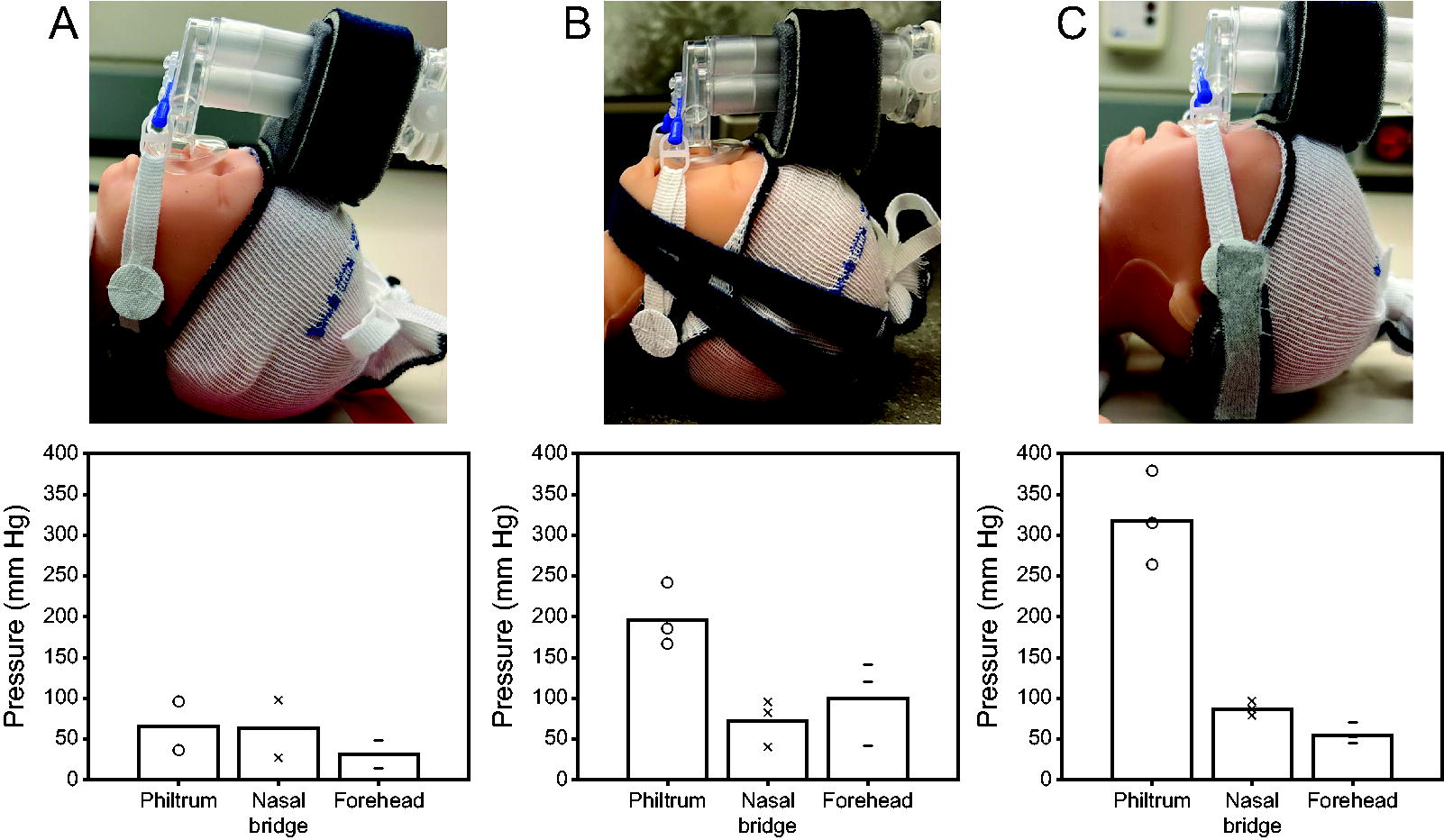
Depiction of the inter-individual variability exhibited, represented by nasal mask, trunk, and bonnet placement and resultant pressure measurements corresponding to the
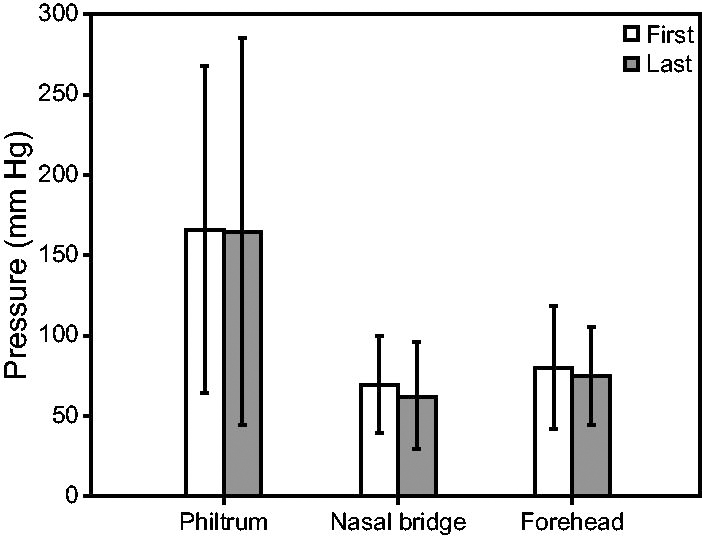
The pooled mean interface pressure recorded at the philtrum, nasal bridge, and forehead from the first to the third measurement of the triplicate decreased but were not statistically significant at a significance level of 0.05 (philtrum t 22 = 0.02, P = .98; nasal bridge t 22 = 0.57, P = .58; and forehead t 22 = 0.35, P = .73).
Discussion
The objective of this study was to characterize the interface pressure between a CPAP nasal mask and a neonatal simulator (manikin) to evaluate the variation that occurs due to inter-individual variability in implementation of the CPAP components. A commercially available mask was fitted to the neonatal simulator by 13 unique UCMC NICU staff. Mask interface pressures were measured using a custom-built, clinical research–grade sensor modified for use in contact pressure measurements. The study demonstrates that the nasal mask exerts substantial pressures on the philtrum and that the pressures are widely variable depending on CPAP fixation technique. To the authors’ knowledge, this is the first study that has developed a pressure sensor intended for contact pressure measurements in the neonatal setting and quantified inter-individual variability previously observed in CPAP placement across clinical staff.
While the exact etiology and evolution of pressure injury are not yet known, it is thought to be related in large part to capillary occlusion. External pressure that exceeds the arterial diastolic blood pressure has been hypothesized to induce an injury. 26 Diastolic blood pressures in the neonatal population have been reported as 24–57 mm Hg on day 1 of life. 27,28 The mean pressure measured at each site of the CPAP nasal mask exceeds this pressure threshold (philtrum 173.9 mm Hg, nasal bridge 67.8 mm Hg, and forehead 79.0 mm Hg). The pressures identified in this study exhibited overlapping range (Fig. 5), markedly higher maximum pressures, and higher pressures at the philtrum compared to those reported in prior studies. 14 –19
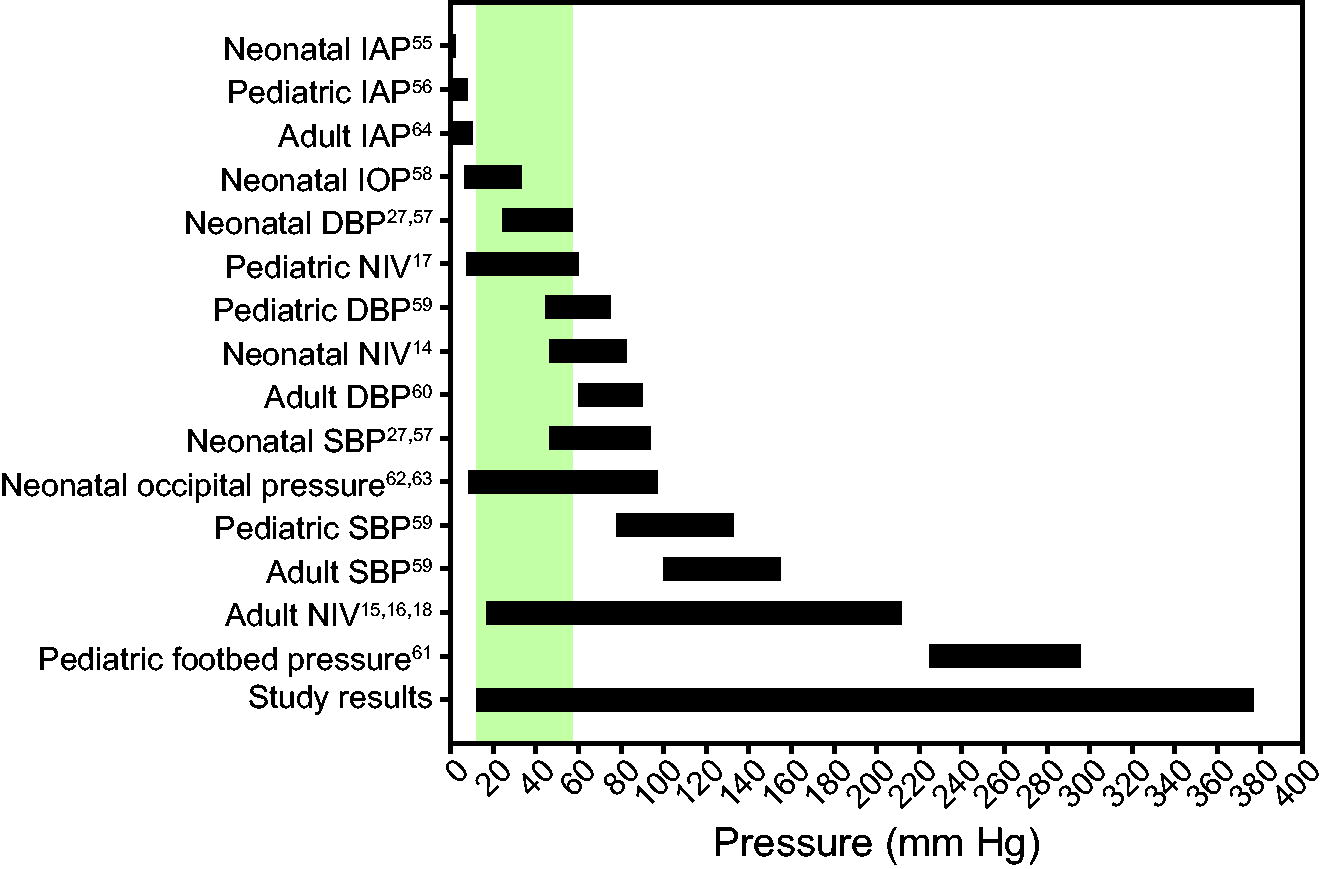
Ranges reported for various physiologic pressure measurements in neonatal, pediatric, and adult populations with associated reference numbers. Treated and untreated adult blood pressures were reported in the range. Large pressure ranges for noninvasive ventilation measurement in the literature reflect variations due to measurement methodology, equipment type, and human or simulator use. Large pressure ranges from the present study results reflect inter-individual differences in fixation technique. The shaded region denotes the overlap between the study findings and neonatal diastolic blood pressure (hypothesized threshold for pressure injury), indicating the ability to achieve a desired nasal mask CPAP pressure while minimizing the potential for pressure injury. IAP, intra-abdominal pressure; IOP, intraocular pressure; DBP, diastolic blood pressure; NIV, noninvasive ventilation; SBP, systolic blood pressure.
Contact pressure measurement is a decades-old engineering challenge that despite significant improvements in technology is still met with limitations. 29 –33 Accurate and repeatable contact pressure measurements are highly dependent on a uniform pressure distribution across the sensing element, a flat sensing surface, contact surface materials, and consistent sensor calibration. 32 –39 Commercially available pressure transducers and force-sensitive resistors (FSRs) are limited by sensor size such that the sensor contact area is greater than that of a neonatal nasal mask, therefore reducing the ability to maintain a uniform pressure distribution across the sensor. The modified MEMS sensor utilized for these pressure measurements was smaller than the mask contact area (enforcing a uniform pressure distribution across the sensor pad) (Fig. 1), flat, and was calibrated before each data collection activity.
When the contacting surface is smaller than the sensor, this may result in an overestimate of pressure. Conversely, when the contacting surface is larger than the sensor, this may result in an underestimate of pressure. 34,40 Pressure can be artificially augmented by the thickness of the sensor causing a stress concentration in the contact surface being measured. 38 Further, embedding a sensor in silicone changes the properties of a sensor and requires sensor-specific calibration 24,25,34,36 (Supplementary Figure S3). Of the studies cited here that previously measured CPAP mask interface pressure, three out of six of the pressure sensors utilized were larger than the contacting surface (presumably underestimated the contact pressure), two studies utilized commercially available pressure mapping handles not intended for small surface areas, and one study embedded FSR sensors in silicone without recalibrating. To minimize sources of measurement error, we utilized a 1F sensor, with a sensor pad smaller than the contact surface, ensured that the silicone was no more than 1.3 mm in thickness (the minimum required for embedding) to minimize the generation of stress concentrations during data acquisition, and calibrated each sensor post–silicone embedding.
During pressure measurements, consistent with previous studies, 16 it was immediately evident that even small differences in mask positioning caused by fixation technique or movements (< 2 mm) of the interface components or neonatal simulator resulted in large changes in pressure (Fig. 3). Movements of the interface components or neonatal simulator were due to the process of repositioning the sensor but may be similar to movements that naturally occur during routine care times, infant repositioning, or normal infant movement. While statistical significance was not achieved, the mean pressure of the first measurement was different than the mean pressure at the third measurement (Fig. 4). This suggests that further studies on interface pressure changes throughout infant repositioning and movement may be necessary. While a minimum pressure mask fit may be achieved during supine infant care, this may not be maintained during repositioning or normal infant movement.
Similarly, mask placements that resulted in higher pressures had less variation in measurements across triplicates, suggesting that a lower pressure mask fit was susceptible to movement and larger shifts in pressure. This is an important finding because it highlights that even if the staff achieve an ideal fit and seal with minimal pressure as defined by the manufacturer guidelines small movements may immediately break the seal. Furthermore, we found a markedly higher pressure at the philtrum (173.9 mm Hg) than at the nasal bridge (67.8 mm Hg) or forehead (79.0 mm Hg), whereas previous studies reported pressures consistently higher at the nasal bridge. 14 –19 This finding reflects the variation in pressure distribution due to the equipment brand and design. Since the CPAP generator interface devices varied across previously reported studies, future studies should include multiple brands of CPAP generator interface devices to determine device-specific pressure distributions.
Several studies have noted anecdotally the importance of individual clinical staff experience and training with mask fitting for the reduction of pressure injury and improvement of NIV outcomes. 12,20 –23 The inter-individual differences in pressure measurements exhibited in the data quantitatively demonstrate the effect of individual differences in nasal mask placement due to fixation technique. By visual inspection (Fig. 3), hat placement, FlexiTrunk angle, nasal mask compression, and use of hook and loop extenders were notably variable. Use of chin strap was variable; however, the neonatal simulator chin was stiff enough such that the chin strap was not always effective, and a seal was checked by manually holding the chin shut. Fixation techniques have a substantial impact on the interface pressures required to achieve a seal. The visual variations in fixation technique correspond to differences in pressure. The biggest differences may be appreciated when comparing the minimum pressure (Fig. 3A) and the maximum pressure (Fig. 3C). In Fig. 3A, the bonnet is positioned per manufacturer guidelines at the nape and along the eyebrow line, the nasal mask is not overly compressed, and the FlexiTrunk interface is level. In Fig. 3C, the bonnet reaches the eyebrow line but does not reach the nape, and the nasal mask is compressed nearly flat. At the philtrum, these differences in fixation technique caused nearly a 5-fold increase in pressure.
This suggests that there are opportunities for improving mask fixation techniques through education. Clinical bedside markers for an ideal (minimum pressure required for a robust seal) mask placement should be determined and incorporated into the training efforts to reduce NIV-associated pressure injury. The bonnet acts as the foundation of the interface and if positioned improperly can have substantial effects on the interface pressures (Fig. 3C vs Fig. 3A). As such, improved bonnet sizing and material (reduced stretch [Supplementary Figure S9]) may also improve the ability of staff to achieve and maintain a robust seal with a minimum pressure. Other NICUs have also found the bonnet to be similarly limiting, which has led to the development of innovative bonnets (eg, versatile sizing, no stretch material) and accessories that are compatible with all major CPAP generators 41 and used in hospital systems across Australia. 42 Hence, testing alternative CPAP interface systems will inform redesign prioritization and efforts.
Nasal interfaces are considered a primary limitation to the effective use of NIV 43 as they are associated with increased risk of nasal trauma. Custom masks in older children and adults have been demonstrated to reduce the peak pressures exerted by nasal masks on the facial skin and improve comfort and adherence but are largely intended for patients with craniofacial anomalies or those undergoing long-term CPAP, such as in the treatment of obstructive sleep apnea. 14,19,44 –52 Lengthy workflow times for the production of custom masks do not translate well for use in the NICU. 18,53 Furthermore, head molding, facial anatomical changes, and facial edema require frequent changes of custom masks.
Improved NIV interfaces can greatly increase the effectiveness of early CPAP initiation. 54 Further research into the improvement of nasal interfaces is necessary. The benchtop studies presented here have validated a method of measuring interface pressure of nasal masks in the neonatal population using a neonatal simulator, a necessary first step that motivates future studies on neonatal human subjects. The high-fidelity neonatal simulator provides a consistent method of evaluating the variability in pressure experienced due to application and fixation techniques. The limitations associated with the simulator, such as simulated skin material, lack of movement, and lack of anatomical variability, motivate the need to validate the findings on human infants. Validating the findings on human infants will enable observational studies to evaluate the relationship between interface pressure and pressure injury.
Conclusions
The modified MEMS pressure sensor found interface pressures at the NIV nasal mask interface to be substantially higher at the philtrum, nasal bridge, and forehead than those previously reported. 14 –19 The mean pressure at the philtrum was significantly higher (174 mm Hg) than both the nasal bridge (67.8 mm Hg) and forehead (79.0 mm Hg). Quantitative pressure values and interface placement (from images) varied among individuals. Specific training on NIV interface fixation may minimize pressure-associated injury until design improvements can be realized.
Footnotes
Acknowledgments
The authors would like to acknowledge the assistance and invaluable feedback from the UCMC NICU staff.
Authors' Contributions
Drs Hoilett and Nauman contributed equally as senior authors.
Author Disclosure Statement
Dr Hoilett is a cofounder of Predictive Wear, Inc; a co-inventor on patents US11672288B2 and US12063993B2 issued to Predictive Wear, Inc; a co-inventor on patents US20230263248A1 and US20210052026A1 pending to Predictive Wear, Inc; a co-inventor on patents US11737708B2, US11633152B2, US12029548B2, US11737708B2, US10786201B2, US11090649B2, US11628434B2, and US11529626B2 issued to Purdue Research Foundation; a co-inventor on patents WO2020009798A1, US20240000384A1, and WO2017184665A1 pending to Purdue Research Foundation; and a co-inventor on a provisional application for a biometric sensor vest filed by the University of Cincinnati. Drs Zakrajsek, Hoilett, and Nauman are co-inventors on a provisional application for a method of creating hydrocolloid dressings filed by the University of Cincinnati. Dr Nauman is a co-founder of Advanced Regenerative Technologies; a co-founder of IFBattery; a co-inventor on patents US8,702,809, US8,764,688, US9,155,607, US9,364,584, US10,857,006, US10,898,331, and US10,948,489 issued to Purdue Research Foundation; a co-inventor on patent US10,665,868 issued to Purdue Research Foundation and IFBattery; a co-inventor on patent 10,675,512 issued to the Indiana Pacers of the National Basketball Association; a co-inventor on patent US10,966,816 issued to Sparta Therapeutics; and a co-inventor on a pending US patent application: US-20210013531-A1. Drs Nauman and Zakrajsek are co-inventors on patents US9,056,983, US9,394,959, US9,839,250, and US10,183,423 issued to Purdue Research Foundation. The remaining authors have disclosed no conflicts of interest.
Supplementary Material
Supplementary Figure S1
Supplementary Figure S2
Supplementary Figure S3
Supplementary Figure S4
Supplementary Figure S5
Supplementary Figure S6
Supplementary Figure S7
Supplementary Figure S8
Supplementary Figure S9
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
